MAJOR ARTICLE. identifiable cause of purulent skin infections, that is, abscess and purulent cellulitis [1, 2]. The frequency of
|
|
|
- Audra O’Connor’
- 6 years ago
- Views:
Transcription
1 MAJOR ARTICLE Clinical Trial: Comparative Effectiveness of Cephalexin Plus Trimethoprim- Sulfamethoxazole Versus Cephalexin Alone for Treatment of Uncomplicated Cellulitis: A Randomized Controlled Trial Daniel J. Pallin, 1,2 William D. Binder, 3 Matthew B. Allen, 1,4 Molly Lederman, 1,5 Siddharth Parmar, 1 Michael R. Filbin, 3 David C. Hooper, 6 and Carlos A. Camargo Jr 3 1 Department of Emergency Medicine, Brigham and Women s Hospital, 2 Division of Emergency Medicine, Boston Children s Hospital, and 3 Department of Emergency Medicine, Massachusetts General Hospital, Boston; 4 Perelman School of Medicine at the University of Pennsylvania, Philadelphia; 5 Department of Pediatrics, and 6 Division of Infectious Diseases, Department of Medicine, Massachusetts General Hospital, Boston (See the Editorial Commentary by Chambers on pages ) Background. Community-associated methicillin-resistant S. aureus (CA-MRSA) is the most common organism isolated from purulent skin infections. Antibiotics are usually not beneficial for skin abscess, and national guidelines do not recommend CA-MRSA coverage for cellulitis, except purulent cellulitis, which is uncommon. Despite this, antibiotics targeting CA-MRSA are prescribed commonly and increasingly for skin infections, perhaps due, in part, to lack of experimental evidence among cellulitis patients. We test the hypothesis that antibiotics targeting CA- MRSA are beneficial in the treatment of cellulitis. Methods. We performed a randomized, multicenter, double-blind, placebo-controlled trial from 2007 to We enrolled patients with cellulitis, no abscesses, symptoms for <1 week, and no diabetes, immunosuppression, peripheral vascular disease, or hospitalization (clinicaltrials.gov NCT ). All participants received cephalexin. Additionally, each was randomized to trimethoprim-sulfamethoxazole or placebo. We provided 14 days of antibiotics and instructed participants to continue therapy for 1 week, then stop 3 days after they felt the infection to be cured. Our main outcome measure was the risk difference for treatment success, determined in person at 2 weeks, with telephone and medical record confirmation at 1 month. Results. We enrolled 153 participants, and 146 had outcome data for intent-to-treat analysis. Median age was 29, range Of intervention participants, 62/73 (85%) were cured versus 60/73 controls (82%), a risk difference of 2.7% (95% confidence interval, 9.3% to 15%; P =.66). No covariates predicted treatment response, including nasal MRSA colonization and purulence at enrollment. Conclusions. Among patients diagnosed with cellulitis without abscess, the addition of trimethoprim-sulfamethoxazole to cephalexin did not improve outcomes overall or by subgroup. Clinical Trials Registration. NCT Keywords. cellulitis; community-associated methicillin-resistant Staphylococcus aureus; comparative effectiveness; trimethoprim-sulfamethoxazole; cephalexin. Community-associated methicillin-resistant Staphylococcus aureus (CA-MRSA) is the most common identifiable cause of purulent skin infections, that is, abscess and purulent cellulitis [1, 2]. The frequency of Received 5 December 2012; accepted 22 January 2013; electronically published 1 March Correspondence: Daniel J. Pallin, 75 Francis St, Neville House 304-D, Boston, MA (dpallin@partners.org). Clinical Infectious Diseases 2013;56(12): The Author Published by Oxford University Press on behalf of the Infectious Diseases Society of America. All rights reserved. For Permissions, please journals.permissions@oup.com. DOI: /cid/cit CID 2013:56 (15 June) Pallin et al
2 Table 1. Inclusion and Exclusion Criteria and Drug Dosing I. Inclusion Criteria A. Must have cellulitis as defined here: 1. Definition A (preferred definition): Recent onset of soft tissue erythema, considered by the treating clinician to be bacterial in origin, and associated with signs of infection that include at least 2 of the following: pain, swelling, warmth, fever, lymphangitis, induration, or ulceration. 2. Definition B (only for darkly pigmented patients who cannot use definition A): Recent onset of soft tissue color change, pain, or swelling, considered by the treating clinician to be bacterial in origin, and at least 1 of the following: warmth, fever, induration, or ulceration B. Clinical (nonresearch) attending physician agrees with treatment with cephalexin until 3 d after all symptoms gone, using our weight-based dosing C. Responsible clinical attending physician comfortable with adding trimethoprim-sulfamethoxazole vs placebo to the above D. Patient understands the study and signs written informed consent E. Patient agrees to drink at least 1 L of fluid per day F. Patient will commit to all follow-up appointments II. Exclusion Criteria A. Age <12 mo or weight <15 kg B. Current skin infection has already been treated C. Allergy to sulfa drugs D. History of severe allergic reaction to penicillin (defined as anaphylactoid reaction, angioedema, bronchospasm) E. Current use of any antibiotic (other than topicals) F. Diabetes mellitus G. Cellulitis complicated by underlying peripheral vascular disease H. Renal insufficiency, defined as patient report, clinical suspicion, or creatinine >1.3 or estimated glomerular filtration rate < 60 on the last available set of chemistry results in our computer system I. Hospital admission required J. Presence of >1 cc of purulent discharge at any time K. Cellulitis involving an indwelling vascular, enteric, or urinary catheter L. Immunocompromise of any etiology M. Pregnancy N. Breast feeding O. Facial cellulitis (infection is above the clavicles) P. Cellulitis associated with marine or freshwater injury or animal or human bite (insect bites not excluded) Q. History of glucose-6-phosphate dehydrogenase deficiency R. Taking Coumadin (warfarin), methotrexate, cisapride, phenytoin (Dilantin), digoxin, or dofetilide S. Known megaloblastic anemia due to folate deficiency. III. Antibiotic dosing Weight Cephalexin Trimethoprim-sulfamethoxazole (mg trimethoprim) Children <30 kg: kg: 300 mg 4 times daily 40/200 mg qid kg: 400 mg 4 times daily 60/300 mg qid kg: 500 mg 4 times daily 72/360 mg qid Adults and children 30 kg: <60 kg: 500 mg 4 times daily 80/400 mg qid kg: 1000 mg 3 times daily 160/800 mg tid >80 kg: 1000 mg 4 times daily 160/800 mg qid skin infections increased with its advent, implying that CA-MRSA caused an epidemic of new disease, rather than merely substituting for methicillin-sensitive S. aureus in infections that would have occurred anyway [3]. Multiple studies have found that patients with CA-MRSA abscesses do not benefit from antibiotics; the mainstay of treatment being incision and drainage [4 6]. For most cases of cellulitis, Infectious Diseases Society of America (IDSA) guidelines recommend antibiotics that target streptococci, not CA-MRSA [7]. They do recommend antibiotics that target CA-MRSA for purulent cellulitis, but this accounts for only 8% of purulent skin infections and a smaller proportion of all skin infections [2]. RCT of CA-MRSA Coverage in Cellulitis CID 2013:56 (15 June) 1755
3 Figure 1. CONSORT Diagram. In the years preceding publication of the guidelines, antibiotics that target CA-MRSA were prescribed increasingly for skin infections [3, 8, 9]. This may reflect the fact that limited evidence was available to both practicing clinicians and guideline authors regarding cellulitis treatment. To date, no experimental trials have been published, and microbiological data relating to cellulitis are usually not obtainable [10]. We conducted a pragmatic comparative effectiveness study, hypothesizing that cephalexin plus trimethoprim-sulfamethoxazole would be more effective than cephalexin alone in the treatment of cellulitis [11]. METHODS Trial Design We conducted a double-blind, randomized, placebo-controlled trial from June 2007 through December 2011, with 1:1 parallel group allocation. Potential participants were patients registered for care in 1 of 3 emergency departments located in an area endemic for CA-MRSA [12]. Cellulitis was diagnosed during routine clinical care by attending physicians, board-certified or board-eligible in emergency medicine or pediatric emergency medicine, using explicit criteria (Table 1). Physician coinvestigators enrolled the participants, with trained research coordinators. Participants The inclusion and exclusion criteria (Table 1) defined a group of generally-healthy patients with uncomplicated cellulitis and no abscess. Pustules <3 mm in maximal diameter were not considered abscesses. Patients with purulent cellulitis or wound infection could be enrolled if <1 cc of pus was observed or reported by the patient. We collected data on enrolled and nonenrolled patients as specified by CONSORT (Figure 1)[13]. Interventions We compared cephalexin plus trimethoprim-sulfamethoxazole to cephalexin plus placebo. For adults, guidelines recommend trimethoprim-sulfamethoxazole doses of 160/800 to 320/1600 mg twice daily; cephalexin is commonly prescribed at 500 mg 4 times daily, though twice-daily dosing has been found effective [7, 14]. We used higher, weight-based doses and matched cephalexin and study drug scheduling (Table 1). Participants were instructed to stop taking the antibiotics 3 days after they believed the infection to be cured, for a minimum of 7 days and a maximum of 14 [15]. We monitored medication adherence via a log filled out by the participants. Outcomes The primary outcome was the risk difference for cure in the intent-to-treat group, defined as resolution of symptoms other 1756 CID 2013:56 (15 June) Pallin et al
4 Table 2. Baseline Characteristics Patient Characteristic All Participants (N = 146) Trimethoprim- Sulfamethoxazole n Placebo n Age (median [range, interquartile range]) 29 (3 74, 23 43) 31 (12 71, 25 47) 27 (3 74, 22 40) Race (%) White 88 (60) 44 (60) 44 (60) Black 24 (16) 13 (18) 11 (15) Asian 3 (2) 1 (1.4) 2 (2.7) Native American 0 (0) 0 (0) 0 (0) Pacific Islander 0 (0) 0 (0) 0 (0) Other 1 (0.68) 0 (0) 1 (1.4) Hispanic 38 (26) 19 (26) 19 (26) Antibiotics in the past year (%) 57 (39) 24 (33) 33 (45) Healthcare worker (%) 31 (21) 21 (29) 10 (14) Past history of skin infection (%) 50 (34) 25 (34) 25 (34) Physical contact with someone with similar infection (%) 10 (6.9) 4 (5.5) 6 (8.2) Ever diagnosed with MRSA (%) 3 (2.1) 2 (2.7) 1 (1.4) Physical contact with someone with MRSA (%) 17 (12) 10 (14) 7 (9.7) Participated in any of the following sports in the past year: football, hockey, wrestling, rugby, boxing, martial arts, dance (%) 43 (30) 19 (26) 24 (33) Homeless in the past year (%) 9 (6.2) 3 (4.1) 6 (8.2) Ever diagnosed with human immunodeficiency virus (%) 0 (0) 0 (0) 0 (0) Ever used IV drugs (%) 4 (2.7) 0 (0) 4 (5.5) Physical contact in past year with someone who uses IV 14 (9.6) 5 (6.9) 9 (12) drugs (%) Ever been in jail (%) 24 (16) 12 (16) 12 (16) Physical contact in past year with person who has ever 30 (21) 13 (18) 17 (23) been in jail (%) Stayed overnight in the hospital in the past year (%) 28 (19) 17 (23) 11 (15) Live in any long-term care facility or care institution (%) 2 (1.4) 1 (1.4) 1 (1.4) Indwelling line (peripherally inserted central catheter, a 2 (1.4) 1 (1.4) 1 (1.4) Hickman, or a Porta-Cath) (%) Physical contact in past year with someone with 12 (8.2) 8 (11) 4 (5.5) indwelling line (%) Adult patient with child in daycare or patient is a child in 16 (11) 8 (11) 8 (11) daycare (%) Homosexual relations in past year (%) 8 (5.5) 6 (8.2) 2 (2.7) History of splenectomy (%) 2 (1.4) 2 (2.7) 0 (0) Ever diagnosed with eczema (%) 9 (6.2) 3 (4.1) 6 (8.2) Nasal MRSA colonization (data not available at 7 (4.9) 4 (5.6) 3 (4.2) enrollment) (%) Infection Characteristics at Enrollment (%) Purulence 19 (13) 8 (11) 11 (15) Warmth 136 (94) 69 (96) 67 (92) Fever 19 (13) 9 (12) 10 (14) Lymphangitis 16 (12) 8 (13) 8 (12) Induration 65 (46) 33 (47) 32 (44) Ulceration 18 (13) 11 (15) 7 (10) Pain 138 (95) 69 (95) 69 (95) Edema 106 (74) 58 (81) 48 (68) RCT of CA-MRSA Coverage in Cellulitis CID 2013:56 (15 June) 1757
