Cases of Cat-Associated Human Plague in the Western US,
|
|
|
- Annabelle Richardson
- 6 years ago
- Views:
Transcription
1 893 Cases of Cat-Associated Human Plague in the Western US, Kenneth L. Gage, 1 David T. Dennis, 1 Kathy A. Orloski, 1 Paul Ettestad, 2 Ted L. Brown, 3 Pamela J. Reynolds, 3 W. John Pape, 4 Curtis L. Fritz, 5 Leon G. Carter, 1 and John D. Stein 1 From the 1 Bacterial Zoonoses Branch, Division of Vector-Borne Infectious Diseases, National Center for Infectious Diseases, Centers for Disease Control and Prevention, Fort Collins; 2 Colorado Department of Public Health and Environment, Denver, Colorado; New Mexico Departments of 3 Health and 4 Environment, Santa Fe, New Mexico; and 5 California Department of Health Services, California Exposure to cats infected with Yersinia pestis is a recently recognized risk for human plague in the US. Twenty-three cases of cat-associated human plague (5 of which were fatal) occurred in 8 western states from 1977 through 1998, which represent 7.7% of the total 297 cases reported in that period. Bites, scratches, or other contact with infectious materials while handling infected cats resulted in 17 cases of bubonic plague, 1 case of primary septicemic plague, and 5 cases of primary pneumonic plague. The 5 fatal cases were associated with misdiagnosis or delays in seeking treatment, which resulted in overwhelming infection and various manifestations of the systemic inflammatory response syndrome. Unlike infections acquired by flea bites, the occurrence of cat-associated human plague did not increase significantly during summer months. Plague epizootics in rodents also were observed less frequently at exposure sites for cases of cat-associated human plague than at exposure sites for other cases. The risk of cat-associated human plague is likely to increase as residential development continues in areas where plague foci exist in the western US. Enhanced awareness is needed for prompt diagnosis and treatment. Plague is a bacterial zoonosis caused by infection with Yersinia pestis, a gram-negative coccobacillus. Active foci exist in wild rodent and flea populations in many regions of the world, including the western US (where the disease probably was first introduced in by rat- and flea-infested ships) [1 3]. To date, surveillance programs have identified evidence of Y. pestis infection in mammal or flea samples collected from 17 western states (K.L.G., Plague Section, Centers for Disease Control and Prevention [CDC], unpublished data). A total of 297 human cases of plague were identified in 13 of these states from 1977 through Most cases of human plague in the US result from bites by infectious fleas, but nearly 20% of all cases with identified modes of transmission are acquired through contact with Y. pestis infected mammals, including domestic cats (Felis catus) [4 8]. The first well-documented case of cat-associated human plague in the US occurred in 1977 in Arizona [7]. Since then, 22 further cases have been identified (total of 23 cases in the 22-year period from 1977 through 1998), which suggests that exposure to infected cats is a significant plague risk in the US. Cases of cat-associated human plague are noteworthy because Received 18 May 1999; revised 4 December 1999; electronically published 13 June Reprints or correspondence: Dr. Kenneth L. Gage, Centers for Disease Control and Prevention, P. O. Box 2087, Fort Collins, CO Clinical Infectious Diseases 2000;30: by the Infectious Diseases Society of America. All rights reserved /2000/ $03.00 they have a broad range of clinical presentations (including primary pneumonic plague) and unusual circumstances of exposure. Further, cases appear to be largely restricted to the US or have yet to be recognized routinely in other countries. Thornton et al. [9] reported a single case from South Africa, and Simpson [10] reported another likely case from that country. Mall and O Leary [11] also presented anecdotal evidence of catassociated human plague in Argentina and Brazil. The purposes of this article are to review the epidemiological, ecological, and clinical aspects of the 23 cases of cat-associated human plague identified to date in the US, to provide detailed discussions of 4 recently identified cases of cat-associated human plague, and to discuss the risks cats may pose to their owners, veterinary staff, and other persons. Although a small number of cases of cat-associated human plague have been reported by other researchers, this article is the first comprehensive review of this topic [7, 12 16]. The review is intended to increase recognition and understanding of the problem and thereby reduce further preventable morbidity and deaths. Methods Under the provisions of the International Health Regulations for quarantinable diseases (plague, yellow fever, and cholera), all cases of plague must be investigated and reported to the World Health Organization [8]. Case investigations in the US involve the cooperative efforts of the CDC and state and local public health agencies. Records of these investigations are maintained in the Plague Section at the CDC. Our study included all cases of catassociated human plague in the CDC records that met the following
2 894 Gage et al. CID 2000;30 (June) Table 1. Summary of data on cases of cat-associated human plague in the US, Case State, county Date of onset Age (y), sex, ethnicity of patient Outcome Clinical presentation Type of exposure 1 AZ, Coconino 6/13/77 23, F, W Recovered Bubonic (cervical) Face-to-face contact 2 NM, Cibola 9/6/77 6, M, W Recovered Bubonic (axillary) a Bite or scratch 3 NV, Douglas 12/25/78 77, F, W Died Septicemic Cared for sick cat 4 CA, El Dorado 10/2/80 47, F, W Died Pneumonic Inhalation 5 CO, Jefferson 4/15/81 49, M, W Recovered Bubonic (axillary) Bite (veterinarian) 6 WY, Laramie 6/22/82 22, F, W Recovered Pneumonic Inhalation (veterinary technician) 7 OR, Jefferson 8/15/82 10, F, NA Recovered Bubonic (axillary) Scratched by cat that presumably had Yersinia pestis on its claws [8] 8 NM, McKinley 10/2/83 14, M, H Recovered Bubonic (axillary) Handled sick cat 9 NM, McKinley 3/26/84 11, M, NA Recovered Bubonic (axillary) Cared for sick cat 10 CA, Los Angeles 3/30/84 35, M, W Recovered Bubonic (axillary) Cared for sick cat 11 CA, Kern 5/3/84 24, M, W Died Bubonic (axillary) Buried dead cat that had been sick 12 NM, Rio Arriba 3/28/87 27, M, W Recovered Bubonic (axillary) Bite 13 AZ, Apache 11/16/87 9, M, NA Recovered Bubonic (axillary) Handled and slept with sick cat 14 CO, Montrose 10/19/91 41, F, W Recovered Bubonic (axillary) Cared for sick cat (veterinary technician) 15 NM, San Miguel 7/13/92 28, M, H Recovered Bubonic (axillary) Bite 16 CO, Chaffee 8/23/92 31, M, W Died Pneumonic Inhalation; face-to-face contact while retrieving sick cat from under house 17 NM, Sandoval 3/13/93 44, M, W Recovered Bubonic (axillary Scratch and epitrochlear) 18 CO, Boulder 5/19/93 31, F, W Recovered Pneumonic Inhalation (veterinarian) 19 UT, Box Elder 7/6/94 15, F, W Recovered Pneumonic Inhalation; face-to-face contact 20 CO, Delta 8/17/96 16, F, W Died Bubonic (axillary) Cared for sick cat with submandibular abscess 21 CA, Modoc 8/7/97 58, M, W Recovered Bubonic (axillary) Scratch (veterinarian) 22