5 Table 2 continued. Patient Characteristic All Participants (N = 146) Trimethoprim- Sulfamethoxazole n Placebo n Portal of entry (any break in the skin preceding the 86 (59) 41 (56) 45 (62) infection) Reported insect bite 34 (23) 15 (21) 19 (26) Reported spider bite 8 (5.5) 2 (2.7) 6 (8.2) Varying Events During the Trial Received up to 24 h of intravenous therapy at index 40 (27) 18 (25) 22 (30) visit Did not attend in-person follow-up visit 19 (13) 7 (9.6) 12 (16) Telephone plus medical record review follow-up 15 (11) 6 (8) 9 (13) Medical record review follow-up only 4 (2.7) 1 (1.4) 3 (4.1) Abbreviations: IV, intravenous; MRSA, methicillin-resistant Staphylococcus aureus. than slight residual erythema or edema. This was determined by in-person examination at 12 ± 2 days by any available emergency department physician. We also contacted participants and reviewed medical records at 1 month to be sure that no delayed outcomes were missed. The final determination of cure or failure was made at 1 month and included all available data. Failure was defined as subsequent hospitalization for the same infection, change in antibiotics for any reason, surgical or needle drainage of an abscess, or recurrence of infection within 30 days. For participants who did not attend the inperson determination of cure visit, we used telephone followup at 2 weeks and 1 month and medical record review to determine outcome. The secondary outcome was the association of nasal MRSA colonization at enrollment with treatment response (CHRO- Magar, Becton Dickinson, Sparks, MD). We examined a number of other possible predictors of response to trimethoprim-sulfamethoxazole (Table 2). These are patient characteristics identified previously as risk factors for CA-MRSA and characteristics of the infection at presentation. These included whether the presence of purulence at enrollment or receipt of initial intravenous therapy could predict treatment response [2]. At the discretion of the treating (nonresearch) physician, some participants received 24 hours of intravenous cefazolin or nafcillin. Simultaneously, they received the oral study drug. They commenced oral cephalexin upon stopping intravenous therapy. We assessed the effect of nonadherence via logistic regression, using treatment assignment and number of nonadherent days to predict outcome. Cellulitis is a clinical diagnosis without proof of a bacterial infection in most cases [10]. Therefore, it was inevitable that some participants would ultimately be found not to have had infectious cellulitis. We determined the final diagnosis using all available information, including subsequent radiographic results, surgical results, and clinical course, and examined this in per-protocol analysis. Sample Size Prior studies of beta lactams for treatment of cellulitis have found failure rates of 13% 18.7% [16, 17]. Our sample size calculations indicated a requirement of 144 participants to achieve 80% power to detect a response rate difference of at least 13%, with 85% responding in the control group and 98% in the intervention group, with 2-sided alpha 0.05 [16, 17]. No data were analyzed until study completion. The trial was stopped because the sample size was achieved. Randomization The research pharmacy generated a randomization sequence with blocks of 4. Medications were put into blinded containers at the time of each enrollment. Pharmacists had no knowledge of clinical characteristics. Clinicians, coordinators, and participants had no access to the randomization sequence and no way to know the contents of each container. Blinding We administered cephalexin as 500-mg capsules and study drug as capsules of trimethoprim-sulfamethoxazole 80/400 mg versus placebo encased in larger opaque gelatin capsules. For children unable to swallow capsules, we administered suspensions of cephalexin 50 mg/cc and trimethoprim-sulfamethoxazole 8 mg/cc trimethoprim + 40 mg/cc of sulfamethoxazole. The placebo contained cherry simple syrup, ora-sweet, ora-plus suspension agent, and cellulose powder CID 2013:56 (15 June) Pallin et al
6 Table 3. Main Results Total Participants: 146 Trimethoprim- Sulfamethoxazole n Placebo n Risk Difference % (95%CI) P Value Cure (no failure by final 62 (85) 60 (82) 2.7 ( 9.3% to 15%).66 follow-up at 30 d) (%) Progression to abscess (%) 5 (6.8) 5 (6.8) 0 ( 8.2% to 8.2%) 1.0 Any adverse event (%) 36 (49) 39 (53) 4.1 ( 20% to 12%).62 Diarrhea (%) 21 (29) 25 (34) 5.5 ( 21% to 10%).48 Nausea (%) 15 (21) 13 (18) 2.7 ( 10% to 16%).67 Vomiting (%) 5 (6.9) 8 (11) 4.1 ( 13% to 5.1%).38 Rash (%) 4 (5.5) 3 (4.1) 1.4 ( 5.6% to 8.3%).70 Pruritus (%) 5 (6.9) 3 (4.1) 2.7 ( 4.6% to 10%).47 Candidiasis (%) 1 (1.4) 3 (4.1) 2.7 ( 8.0% to 2.5%).31 Clostridium difficile colitis (%) 0 (0) 1 (1.4) 1.4 ( 4.0% to 1.3%).32 Other a (%) 3 (4.1) 3 (4.1) 0 ( 6.4% to 6.4%) 1.0 Abbreviation: CI, confidence interval. a Other adverse events included constipation, flatus, heartburn, bloating, and dysphoria. There were no other reported adverse events. Statistical Methods We assessed outcomes according to intent to treat, with the primary outcome being the bivariate risk difference for cure with its 95% confidence interval (CI) and χ 2 testing. To seek evidence of confounding despite randomization, we performed a secondary analysis, using stepwise logistic regression with liberal entry and stay criteria of 0.20 and using all predictors shown in Table 2 as candidates. We also sought to identify whether subgroups responded to the intervention differently, that is, whether there was effect modification. For each characteristic, we constructed a logistic regression model with cure as the dependent variable. The independent variables were intervention, characteristic, and the interaction of the two. We deemed interaction P values of.05 to be significant an inclusive criterion, since we tested many possible interactions. We used SAS 9.2 for all analyses (SAS Institute, Carey, NC). The institutional review boards from the participating hospitals approved the study, and each patient (or parent) provided written informed consent. This trial was registered with Clinicaltrials.gov, record number NCT RESULTS We randomized 153 participants. Four were randomized in error and did not receive the study drug, 1 received 2 doses before it was discovered that he was ineligible, 1 was lost to follow-up, and 1 withdrew voluntarily in the first few days after enrollment, leaving 146 for intent-to-treat analysis. Of these, 15 (11%) did not return for in-person assessment (12 ± 2 days) but were reached by telephone. Four (2.7%) had outcomes determined only by medical record review (Table 2). Clinical cure was achieved in 62 of 73 (85%) intervention participants versus 60 of 73 (82%) control participants (Table 3), for a nonsignificant risk difference of 2.7% (95% CI, 9.3% to 15%; P =.66). Progression to abscess occurred in 5 participants (6.8%) in each group (risk difference 0%; 95% CI, 8.2% to 8.2%; P = 1.0). Of 146 participants, 24 (16%) failed treatment. Each subject could fail treatment for more than 1 reason, and thus reason for failure categories overlap. Reasons for failure included the following: 21 were prescribed additional antibiotics due to clinicians perception of treatment failure, 6 required incision and drainage, and 3 failed due to drug intolerance (2 controls with allergy, 1 intervention patient with diarrhea). Exclusion of the latter 3 did not change the results (risk difference 1.6%; 95 % CI, 10% to 13%; P =.79). We found no evidence of negative confounding, as the main effect remained null despite our liberal search for confounders (see the Methods section). We also found no evidence of effect modification, as none of the covariates could identify a differential response to the intervention for a subgroup of participants. Nasal colonization data were available for 142 of 146 (97%). Of these, 7 (4.9%) were colonized with MRSA, and this was not associated with response to therapy (interaction P =.67). Purulence was present at enrollment in 19 (13%) and did not predict response. The intervention:placebo cure rates were 75%:91% with purulence and 86%:81% without (interaction P =.26). Up to 24 hours of initial intravenous therapy with cefazolin or nafcillin was administered to 40 participants (27%; Table 2); they took oral study drug while receiving the intravenous beta lactam. The intervention:placebo cure rates were 72%:82% with intravenous therapy and 89%:82% without RCT of CA-MRSA Coverage in Cellulitis CID 2013:56 (15 June) 1759
7 (interaction P =.24). Adherence to prescribed antibiotics did not affect the outcome (see the Methods section; adjusted risk difference 1.8%; 95% CI, 14% to 11%; P =.78). Of the 146 intent-to-treat participants, 4 (2.7%) were ultimately found not to have had cellulitis (2 intervention, 2 control). These were included in the intent-to-treat analysis because, in clinical practice, it is difficult to differentiate bacterial cellulitis from other causes of inflammation. Results were unchanged upon per-protocol analysis of the remaining 142, with a nonsignificant risk difference of 4.2% (95% CI, 7.4% to 16%; P =.45). Among the 146 intent-to-treat participants, 75 (51%) had adverse events (Table 3). This was similar in intervention (49%) versus control (53%) groups (risk difference 4.1%; 95% CI, 20% to 12%; P =.62). There was 1 serious adverse event a case of Clostridium difficile colitis in the placebo group. DISCUSSION We observed no benefit from the addition of trimethoprimsulfamethoxazole to cephalexin in the outpatient treatment of cellulitis. This supports the guidelines of the IDSA, which recommend against targeting CA-MRSA for nonpurulent cellulitis [7]. Practice in recent years has not corresponded to these guidelines [3, 8, 9]. One study found that 38% of antibiotic regimens for skin infections treated in US emergency departments in 2005 included agents typically active against CA-MRSA [3]. By 2010, according to data from the National Hospital Ambulatory Medical Care Survey, 74% of all antibiotic regimens prescribed at emergency department visits for skin infections included an agent typically active against CA-MRSA (our own unpublished analysis). This nonconcordance of clinical practice and the IDSA guidelines is probably due to the time period when the guidelines were introduced and also the limited evidence base available to inform practice and guideline development regarding cellulitis. Non-Purulent Cellulitis The guidelines suggest that cellulitis is usually streptococcal, not staphylococcal. However, this cannot be supported by microbiological arguments, because microbiological data are not obtainable for most cases of cellulitis, with blood cultures positive in only 4%, needle aspiration in 29%, and punch biopsy in 18% [10, 18]. This means that >70% of cellulitis cases are not amenable to microbiological diagnosis. Moreover, among cellulitis cases with cultivable bacteria, S. aureus is a common finding [10]. Experimental evidence has also not been available because no trials have been reported, only observational studies [19 23]. These studies have been inconsistent, with 1 retrospective observational study suggesting benefit from antibiotics with activity against CA-MRSA [24]. That study made the nuanced observation that receipt of an inactive agent... before zero time was not associated with later treatment failure. [24] Such subtle caveats emphasize the importance of prospective experimental evidence to justify treatment recommendations. One line of indirect evidence that supports the guidelines is that epidemiological studies have found that CA-MRSA might be associated with abscesses but not cellulitis [19, 25]. Why CA-MRSA might be associated with abscesses but not cellulitis remains a subject of speculation. CA-MRSA produces exotoxins, including Panton-Valentine leukocidin, that have been found to be modulators of neutrophil chemotaxis, apoptosis, and other properties that might result in purulence and necrosis [26 28]. The observation that abscesses, not cellulitis, increased during the spread of CA-MRSA implies that cellulitis and CA-MRSA are not linked [19]. Once again, this provides indirect evidence to support the guideline s recommendation against CA-MRSA coverage for most cases of cellulitis. Cellulitis is poorly understood, despite being so common. It remains a condition that is diagnosed purely on clinical grounds, with help from no objective diagnostic test. When discussed in the medical literature, it is described phenomenologically, with reference not to pathology but rather to predisposing conditions and clinical characteristics [10]. Even a major pathology textbook provides no description of the pathology of this common condition [29]. As discussed above, attempts to prove a bacterial etiology usually fail [10, 18]. Thus, when an epidemic of skin infections followed the emergence of CA- MRSA, clinical equipoise resulted, with nationwide practice variation and surging use of agents active against CA-MRSA among patients with skin infections [3, 8, 9]. Purulent Cellulitis For the uncommon case of purulent cellulitis, the guidelines recommend empirical therapy for CA-MRSA... pending culture results. [7] This recommendation cites a single observational study, which found CA-MRSA to be present in purulent cellulitis. However, that study also found no association of antibiotic susceptibility and outcome [2]. In our study, the presence of purulence at enrollment was not associated with response to trimethoprim-sulfamethoxazole. Abscess Considering abscess, the guidelines recommend antibiotics only for abscesses associated with... severe or extensive disease... rapid progression in presence of associated cellulitis, signs and symptoms of systemic illness, associated comorbidities or immunosuppression, extremes of age, abscess in an area difficult to drain... associated septic phlebitis, and lack of response to incision and drainage alone. The recommendation 1760 CID 2013:56 (15 June) Pallin et al
8 that antibiotics be used in such settings was based solely on expert opinion [7]. The present study is typical of pragmatic trials [11]. One feature of comparative effectiveness research is that its pragmatic approach can produce results that contradict what would be logically predictable from biological knowledge. We are in the midst of an epidemic of skin infections caused by CA-MRSA, but antibiotics targeting this organism have not been found helpful in pragmatic studies. Limitations The chief limitation of our study is intrinsic to most cases of cellulitis: there is no objective way to make an etiologic diagnosis. Our study was also limited to outpatients, including, for example, no patients with cellulitis complicating lymphedema, for whom blood cultures and intravenous antibiotics are recommended [10]. Our study provides no direct information about the management of such complicated skin infections in hospitalized patients, though it does provide support for the general concept that CA-MRSA is not important in cellulitis. Treatment of patients with life-threatening infections such as necrotizing fasciitis is not informed by this study. In this context, it bears mentioning that we avoided the common term skin and soft-tissue infections, in favor of simply, skin infections. Soft tissue infections, which are not skin infections, include such syndromes as pyomyositis and fasciitis and are rare. This study does not inform the treatment of such conditions. We excluded diabetics. We are aware of no evidence that diabetics are at higher risk for CA-MRSA associated skin infections, and thus our results may be generalizable to them. Most episodes of cellulitis in diabetics are treated as in nondiabetics [7]. While diabetic foot infections do require broader coverage, whether they require coverage for CA-MRSA remains unknown [10]. We assessed MRSA colonization nasally. Only 67% of MRSA-colonized skin infection patients have positive nasal swabs, with other common sites of colonization being the axillae, groin, and perineum [30]. Had we also swabbed the axillae and groin, we might have observed an interaction of colonization and treatment response. While CA-MRSA is almost universally susceptible to trimethoprim-sulfamethoxazole in vitro, it is possible that other agents, such as clindamycin and tetracyclines, might be more effective in vivo. Using diaries, participants indicated whether they took all of their medications each day until self-reported cure, and this is analyzed above. However, we did not record the total duration of therapy (ie, we cannot distinguish stopped therapy from a day of partial adherence). This reflects the study s nature as an effectiveness study, rather than an efficacy study, in that the practicing clinician controls only the prescription, not subsequent adherence. We did not record lesion area, which would have required photography and parsing of each lesion into measurable geometric shapes. Though it achieved its target, our study was modest in size. This was not an equivalence study, and when we designed the trial, we expected to find a benefit from the intervention. The value of the result is that it is the first evidentiary support for the relevant national guidelines [7]. We know of only 2 other relevant trials: NCT and NCT If the IDSA guidelines are correct, those trials and meta-analyses combining their results with ours will narrow the confidence interval. However, those studies have posted no results on ClinicalTrials. gov (as of 30 November 2012), and thus we do not know when they will contribute relevant data from cellulitis patients or how large their samples of cellulitis patients will be. As clinical trial evidence accumulates, antibiotic prescribing for skin infection patients might even be a reasonable target for antibiotic stewardship interventions, especially given how common skin infections are. Regarding abscesses, the evidence may already be sufficient to consider use of antibiotics for uncomplicated abscesses as a good target for antibiotic stewardship efforts. Regarding cellulitis, we feel that the present trial alone is not sufficient to motivate a stewardship campaign for cellulitis. If the results of the other studies mentioned above echo our own findings, reduction in the use of antibiotics that target CA-MRSA for uncomplicated cellulitis may become a reasonable target. CONCLUSIONS In the first study to provide experimental support for IDSA recommendations against antibiotics targeting CA-MRSA for most cases of cellulitis, we found that adding trimethoprim-sulfamethoxazole to cephalexin conferred no benefit relative to therapy with cephalexin alone in the outpatient treatment of cellulitis. Concerns about polypharmacy and antibiotic stewardship may lead us to rely on beta lactams when treating uncomplicated cellulitis. Notes Acknowledgments. We thank Patricia Kelly, RPh, of Brigham and Women s Hospital, for her invaluable help in developing placebos and planning and executing the study. We are grateful to Shannon Manzi, RPh, of Boston Children s Hospital, for her help in implementation at that site. John T. Nagurney, MD, provided invaluable support, as did Blair Parry, both of Massachusetts General Hospital. We acknowledge gratefully the work of many research assistants, attending physicians, resident physicians, and nurses who made the study happen. None of these contributors were compensated for their help. We are very grateful to the participants. Financial support. This work was supported by a grant from the Eleanor and Miles Shore Fellowship Program of the Brigham and Women s Hospital Department of Emergency Medicine; by the Milton Fund of Harvard University, Boston, MA; and by seed funding from the Department of Emergency Medicine of Massachusetts General Hospital. Potential conflicts of interest. All authors: No reported conflicts. RCT of CA-MRSA Coverage in Cellulitis CID 2013:56 (15 June) 1761
9 All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed. References 1. Herold BC, Immergluck LC, Maranan MC, et al. Community-acquired methicillin-resistant Staphylococcus aureus in children with no identified predisposing risk. JAMA 1998; 279: Moran GJ, Krishnadasan A, Gorwitz RJ, et al. Methicillin-resistant S. aureus infections among patients in the emergency department. N Engl J Med 2006; 355: Pallin DJ, Egan DJ, Pelletier AJ, Espinola JA, Hooper DC, Camargo CA Jr. Increased US emergency department visits for skin and soft tissue infections, and changes in antibiotic choices, during the emergence of community-associated methicillin-resistant Staphylococcus aureus. Ann Emerg Med 2008; 51: Schmitz GR, Bruner D, Pitotti R, et al. Randomized controlled trial of trimethoprim-sulfamethoxazole for uncomplicated skin abscesses in patients at risk for community-associated methicillin-resistant Staphylococcus aureus infection. Ann Emerg Med 2010; 56: Rajendran PM, Young D, Maurer T, et al. Randomized, double-blind, placebo-controlled trial of cephalexin for treatment of uncomplicated skin abscesses in a population at risk for community-acquired methicillin-resistant Staphylococcus aureus infection. Antimicrob Agents Chemother 2007; 51: Duong M, Markwell S, Peter J, Barenkamp S. Randomized, controlled trial of antibiotics in the management of community-acquired skin abscesses in the pediatric patient. Ann Emerg Med 2010; 55: Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillinresistant Staphylococcus aureus infections in adults and children. Clin Infect Dis 2011; 52:e Talan DA, Krishnadasan A, Gorwitz RJ, et al. Comparison of Staphylococcus aureus from skin and soft-tissue infections in US emergency department patients, 2004 and Clin Infect Dis 2011; 53: Herigon JC, Hersh AL, Gerber JS, Zaoutis TE, Newland JG. Antibiotic management of Staphylococcus aureus infections in US children s hospitals, Pediatrics 2010; 125:e Swartz MN. Clinical practice, cellulitis. N Engl J Med 2004; 350: Roland M, Torgerson D. What are pragmatic trials? BMJ 1998; 316: Szumowski JD, Cohen DE, Kanaya F, Mayer KH. Treatment and outcomes of infections by methicillin-resistant Staphylococcus aureus at an ambulatory clinic. Antimicrob Agents Chemother 2007; 51: Schulz KF, Altman DG, Moher D, CONSORT Group. CONSORT 2010 statement: Updated guidelines for reporting parallel group randomized trials. Ann Intern Med 2010; 152: DiMattia AF, Sexton MJ, Smialowicz CR, Knapp WH Jr. Efficacy of two dosage schedules of cephalexin in dermatologic infections. J Fam Pract 1981; 12: Sanford JP, Gilbert DN, Sande MA. The Sanford guide to antimicrobial therapy. Dallas, TX: Antimicrobial Therapy, Inc, Dong SL, Kelly KD, Oland RC, Holroyd BR, Rowe BH. ED management of cellulitis: A review of five urban centers. Am J Emerg Med 2001; 19: Murray H, Stiell I, Wells G. Treatment failure in emergency department patients with cellulitis. CJEM 2005; 7: Duvanel T, Auckenthaler R, Rohner P, Harms M, Saurat JH. Quantitative cultures of biopsy specimens from cutaneous cellulitis. Arch Intern Med 1989; 149: Qualls ML, Mooney MM, Camargo CA Jr, Zucconi T, Hooper DC, Pallin DJ. Emergency department visit rates for abscess versus other skin infections during the emergence of CA-MRSA, Clin Infect Dis 2012; 55: Crawford SE, David MZ, Glikman D, King KJ, Boyle-Vavra S, Daum RS. Clinical importance of purulence in methicillin-resistant Staphylococcus aureus skin and soft tissue infections. J Am Board Fam Med 2009; 22: Eells SJ, Chira S, David CG, Craft N, Miller LG. Non-suppurative cellulitis: Risk factors and its association with Staphylococcus aureus colonization in an area of endemic community-associated methicillinresistant S. aureus infections. Epidemiol Infect 2011; 139: Jeng A, Beheshti M, Li J, Nathan R. The role of beta-hemolytic streptococci in causing diffuse, nonculturable cellulitis: A prospective investigation. Medicine (Baltimore) 2010; 89: Elliott DJ, Zaoutis TE, Troxel AB, Loh A, Keren R. Empiric antimicrobial therapy for pediatric skin and soft-tissue infections in the era of methicillin-resistant Staphylococcus aureus. Pediatrics 2009; 123: e Ruhe JJ, Smith N, Bradsher RW, Menon A. Community-onset methicillin-resistant Staphylococcus aureus skin and soft-tissue infections: Impact of antimicrobial therapy on outcome. Clin Infect Dis 2007; 44: Karamatsu ML, Thorp AW, Brown L. Changes in community-associated methicillin-resistant Staphylococcus aureus skin and soft tissue infections presenting to the pediatric emergency department: Comparing 2003 to Pediatr Emerg Care 2012; 28: Vandenesch F, Naimi T, Enright MC, et al. Community-acquired methicillin-resistant Staphylococcus aureus carrying Panton-Valentine leukocidin genes: Worldwide emergence. Emerg Infect Dis 2003; 9: Naimi TS, LeDell KH, Como-Sabetti K, et al. Comparison of community- and health care-associated methicillin-resistant Staphylococcus aureus infection. JAMA 2003; 290: Genestier AL, Michallet MC, Prevost G, et al. Staphylococcus aureus Panton-Valentine leukocidin directly targets mitochondria and induces bax-independent apoptosis of human neutrophils. J Clin Invest 2005; 115: Kumar V, Robbins SL, Abbas AK. Robbins and Cotran pathologic basis of disease. Philadelphia, PA: Saunders/Elsevier, Yang ES, Tan J, Eells S, Rieg G, Tagudar G, Miller LG. Body site colonization in patients with community-associated methicillin-resistant Staphylococcus aureus and other types of S. aureus skin infections. Clin Microbiol Infect 2010; 16: CID 2013:56 (15 June) Pallin et al
Skin Infections and Antibiotic Stewardship: Analysis of Emergency Department Prescribing Practices,
 Original Research Skin Infections and Antibiotic Stewardship: Analysis of Emergency Department Prescribing Practices, 2007-2010 Daniel J. Pallin, MD, MPH Carlos A. Camargo Jr, MD, DrPH Jeremiah D. Schuur,
Original Research Skin Infections and Antibiotic Stewardship: Analysis of Emergency Department Prescribing Practices, 2007-2010 Daniel J. Pallin, MD, MPH Carlos A. Camargo Jr, MD, DrPH Jeremiah D. Schuur,
INFECTIOUS DISEASE/ORIGINAL RESEARCH
 INFECTIOUS DISEASE/ORIGINAL RESEARCH Randomized Controlled Trial of Trimethoprim-Sulfamethoxazole for Uncomplicated Skin Abscesses in Patients at Risk for Community-Associated Methicillin-Resistant Staphylococcus
INFECTIOUS DISEASE/ORIGINAL RESEARCH Randomized Controlled Trial of Trimethoprim-Sulfamethoxazole for Uncomplicated Skin Abscesses in Patients at Risk for Community-Associated Methicillin-Resistant Staphylococcus
Optimizing Antibiotic Treatment of Skin and Soft Tissue Infections
 Optimizing Antibiotic Treatment of Skin and Soft Tissue Infections 15th Annual Rocky Mountain Hospital Medicine Symposium November 6, 2017 Tim Jenkins, MD Director, Antibiotic Stewardship Program Denver
Optimizing Antibiotic Treatment of Skin and Soft Tissue Infections 15th Annual Rocky Mountain Hospital Medicine Symposium November 6, 2017 Tim Jenkins, MD Director, Antibiotic Stewardship Program Denver
DAYTON CHILDREN S HOSPITAL CLINICAL PRACTICE GUIDELINES
 DAYTON CHILDREN S HOSPITAL CLINICAL PRACTICE GUIDELINES DISCLAIMER: This Clinical Practice Guideline (CPG) generally describes a recommended course of treatment for patients with the identified health
DAYTON CHILDREN S HOSPITAL CLINICAL PRACTICE GUIDELINES DISCLAIMER: This Clinical Practice Guideline (CPG) generally describes a recommended course of treatment for patients with the identified health
Inappropriate Use of Antibiotics and Clostridium difficile Infection. Jocelyn Srigley, MD, FRCPC November 1, 2012
 Inappropriate Use of Antibiotics and Clostridium difficile Infection Jocelyn Srigley, MD, FRCPC November 1, 2012 Financial Disclosures } No conflicts of interest } The study was supported by a Hamilton
Inappropriate Use of Antibiotics and Clostridium difficile Infection Jocelyn Srigley, MD, FRCPC November 1, 2012 Financial Disclosures } No conflicts of interest } The study was supported by a Hamilton
A Prospective Investigation of Nasal Mupirocin, Hexachlorophene Body Wash, and Systemic
 AAC Accepts, published online ahead of print on 14 November 2011 Antimicrob. Agents Chemother. doi:10.1128/aac.01608-10 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions.
AAC Accepts, published online ahead of print on 14 November 2011 Antimicrob. Agents Chemother. doi:10.1128/aac.01608-10 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions.
Management of Skin and Soft-Tissue Infection
 Clinical Decisions Interactive at www.nejm.org Management of Skin and Soft-Tissue Infection This interactive feature addresses the diagnosis or management of a clinical case. A case vignette is followed
Clinical Decisions Interactive at www.nejm.org Management of Skin and Soft-Tissue Infection This interactive feature addresses the diagnosis or management of a clinical case. A case vignette is followed
Randomized, Controlled Trial of Antibiotics in the Management of Community-Acquired Skin Abscesses in the Pediatric Patient
 PEDIATRICS/ORIGINAL RESEARCH Randomized, Controlled Trial of Antibiotics in the Management of Community-Acquired Skin Abscesses in the Pediatric Patient Myto Duong, MD, MS Stephen Markwell, MA John Peter,
PEDIATRICS/ORIGINAL RESEARCH Randomized, Controlled Trial of Antibiotics in the Management of Community-Acquired Skin Abscesses in the Pediatric Patient Myto Duong, MD, MS Stephen Markwell, MA John Peter,
Infections caused by Methicillin-Resistant Staphylococcus
 MRSA infections are no longer limited to hospitals. An infectious disease specialist offers insight on what this means for dermatologists. By Robert S. Jones, DO, Reading, PA Infections caused by Methicillin-Resistant
MRSA infections are no longer limited to hospitals. An infectious disease specialist offers insight on what this means for dermatologists. By Robert S. Jones, DO, Reading, PA Infections caused by Methicillin-Resistant
Antibiotic Prophylaxis in Spinal Surgery Antibiotic Guidelines. Contents
 Antibiotic Prophylaxis in Spinal Antibiotic Guidelines Classification: Clinical Guideline Lead Author: Antibiotic Steering Committee Additional author(s): Authors Division: DCSS & Tertiary Medicine Unique
Antibiotic Prophylaxis in Spinal Antibiotic Guidelines Classification: Clinical Guideline Lead Author: Antibiotic Steering Committee Additional author(s): Authors Division: DCSS & Tertiary Medicine Unique
Le infezioni di cute e tessuti molli
 Le infezioni di cute e tessuti molli SCELTE e STRATEGIE TERAPEUTICHE Pierluigi Viale Clinica di Malattie Infettive Policlinico S. Orsola Malpighi Treatment of complicated skin and skin structure infections
Le infezioni di cute e tessuti molli SCELTE e STRATEGIE TERAPEUTICHE Pierluigi Viale Clinica di Malattie Infettive Policlinico S. Orsola Malpighi Treatment of complicated skin and skin structure infections
S aureus infections: outpatient treatment. Dirk Vogelaers Dept of Infectious Diseases University Hospital Gent Belgium
 S aureus infections: outpatient treatment Dirk Vogelaers Dept of Infectious Diseases University Hospital Gent Belgium Intern Med J. 2005 Feb;36(2):142-3 Intern Med J. 2005 Feb;36(2):142-3 Treatment of
S aureus infections: outpatient treatment Dirk Vogelaers Dept of Infectious Diseases University Hospital Gent Belgium Intern Med J. 2005 Feb;36(2):142-3 Intern Med J. 2005 Feb;36(2):142-3 Treatment of
Treatment Duration for Uncomplicated Community-Acquired Pneumonia: The Evidence in Support of 5 Days
 Treatment Duration for Uncomplicated Community-Acquired Pneumonia: The Evidence in Support of 5 Days Executive Summary National consensus guidelines created jointly by the Infectious Diseases Society of
Treatment Duration for Uncomplicated Community-Acquired Pneumonia: The Evidence in Support of 5 Days Executive Summary National consensus guidelines created jointly by the Infectious Diseases Society of
Cellulitis and Abscess: ED Phase v 1.1
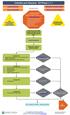 Cellulitis and Abscess: ED Phase v 1.1 Executive Summary Test Your Knowledge PHASE I (E.D.) Explanation of Evidence Ratings Summary of Version Changes! Labs if systemic illness or necrotizing fasciitis
Cellulitis and Abscess: ED Phase v 1.1 Executive Summary Test Your Knowledge PHASE I (E.D.) Explanation of Evidence Ratings Summary of Version Changes! Labs if systemic illness or necrotizing fasciitis
Felipe N. Gutierrez MD, MPH Chief, Infectious Diseases Phoenix VA Healthcare
 Felipe N. Gutierrez MD, MPH Chief, Infectious Diseases Phoenix VA Healthcare 100% of all wounds will yield growth If you get a negative culture you something is wrong! Pseudomonas while ubiquitous does
Felipe N. Gutierrez MD, MPH Chief, Infectious Diseases Phoenix VA Healthcare 100% of all wounds will yield growth If you get a negative culture you something is wrong! Pseudomonas while ubiquitous does
Clinical Management of Skin and Soft Tissue Infections in the U.S. Emergency Departments
 Original Research Clinical Management of Skin and Soft Tissue Infections in the U.S. Emergency Departments Rakesh D. Mistry, MD, MS* Daniel J. Shapiro, BA Monika K. Goyal, MD Theoklis E. Zaoutis Jeffrey
Original Research Clinical Management of Skin and Soft Tissue Infections in the U.S. Emergency Departments Rakesh D. Mistry, MD, MS* Daniel J. Shapiro, BA Monika K. Goyal, MD Theoklis E. Zaoutis Jeffrey
Blood Culture Associations in Children With a Diagnosis of Cellulitis in the Era of Methicillin-Resistant Staphylococcus aureus
 RESEARCH ARTICLE Blood Culture Associations in Children With a Diagnosis of Cellulitis in the Era of Methicillin-Resistant Staphylococcus aureus AUTHORS David Wathen, DO, Donna R. Halloran Department of
RESEARCH ARTICLE Blood Culture Associations in Children With a Diagnosis of Cellulitis in the Era of Methicillin-Resistant Staphylococcus aureus AUTHORS David Wathen, DO, Donna R. Halloran Department of
Outpatient Antimicrobial Stewardship. Jeffrey S Gerber, MD, PhD Division of Infectious Diseases The Children s Hospital of Philadelphia
 Outpatient Antimicrobial Stewardship Jeffrey S Gerber, MD, PhD Division of Infectious Diseases The Children s Hospital of Philadelphia Overview The case for outpatient antimicrobial stewardship Interventions
Outpatient Antimicrobial Stewardship Jeffrey S Gerber, MD, PhD Division of Infectious Diseases The Children s Hospital of Philadelphia Overview The case for outpatient antimicrobial stewardship Interventions
Impact of a Standardized Protocol to Address Outbreak of Methicillin-resistant
 Impact of a Standardized Protocol to Address Outbreak of Methicillin-resistant Staphylococcus Aureus Skin Infections at a large, urban County Jail System Earl J. Goldstein, MD* Gladys Hradecky, RN* Gary
Impact of a Standardized Protocol to Address Outbreak of Methicillin-resistant Staphylococcus Aureus Skin Infections at a large, urban County Jail System Earl J. Goldstein, MD* Gladys Hradecky, RN* Gary
Skin and Soft Tissue Infections Emerging Therapies and 5 things to know
 2011 MFMER slide-1 Skin and Soft Tissue Infections Emerging Therapies and 5 things to know Aaron Tande, MD Assistant Professor of Medicine October 27, 2017 Division of INFECTIOUS DISEASES 2011 MFMER slide-2
2011 MFMER slide-1 Skin and Soft Tissue Infections Emerging Therapies and 5 things to know Aaron Tande, MD Assistant Professor of Medicine October 27, 2017 Division of INFECTIOUS DISEASES 2011 MFMER slide-2
Suitability of Antibiotic Treatment for CAP (CAPTIME) The duration of antibiotic treatment in community acquired pneumonia (CAP)
 STUDY PROTOCOL Suitability of Antibiotic Treatment for CAP (CAPTIME) Purpose The duration of antibiotic treatment in community acquired pneumonia (CAP) lasts about 9 10 days, and is determined empirically.
STUDY PROTOCOL Suitability of Antibiotic Treatment for CAP (CAPTIME) Purpose The duration of antibiotic treatment in community acquired pneumonia (CAP) lasts about 9 10 days, and is determined empirically.
New Antibiotics for MRSA
 New Antibiotics for MRSA Faculty Warren S. Joseph, DPM, FIDSA Consultant, Lower Extremity Infectious Diseases Roxborough Memorial Hospital Philadelphia, Pennsylvania Faculty Disclosure Dr. Joseph: Speaker
New Antibiotics for MRSA Faculty Warren S. Joseph, DPM, FIDSA Consultant, Lower Extremity Infectious Diseases Roxborough Memorial Hospital Philadelphia, Pennsylvania Faculty Disclosure Dr. Joseph: Speaker
Necrotizing Soft Tissue Infections: Emerging Bacterial Resistance
 Necrotizing Soft Tissue Infections: Emerging Bacterial Resistance Eileen M. Bulger, MD Professor of Surgery Harborview Medical Center University of Washington Objectives Review definition & diagnostic
Necrotizing Soft Tissue Infections: Emerging Bacterial Resistance Eileen M. Bulger, MD Professor of Surgery Harborview Medical Center University of Washington Objectives Review definition & diagnostic
FM - Male, 38YO. MRSA nasal swab (+) Due to positive MRSA nasal swab test, patient will be continued on Vancomycin 1500mg IV q12 for MRSA treatment...
 Jillian O Keefe Doctor of Pharmacy Candidate 2016 September 15, 2015 FM - Male, 38YO HPI: Previously healthy male presents to ED febrile (102F) and in moderate distress ~2 weeks after getting a tattoo
Jillian O Keefe Doctor of Pharmacy Candidate 2016 September 15, 2015 FM - Male, 38YO HPI: Previously healthy male presents to ED febrile (102F) and in moderate distress ~2 weeks after getting a tattoo
ANTIMICROBIAL STEWARDSHIP FOR AMBULATORY CARE SETTINGS
 ANTIMICROBIAL STEWARDSHIP FOR AMBULATORY CARE SETTINGS Jeffrey S Gerber, MD, PhD Children s Hospital of Philadelphia University of Pennsylvania School of Medicine DISCLOSURE STATEMENT I have no conflicts
ANTIMICROBIAL STEWARDSHIP FOR AMBULATORY CARE SETTINGS Jeffrey S Gerber, MD, PhD Children s Hospital of Philadelphia University of Pennsylvania School of Medicine DISCLOSURE STATEMENT I have no conflicts
Combination vs Monotherapy for Gram Negative Septic Shock
 Combination vs Monotherapy for Gram Negative Septic Shock Critical Care Canada Forum November 8, 2018 Michael Klompas MD, MPH, FIDSA, FSHEA Professor, Harvard Medical School Hospital Epidemiologist, Brigham
Combination vs Monotherapy for Gram Negative Septic Shock Critical Care Canada Forum November 8, 2018 Michael Klompas MD, MPH, FIDSA, FSHEA Professor, Harvard Medical School Hospital Epidemiologist, Brigham
UPDATE ON ANTIMICROBIAL STEWARDSHIP REGULATIONS AND IMPLEMENTATION OF AN AMS PROGRAM
 UPDATE ON ANTIMICROBIAL STEWARDSHIP REGULATIONS AND IMPLEMENTATION OF AN AMS PROGRAM Diane Rhee, Pharm.D. Associate Professor of Pharmacy Practice Roseman University of Health Sciences Chair, Valley Health
UPDATE ON ANTIMICROBIAL STEWARDSHIP REGULATIONS AND IMPLEMENTATION OF AN AMS PROGRAM Diane Rhee, Pharm.D. Associate Professor of Pharmacy Practice Roseman University of Health Sciences Chair, Valley Health
Methicillin-Resistant Staphylococcus aureus Nasal Swabs as a Tool in Antimicrobial Stewardship
 Methicillin-Resistant Staphylococcus aureus Nasal Swabs as a Tool in Antimicrobial Stewardship Natalie R. Tucker, PharmD Antimicrobial Stewardship Pharmacist Tyson E. Dietrich, PharmD PGY2 Infectious Diseases
Methicillin-Resistant Staphylococcus aureus Nasal Swabs as a Tool in Antimicrobial Stewardship Natalie R. Tucker, PharmD Antimicrobial Stewardship Pharmacist Tyson E. Dietrich, PharmD PGY2 Infectious Diseases
Antibiotic stewardship in long term care
 Antibiotic stewardship in long term care Shira Doron, MD Associate Professor of Medicine Division of Geographic Medicine and Infectious Diseases Tufts Medical Center Boston, MA Consultant to Massachusetts
Antibiotic stewardship in long term care Shira Doron, MD Associate Professor of Medicine Division of Geographic Medicine and Infectious Diseases Tufts Medical Center Boston, MA Consultant to Massachusetts
An Approach to Appropriate Antibiotic Prescribing in Outpatient and LTC Settings?
 An Approach to Appropriate Antibiotic Prescribing in Outpatient and LTC Settings? Dr. Andrew Morris Antimicrobial Stewardship ProgramMt. Sinai Hospital University Health Network amorris@mtsinai.on.ca andrew.morris@uhn.ca
An Approach to Appropriate Antibiotic Prescribing in Outpatient and LTC Settings? Dr. Andrew Morris Antimicrobial Stewardship ProgramMt. Sinai Hospital University Health Network amorris@mtsinai.on.ca andrew.morris@uhn.ca
A Placebo-Controlled Trial of Antibiotics for Smaller Skin Abscesses
 Original Article A Placebo-Controlled Trial of Antibiotics for Smaller Skin Abscesses Robert S. Daum, M.D., C.M., Loren G. Miller, M.D., M.P.H., Lilly Immergluck, M.D., Stephanie Fritz, M.D., M.S.C.I.,
Original Article A Placebo-Controlled Trial of Antibiotics for Smaller Skin Abscesses Robert S. Daum, M.D., C.M., Loren G. Miller, M.D., M.P.H., Lilly Immergluck, M.D., Stephanie Fritz, M.D., M.S.C.I.,
Replaces:04/14/16. Formulated: 1997 SKIN AND SOFT TISSUE INFECTION
 Effective Date: 04/13/17 Replaces:04/14/16 Page 1 of 7 POLICY To standardize the clinical management and housing of offenders with skin and soft tissue infections, thereby reducing the transmission and
Effective Date: 04/13/17 Replaces:04/14/16 Page 1 of 7 POLICY To standardize the clinical management and housing of offenders with skin and soft tissue infections, thereby reducing the transmission and
CA-MRSA lesions: What works, what doesn t
 For mass reproduction, content licensing and permissions contact Dowden Health Media. FAMILY David McBride, MD University Student Health Services and the Department of Family Medicine, Boston University
For mass reproduction, content licensing and permissions contact Dowden Health Media. FAMILY David McBride, MD University Student Health Services and the Department of Family Medicine, Boston University
Active Bacterial Core Surveillance Site and Epidemiologic Classification, United States, 2005a. Copyright restrictions may apply.
 Impact of routine surgical ward and intensive care unit admission surveillance cultures on hospital-wide nosocomial methicillin-resistant Staphylococcus aureus infections in a university hospital: an interrupted
Impact of routine surgical ward and intensive care unit admission surveillance cultures on hospital-wide nosocomial methicillin-resistant Staphylococcus aureus infections in a university hospital: an interrupted
Community-Onset Methicillin-Resistant Staphylococcus aureus Skin and Soft-Tissue Infections: Impact of Antimicrobial Therapy on Outcome
 MAJOR ARTICLE Community-Onset Methicillin-Resistant Staphylococcus aureus Skin and Soft-Tissue Infections: Impact of Antimicrobial Therapy on Outcome Jörg J. Ruhe, 1,2 Nathaniel Smith, 1,3 Robert W. Bradsher,
MAJOR ARTICLE Community-Onset Methicillin-Resistant Staphylococcus aureus Skin and Soft-Tissue Infections: Impact of Antimicrobial Therapy on Outcome Jörg J. Ruhe, 1,2 Nathaniel Smith, 1,3 Robert W. Bradsher,
Appropriate Management of Common Pediatric Infections. Blaise L. Congeni M.D. Akron Children s Hospital Division of Pediatric Infectious Diseases
 Appropriate Management of Common Pediatric Infections Blaise L. Congeni M.D. Akron Children s Hospital Division of Pediatric Infectious Diseases It s all about the microorganism The common pathogens Viruses
Appropriate Management of Common Pediatric Infections Blaise L. Congeni M.D. Akron Children s Hospital Division of Pediatric Infectious Diseases It s all about the microorganism The common pathogens Viruses
Antimicrobial Stewardship in the Hospital Setting
 GUIDE TO INFECTION CONTROL IN THE HOSPITAL CHAPTER 12 Antimicrobial Stewardship in the Hospital Setting Authors Dan Markley, DO, MPH, Amy L. Pakyz, PharmD, PhD, Michael Stevens, MD, MPH Chapter Editor
GUIDE TO INFECTION CONTROL IN THE HOSPITAL CHAPTER 12 Antimicrobial Stewardship in the Hospital Setting Authors Dan Markley, DO, MPH, Amy L. Pakyz, PharmD, PhD, Michael Stevens, MD, MPH Chapter Editor
Mrsa abscess and cellulitis
 Search Mrsa abscess and cellulitis An abscess is a collection of pus that has built up within the tissue of the body. Signs and symptoms of abscesses include redness, pain, warmth, and swelling. The. Staph
Search Mrsa abscess and cellulitis An abscess is a collection of pus that has built up within the tissue of the body. Signs and symptoms of abscesses include redness, pain, warmth, and swelling. The. Staph
Objectives 4/26/2017. Co-Investigators Sadie Giuliani, PharmD, BCPS Claude Tonnerre, MD Jayme Hartzell, PharmD, MS, BCPS
 IMPLEMENTATION AND ASSESSMENT OF A GUIDELINE-BASED TREATMENT ALGORITHM FOR COMMUNITY-ACQUIRED PNEUMONIA (CAP) Lucas Schonsberg, PharmD PGY-1 Pharmacy Practice Resident Providence St. Patrick Hospital Missoula,
IMPLEMENTATION AND ASSESSMENT OF A GUIDELINE-BASED TREATMENT ALGORITHM FOR COMMUNITY-ACQUIRED PNEUMONIA (CAP) Lucas Schonsberg, PharmD PGY-1 Pharmacy Practice Resident Providence St. Patrick Hospital Missoula,
IMPLEMENTATION AND ASSESSMENT OF A GUIDELINE-BASED TREATMENT ALGORITHM FOR COMMUNITY-ACQUIRED PNEUMONIA (CAP)
 IMPLEMENTATION AND ASSESSMENT OF A GUIDELINE-BASED TREATMENT ALGORITHM FOR COMMUNITY-ACQUIRED PNEUMONIA (CAP) Lucas Schonsberg, PharmD PGY-1 Pharmacy Practice Resident Providence St. Patrick Hospital Missoula,
IMPLEMENTATION AND ASSESSMENT OF A GUIDELINE-BASED TREATMENT ALGORITHM FOR COMMUNITY-ACQUIRED PNEUMONIA (CAP) Lucas Schonsberg, PharmD PGY-1 Pharmacy Practice Resident Providence St. Patrick Hospital Missoula,
Barriers to Intravenous Penicillin Use for Treatment of Nonmeningitis
 JCM Accepts, published online ahead of print on 7 July 2010 J. Clin. Microbiol. doi:10.1128/jcm.01012-10 Copyright 2010, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
JCM Accepts, published online ahead of print on 7 July 2010 J. Clin. Microbiol. doi:10.1128/jcm.01012-10 Copyright 2010, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
Development of Drugs for Skin Infections
 EFPIA - Skin Infection comments 1 Development of Drugs for Skin Infections John H Rex, MD EFPIA - Skin Infection comments 2 Skin Infections Significant recent debate: Acceptable forms: A focus on fever
EFPIA - Skin Infection comments 1 Development of Drugs for Skin Infections John H Rex, MD EFPIA - Skin Infection comments 2 Skin Infections Significant recent debate: Acceptable forms: A focus on fever
11/22/2016. Antimicrobial Stewardship Update Disclosures. Outline. No conflicts of interest to disclose
 Antimicrobial Stewardship Update 2016 APIC-CI Conference November 17 th, 2016 Jay R. McDonald, MD Chief, ID Section VA St. Louis Health Care System Assistant Professor of medicine Washington University
Antimicrobial Stewardship Update 2016 APIC-CI Conference November 17 th, 2016 Jay R. McDonald, MD Chief, ID Section VA St. Louis Health Care System Assistant Professor of medicine Washington University
Commonwealth of Kentucky Antibiotic Stewardship Practice Assessment For Long-Term Care Facilities
 Commonwealth of Kentucky Antibiotic Stewardship Practice Assessment For Long-Term Care Facilities Introduction As the problem of antibiotic resistance continues to worsen in all healthcare setting, we
Commonwealth of Kentucky Antibiotic Stewardship Practice Assessment For Long-Term Care Facilities Introduction As the problem of antibiotic resistance continues to worsen in all healthcare setting, we
Host, Syndrome, Bug, Drug: Introducing 2 Frameworks to Approach Infectious Diseases Cases with an Antimicrobial Stewardship Focus
 Host, Syndrome, Bug, Drug: Introducing 2 Frameworks to Approach Infectious Diseases Cases with an Antimicrobial Stewardship Focus Montana ACP Meeting 2018 September 8, 2018 Staci Lee, MD, MEHP Billings
Host, Syndrome, Bug, Drug: Introducing 2 Frameworks to Approach Infectious Diseases Cases with an Antimicrobial Stewardship Focus Montana ACP Meeting 2018 September 8, 2018 Staci Lee, MD, MEHP Billings
HEALTH SERVICES POLICY & PROCEDURE MANUAL
 PAGE 1 of 3 PURPOSE To assure that DOP inmates with Soft Tissue Infections are receiving high quality Primary Care for their infections and that the risk of infecting other inmates or staff is minimized.
PAGE 1 of 3 PURPOSE To assure that DOP inmates with Soft Tissue Infections are receiving high quality Primary Care for their infections and that the risk of infecting other inmates or staff is minimized.
Antimicrobial stewardship: Quick, don t just do something! Stand there!
 Antimicrobial stewardship: Quick, don t just do something! Stand there! Stanley I. Martin, MD, FACP, FIDSA Director, Division of Infectious Diseases Director, Antimicrobial Stewardship Program Geisinger
Antimicrobial stewardship: Quick, don t just do something! Stand there! Stanley I. Martin, MD, FACP, FIDSA Director, Division of Infectious Diseases Director, Antimicrobial Stewardship Program Geisinger
Bacterial skin infection
 D i v i s i o n o f P e d i a t r i c E m e r g e n c y M e d i c i n e P a g e 1 Bacterial skin infection Cellulitis w/o abscess Abscess Deep tissue involvement Multiple abscesses Perirectal Anterior
D i v i s i o n o f P e d i a t r i c E m e r g e n c y M e d i c i n e P a g e 1 Bacterial skin infection Cellulitis w/o abscess Abscess Deep tissue involvement Multiple abscesses Perirectal Anterior
Optimizing Antibiotic Stewardship in the ED
 Optimizing Antibiotic Stewardship in the ED Michael Pulia, MD MS FAAEM FACEP Director, UW EM Antibiotic Stewardship Research Program Chair, AAEM Antimicrobial Stewardship Task Force @DrMichaelPulia Learning
Optimizing Antibiotic Stewardship in the ED Michael Pulia, MD MS FAAEM FACEP Director, UW EM Antibiotic Stewardship Research Program Chair, AAEM Antimicrobial Stewardship Task Force @DrMichaelPulia Learning
Impact of Systemic Antibiotics on Staphylococcus aureus Colonization and Recurrent Skin Infection
 Clinical Infectious Diseases MAJOR ARTICLE Impact of Systemic Antibiotics on Staphylococcus aureus Colonization and Recurrent Skin Infection Patrick G. Hogan, 1 Marcela Rodriguez, 2 Allison M. Spenner,
Clinical Infectious Diseases MAJOR ARTICLE Impact of Systemic Antibiotics on Staphylococcus aureus Colonization and Recurrent Skin Infection Patrick G. Hogan, 1 Marcela Rodriguez, 2 Allison M. Spenner,
Antimicrobial Stewardship in the Long Term Care and Outpatient Settings. Carlos Reyes Sacin, MD, AAHIVS
 Antimicrobial Stewardship in the Long Term Care and Outpatient Settings Carlos Reyes Sacin, MD, AAHIVS Disclosure Speaker and consultant in HIV medicine for Gilead and Jansen Pharmaceuticals Objectives
Antimicrobial Stewardship in the Long Term Care and Outpatient Settings Carlos Reyes Sacin, MD, AAHIVS Disclosure Speaker and consultant in HIV medicine for Gilead and Jansen Pharmaceuticals Objectives
Optimizing Antimicrobial Stewardship Activities Based on Institutional Resources
 Optimizing Antimicrobial Stewardship Activities Based on Institutional Resources Andrew Hunter, PharmD, BCPS Infectious Diseases Clinical Pharmacy Specialist Michael E. DeBakey VA Medical Center Andrew.hunter@va.gov
Optimizing Antimicrobial Stewardship Activities Based on Institutional Resources Andrew Hunter, PharmD, BCPS Infectious Diseases Clinical Pharmacy Specialist Michael E. DeBakey VA Medical Center Andrew.hunter@va.gov
Physician Rating: ( 23 Votes ) Rate This Article:
 From Medscape Infectious Diseases Conquering Antibiotic Overuse An Expert Interview With the CDC Laura A. Stokowski, RN, MS Authors and Disclosures Posted: 11/30/2010 Physician Rating: ( 23 Votes ) Rate
From Medscape Infectious Diseases Conquering Antibiotic Overuse An Expert Interview With the CDC Laura A. Stokowski, RN, MS Authors and Disclosures Posted: 11/30/2010 Physician Rating: ( 23 Votes ) Rate
Diabetic Foot Infection. Dr David Orr Consultant Microbiologist Lancashire Teaching Hospitals
 Diabetic Foot Infection Dr David Orr Consultant Microbiologist Lancashire Teaching Hospitals History of previous amputation [odds ratio (OR)=19.9, P=.01], Peripheral vascular disease (OR=5.5, P=.007)
Diabetic Foot Infection Dr David Orr Consultant Microbiologist Lancashire Teaching Hospitals History of previous amputation [odds ratio (OR)=19.9, P=.01], Peripheral vascular disease (OR=5.5, P=.007)
Treatment of septic peritonitis
 Vet Times The website for the veterinary profession https://www.vettimes.co.uk Treatment of septic peritonitis Author : Andrew Linklater Categories : Companion animal, Vets Date : November 2, 2016 Septic
Vet Times The website for the veterinary profession https://www.vettimes.co.uk Treatment of septic peritonitis Author : Andrew Linklater Categories : Companion animal, Vets Date : November 2, 2016 Septic
Objective 1/20/2016. Expanding Antimicrobial Stewardship into the Outpatient Setting. Disclosure Statement of Financial Interest
 Expanding Antimicrobial Stewardship into the Outpatient Setting Michael E. Klepser, Pharm.D., FCCP Professor Pharmacy Practice Ferris State University College of Pharmacy Disclosure Statement of Financial
Expanding Antimicrobial Stewardship into the Outpatient Setting Michael E. Klepser, Pharm.D., FCCP Professor Pharmacy Practice Ferris State University College of Pharmacy Disclosure Statement of Financial
Standing Orders for the Treatment of Outpatient Peritonitis
 Standing Orders for the Treatment of Outpatient Peritonitis 1. Definition of Peritonitis: a. Cloudy effluent. b. WBC > 100 cells/mm3 with >50% polymorphonuclear (PMN) cells with minimum 2 hour dwell. c.
Standing Orders for the Treatment of Outpatient Peritonitis 1. Definition of Peritonitis: a. Cloudy effluent. b. WBC > 100 cells/mm3 with >50% polymorphonuclear (PMN) cells with minimum 2 hour dwell. c.
11/10/2016. Skin and Soft Tissue Infections. Disclosures. Educational Need/Practice Gap. Objectives. Case #1
 Disclosures Selecting Antimicrobials for Common Infections in Children FMR-Contemporary Pediatrics 11/2016 Sean McTigue, MD Assistant Professor of Pediatrics, Pediatric Infectious Diseases Medical Director
Disclosures Selecting Antimicrobials for Common Infections in Children FMR-Contemporary Pediatrics 11/2016 Sean McTigue, MD Assistant Professor of Pediatrics, Pediatric Infectious Diseases Medical Director
Cellulitis. Assoc Prof Mark Thomas. Conference for General Practice Auckland Saturday 28 July 2018
 Cellulitis Assoc Prof Mark Thomas Conference for General Practice Auckland Saturday 28 July 2018 Summary Cellulitis Usual treatment flucloxacillin for 5 days Frequent recurrences consider penicillin 250mg
Cellulitis Assoc Prof Mark Thomas Conference for General Practice Auckland Saturday 28 July 2018 Summary Cellulitis Usual treatment flucloxacillin for 5 days Frequent recurrences consider penicillin 250mg
Scottish Medicines Consortium
 Scottish Medicines Consortium tigecycline 50mg vial of powder for intravenous infusion (Tygacil ) (277/06) Wyeth 9 June 2006 The Scottish Medicines Consortium (SMC) has completed its assessment of the
Scottish Medicines Consortium tigecycline 50mg vial of powder for intravenous infusion (Tygacil ) (277/06) Wyeth 9 June 2006 The Scottish Medicines Consortium (SMC) has completed its assessment of the
5/17/2012 DISCLOSURES OBJECTIVES CONTEMPORARY PEDIATRICS
 CONTEMPORARY PEDIATRICS Surgical Management of MRSA Soft Tissue Infections John M. Draus, Jr., M.D. Assistant Professor of Surgery and Pediatrics Kentucky Children s Hospital University of Kentucky DISCLOSURES
CONTEMPORARY PEDIATRICS Surgical Management of MRSA Soft Tissue Infections John M. Draus, Jr., M.D. Assistant Professor of Surgery and Pediatrics Kentucky Children s Hospital University of Kentucky DISCLOSURES
Intra-Abdominal Infections. Jessica Thompson, PharmD, BCPS (AQ-ID) Infectious Diseases Pharmacy Clinical Specialist Renown Health April 19, 2018
 Intra-Abdominal Infections Jessica Thompson, PharmD, BCPS (AQ-ID) Infectious Diseases Pharmacy Clinical Specialist Renown Health April 19, 2018 Select guidelines Mazuski JE, et al. The Surgical Infection
Intra-Abdominal Infections Jessica Thompson, PharmD, BCPS (AQ-ID) Infectious Diseases Pharmacy Clinical Specialist Renown Health April 19, 2018 Select guidelines Mazuski JE, et al. The Surgical Infection
Scottish Medicines Consortium
 Scottish Medicines Consortium daptomycin 350mg powder for concentrate for solution for infusion (Cubicin ) Chiron Corporation Limited No. (248/06) 10 March 2006 The Scottish Medicines Consortium (SMC)
Scottish Medicines Consortium daptomycin 350mg powder for concentrate for solution for infusion (Cubicin ) Chiron Corporation Limited No. (248/06) 10 March 2006 The Scottish Medicines Consortium (SMC)
Author - Dr. Josie Traub-Dargatz
 Author - Dr. Josie Traub-Dargatz Dr. Josie Traub-Dargatz is a professor of equine medicine at Colorado State University (CSU) College of Veterinary Medicine and Biomedical Sciences. She began her veterinary
Author - Dr. Josie Traub-Dargatz Dr. Josie Traub-Dargatz is a professor of equine medicine at Colorado State University (CSU) College of Veterinary Medicine and Biomedical Sciences. She began her veterinary
Incidence of hospital-acquired Clostridium difficile infection in patients at risk
 Baptist Health South Florida Scholarly Commons @ Baptist Health South Florida All Publications 5-20-2016 Incidence of hospital-acquired Clostridium difficile infection in patients at risk Christine Ibarra
Baptist Health South Florida Scholarly Commons @ Baptist Health South Florida All Publications 5-20-2016 Incidence of hospital-acquired Clostridium difficile infection in patients at risk Christine Ibarra
Standing Orders for the Treatment of Outpatient Peritonitis
 Standing Orders for the Treatment of Outpatient Peritonitis 1. Definition of Peritonitis: a. Cloudy effluent. b. WBC > 100 cells/mm3 with >50% polymorphonuclear (PMN) cells with minimum 2 hour dwell. c.
Standing Orders for the Treatment of Outpatient Peritonitis 1. Definition of Peritonitis: a. Cloudy effluent. b. WBC > 100 cells/mm3 with >50% polymorphonuclear (PMN) cells with minimum 2 hour dwell. c.
Source: Portland State University Population Research Center (
 Methicillin Resistant Staphylococcus aureus (MRSA) Surveillance Report 2010 Oregon Active Bacterial Core Surveillance (ABCs) Office of Disease Prevention & Epidemiology Oregon Health Authority Updated:
Methicillin Resistant Staphylococcus aureus (MRSA) Surveillance Report 2010 Oregon Active Bacterial Core Surveillance (ABCs) Office of Disease Prevention & Epidemiology Oregon Health Authority Updated:
Antibiotic Abyss. Discussion Points. MRSA Treatment Guidelines
 Antibiotic Abyss Fredrick M. Abrahamian, D.O., FACEP, FIDSA Professor of Medicine UCLA School of Medicine Director of Education Department of Emergency Medicine Olive View-UCLA Medical Center Sylmar, California
Antibiotic Abyss Fredrick M. Abrahamian, D.O., FACEP, FIDSA Professor of Medicine UCLA School of Medicine Director of Education Department of Emergency Medicine Olive View-UCLA Medical Center Sylmar, California
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #407: Appropriate Treatment of Methicillin-Susceptible Staphylococcus Aureus (MSSA) Bacteremia National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES:
Quality ID #407: Appropriate Treatment of Methicillin-Susceptible Staphylococcus Aureus (MSSA) Bacteremia National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES:
8/17/2016 ABOUT US REDUCTION OF CLOSTRIDIUM DIFFICILE THROUGH THE USE OF AN ANTIMICROBIAL STEWARDSHIP PROGRAM
 Mary Moore, MS CIC MT (ASCP) Infection Prevention Coordinator Great River Medical Center, West Burlington REDUCTION OF CLOSTRIDIUM DIFFICILE THROUGH THE USE OF AN ANTIMICROBIAL STEWARDSHIP PROGRAM ABOUT
Mary Moore, MS CIC MT (ASCP) Infection Prevention Coordinator Great River Medical Center, West Burlington REDUCTION OF CLOSTRIDIUM DIFFICILE THROUGH THE USE OF AN ANTIMICROBIAL STEWARDSHIP PROGRAM ABOUT
Cefazolin vs. Antistaphyloccal Penicillins: The Great Debate
 Cefazolin vs. Antistaphyloccal Penicillins: The Great Debate Annie Heble, PharmD PGY2 Pediatric Pharmacy Resident Children s Hospital Colorado Microbiology Rounds March 22, 2017 Image Source: Buck cartoons
Cefazolin vs. Antistaphyloccal Penicillins: The Great Debate Annie Heble, PharmD PGY2 Pediatric Pharmacy Resident Children s Hospital Colorado Microbiology Rounds March 22, 2017 Image Source: Buck cartoons
Antibiotic prophylaxis guideline for colorectal, hepatobiliary and vascular surgery for adult patients.
 Antibiotic prophylaxis guideline for colorectal, hepatobiliary and vascular surgery for adult patients. Full Title of Guideline: Author (include email and role): Division & Speciality: Scope (Target audience,
Antibiotic prophylaxis guideline for colorectal, hepatobiliary and vascular surgery for adult patients. Full Title of Guideline: Author (include email and role): Division & Speciality: Scope (Target audience,
Antibiotic Updates: Part II
 Antibiotic Updates: Part II Fredrick M. Abrahamian, DO, FACEP, FIDSA Health Sciences Clinical Professor of Emergency Medicine David Geffen School of Medicine at UCLA Los Angeles, California Financial Disclosures
Antibiotic Updates: Part II Fredrick M. Abrahamian, DO, FACEP, FIDSA Health Sciences Clinical Professor of Emergency Medicine David Geffen School of Medicine at UCLA Los Angeles, California Financial Disclosures
SKIN AND SOFT TISSUE INFECTIONS OCTOBER 3-4, 2015
 SKIN AND SOFT TISSUE INFECTIONS OCTOBER 3-4, 2015 Disclosures I have no financial conflicts of interest to disclose or report. Steven Tran, PharmD NEFSHP Fall Meeting 2015 Objectives for Pharmacists Review
SKIN AND SOFT TISSUE INFECTIONS OCTOBER 3-4, 2015 Disclosures I have no financial conflicts of interest to disclose or report. Steven Tran, PharmD NEFSHP Fall Meeting 2015 Objectives for Pharmacists Review
Simplicef is Used to Treat Animals with Skin Infections
 Simplicef is Used to Treat Animals with Skin Infections PRODUCT INFO Simplicef tablets are a semi-synthetic cephalosporin antibiotic cefpodoxime proxetil used to cure infections caused by the susceptible
Simplicef is Used to Treat Animals with Skin Infections PRODUCT INFO Simplicef tablets are a semi-synthetic cephalosporin antibiotic cefpodoxime proxetil used to cure infections caused by the susceptible
Safety of an Out-Patient Intravenous Antibiotics Programme
 Safety of an Out-Patient Intravenous Antibiotics Programme Chan VL, Tang ESK, Leung WS, Wong L, Cheung PS, Chu CM Department of Medicine & Geriatrics United Christian Hospital Outpatient Parental Antimicrobial
Safety of an Out-Patient Intravenous Antibiotics Programme Chan VL, Tang ESK, Leung WS, Wong L, Cheung PS, Chu CM Department of Medicine & Geriatrics United Christian Hospital Outpatient Parental Antimicrobial
Period of study: 12 Nov 2002 to 08 Apr 2004 (first subject s first visit to last subject s last visit)
 Study Synopsis This file is posted on the Bayer HealthCare Clinical Trials Registry and Results website and is provided for patients and healthcare professionals to increase the transparency of Bayer's
Study Synopsis This file is posted on the Bayer HealthCare Clinical Trials Registry and Results website and is provided for patients and healthcare professionals to increase the transparency of Bayer's
GUIDELINES FOR THE MANAGEMENT OF COMMUNITY-ACQUIRED PNEUMONIA IN ADULTS
 Version 3.1 GUIDELINES FOR THE MANAGEMENT OF COMMUNITY-ACQUIRED PNEUMONIA IN ADULTS Date ratified June 2008 Updated March 2009 Review date June 2010 Ratified by Authors Consultation Evidence base Changes
Version 3.1 GUIDELINES FOR THE MANAGEMENT OF COMMUNITY-ACQUIRED PNEUMONIA IN ADULTS Date ratified June 2008 Updated March 2009 Review date June 2010 Ratified by Authors Consultation Evidence base Changes
Evaluating the Role of MRSA Nasal Swabs
 Evaluating the Role of MRSA Nasal Swabs Josh Arnold, PharmD PGY1 Pharmacy Resident Pharmacy Grand Rounds February 28, 2017 2016 MFMER slide-1 Objectives Identify the pathophysiology of MRSA nasal colonization
Evaluating the Role of MRSA Nasal Swabs Josh Arnold, PharmD PGY1 Pharmacy Resident Pharmacy Grand Rounds February 28, 2017 2016 MFMER slide-1 Objectives Identify the pathophysiology of MRSA nasal colonization
ANTIBIOTICS IN THE ER:
 ANTIBIOTICS IN THE ER: EXPLORING THE ROLE OF ANTIMICROBIAL STEWARDSHIP IN THE EMERGENCY DEPARTMENT ANGELINA DAVIS, PHARMD, MS, BCPS (AQ-ID) LIAISON CLINICAL PHARMACIST DUKE ANTIMICROBIAL STEWARDSHIP OUTREACH
ANTIBIOTICS IN THE ER: EXPLORING THE ROLE OF ANTIMICROBIAL STEWARDSHIP IN THE EMERGENCY DEPARTMENT ANGELINA DAVIS, PHARMD, MS, BCPS (AQ-ID) LIAISON CLINICAL PHARMACIST DUKE ANTIMICROBIAL STEWARDSHIP OUTREACH
Ca-MRSA Update- Hand Infections. Washington Hand Society September 19, 2007
 Ca-MRSA Update- Hand Infections Washington Hand Society September 19, 2007 Resistant Staph. Aureus Late 1940 s -50% S.Aureus resistant to PCN 1957-80/81 strain- of S.A. highly virulent and easily transmissible
Ca-MRSA Update- Hand Infections Washington Hand Society September 19, 2007 Resistant Staph. Aureus Late 1940 s -50% S.Aureus resistant to PCN 1957-80/81 strain- of S.A. highly virulent and easily transmissible
The CARI Guidelines Caring for Australians with Renal Impairment. 10. Treatment of peritoneal dialysis associated fungal peritonitis
 10. Treatment of peritoneal dialysis associated fungal peritonitis Date written: February 2003 Final submission: July 2004 Guidelines (Include recommendations based on level I or II evidence) The use of
10. Treatment of peritoneal dialysis associated fungal peritonitis Date written: February 2003 Final submission: July 2004 Guidelines (Include recommendations based on level I or II evidence) The use of
Antimicrobial Update Stewardship in Primary Care. Clare Colligan Antimicrobial Pharmacist NHS Forth Valley
 Antimicrobial Update Stewardship in Primary Care Clare Colligan Antimicrobial Pharmacist NHS Forth Valley Setting the Scene! Consequences of Antibiotic Use? Resistance For an individual patient with
Antimicrobial Update Stewardship in Primary Care Clare Colligan Antimicrobial Pharmacist NHS Forth Valley Setting the Scene! Consequences of Antibiotic Use? Resistance For an individual patient with
Treating Rosacea in the Era of Bacterial Resistance. This presentation is sponsored by Galderma Laboratories, L.P.
 Treating Rosacea in the Era of Bacterial Resistance This presentation is sponsored by Galderma Laboratories, L.P. Lecture Discuss rosacea as an inflammatory condition Assess the psychosocial impact of
Treating Rosacea in the Era of Bacterial Resistance This presentation is sponsored by Galderma Laboratories, L.P. Lecture Discuss rosacea as an inflammatory condition Assess the psychosocial impact of
Randomized Controlled Trial on Adjunctive Lavage for Severe Peritoneal Dialysis- Related Peritonitis
 Randomized Controlled Trial on Adjunctive Lavage for Severe Peritoneal Dialysis- Related Peritonitis Steve SM Wong Alice Ho Miu Ling Nethersole Hospital Background PD peritonitis is a major cause of PD
Randomized Controlled Trial on Adjunctive Lavage for Severe Peritoneal Dialysis- Related Peritonitis Steve SM Wong Alice Ho Miu Ling Nethersole Hospital Background PD peritonitis is a major cause of PD
Acute Pyelonephritis POAC Guideline
 Acute Pyelonephritis POAC Guideline Refer full regional pathway http://aucklandregion.healthpathways.org.nz/33444 EXCLUSION CRITERIA: COMPLICATED PYELONEPHRITIS Discuss with relevant specialist for advice
Acute Pyelonephritis POAC Guideline Refer full regional pathway http://aucklandregion.healthpathways.org.nz/33444 EXCLUSION CRITERIA: COMPLICATED PYELONEPHRITIS Discuss with relevant specialist for advice
Clinical Practice Standard
 Clinical Practice Standard 1-20-6-1-010 TITLE: INTRAVENOUS TO ORAL CONVERSION FOR ANTIMICROBIALS A printed copy of this document may not reflect the current, electronic version on OurNH. APPLICABILITY:
Clinical Practice Standard 1-20-6-1-010 TITLE: INTRAVENOUS TO ORAL CONVERSION FOR ANTIMICROBIALS A printed copy of this document may not reflect the current, electronic version on OurNH. APPLICABILITY:
Duration of antibiotic therapy:
 Duration of antibiotic therapy: How low can you go? Thomas Holland, MD Hilton Head, SC July 2017 Disclosures Consulting: The Medicines Company, Basilea Pharmaceutica Adjudication committee: Achaogen Grant
Duration of antibiotic therapy: How low can you go? Thomas Holland, MD Hilton Head, SC July 2017 Disclosures Consulting: The Medicines Company, Basilea Pharmaceutica Adjudication committee: Achaogen Grant
Overview Management of Skin and Soft Tissue Infections in the MRSA Era
 Overview Management of Skin and Soft Tissue Infections in the MRSA Era April 2011 2011 IDSA MRSA Treatment Guidelines Skin and soft tissue infections (SSTIs) Management of Recurrent SSTIs Necrotizing soft
Overview Management of Skin and Soft Tissue Infections in the MRSA Era April 2011 2011 IDSA MRSA Treatment Guidelines Skin and soft tissue infections (SSTIs) Management of Recurrent SSTIs Necrotizing soft
Geriatric Mental Health Partnership
 Geriatric Mental Health Partnership September 8, 2017 First, let s test your knowledge about antibiotics http://www.cdc.gov/getsmart/community/about/quiz.html 2 Get Smart Antibiotics Quiz Antibiotics fight
Geriatric Mental Health Partnership September 8, 2017 First, let s test your knowledge about antibiotics http://www.cdc.gov/getsmart/community/about/quiz.html 2 Get Smart Antibiotics Quiz Antibiotics fight
Trimethoprim Sulfamethoxazole versus Placebo for Uncomplicated Skin Abscess
 The new england journal of medicine Original Article Trimethoprim Sulfamethoxazole versus Placebo for Uncomplicated Skin Abscess David A. Talan, M.D., William R. Mower, M.D., Ph.D., Anusha Krishnadasan,
The new england journal of medicine Original Article Trimethoprim Sulfamethoxazole versus Placebo for Uncomplicated Skin Abscess David A. Talan, M.D., William R. Mower, M.D., Ph.D., Anusha Krishnadasan,
The Impact of meca Gene Testing and Infectious Diseases Pharmacists. Intervention on the Time to Optimal Antimicrobial Therapy for ACCEPTED
 JCM Accepts, published online ahead of print on 7 May 2008 J. Clin. Microbiol. doi:10.1128/jcm.00801-08 Copyright 2008, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
JCM Accepts, published online ahead of print on 7 May 2008 J. Clin. Microbiol. doi:10.1128/jcm.00801-08 Copyright 2008, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
10/9/2017. Evidence-Based Interventions to Reduce Inappropriate Prescription of Antibiotics. Prescribing for Respiratory Tract Infections
 Evidence-Based Interventions to Reduce Inappropriate Prescription of Antibiotics Ann Thomas, MD, MPH Oregon Public Health Division Prescribing for Respiratory Tract Infections Antibiotic use is primary
Evidence-Based Interventions to Reduce Inappropriate Prescription of Antibiotics Ann Thomas, MD, MPH Oregon Public Health Division Prescribing for Respiratory Tract Infections Antibiotic use is primary
EVIDENCE BASED MEDICINE: ANTIBIOTIC RESISTANCE IN THE ELDERLY CHETHANA KAMATH GERIATRIC MEDICINE WEEK
 EVIDENCE BASED MEDICINE: ANTIBIOTIC RESISTANCE IN THE ELDERLY CHETHANA KAMATH GERIATRIC MEDICINE WEEK EPIDEMIOLOGY AND BACKGROUND Every year, more than 2 million people in the United States acquire antibiotic-resistant
EVIDENCE BASED MEDICINE: ANTIBIOTIC RESISTANCE IN THE ELDERLY CHETHANA KAMATH GERIATRIC MEDICINE WEEK EPIDEMIOLOGY AND BACKGROUND Every year, more than 2 million people in the United States acquire antibiotic-resistant
Symptoms of cellulitis (n=396) %
 Cellulitis and lymphoedema Vaughan Keeley May 2012 What is cellulitis? - also called erysipelas, acute inflammatory episodes etc. - bacterial infection of skin + subcutaneous tissues - more common in people
Cellulitis and lymphoedema Vaughan Keeley May 2012 What is cellulitis? - also called erysipelas, acute inflammatory episodes etc. - bacterial infection of skin + subcutaneous tissues - more common in people
Antibiotic Stewardship in the LTC Setting
 Antibiotic Stewardship in the LTC Setting Joe Litsey, Director of Consulting Services Pharm.D., Board Certified Geriatric Pharmacist Thrifty White Pharmacy Objectives Describe the Antibiotic Stewardship
Antibiotic Stewardship in the LTC Setting Joe Litsey, Director of Consulting Services Pharm.D., Board Certified Geriatric Pharmacist Thrifty White Pharmacy Objectives Describe the Antibiotic Stewardship
Community Methicillin- Resistant Staphylococcus aureus. Sixth Plague of Egypt. Epidemiology
 Community Methicillin- Resistant Staphylococcus aureus Henry F. Chambers, M.D. University of California San Francisco San Francisco General Hospital Sixth Plague of Egypt (~ 1200 BCE) So they took soot
Community Methicillin- Resistant Staphylococcus aureus Henry F. Chambers, M.D. University of California San Francisco San Francisco General Hospital Sixth Plague of Egypt (~ 1200 BCE) So they took soot
Appropriate Antimicrobial Therapy for Treatment of
 Appropriate Antimicrobial Therapy for Treatment of Staphylococcus aureus infections ( MRSA ) By : A. Bojdi MD Assistant Professor Inf. Dis. Dep. Imam Reza Hosp. MUMS Antibiotics Still Miracle Drugs Paul
Appropriate Antimicrobial Therapy for Treatment of Staphylococcus aureus infections ( MRSA ) By : A. Bojdi MD Assistant Professor Inf. Dis. Dep. Imam Reza Hosp. MUMS Antibiotics Still Miracle Drugs Paul
File S1: Questionnaire for self-medication with antibiotics
 File S1: Questionnaire for self-medication with antibiotics Part A: Self-medication behaviors 1 2 3 4 5 6 7 8 Have you ever taken antibiotics? If NO, please go to Part B Question 1 Have you ever treated
File S1: Questionnaire for self-medication with antibiotics Part A: Self-medication behaviors 1 2 3 4 5 6 7 8 Have you ever taken antibiotics? If NO, please go to Part B Question 1 Have you ever treated
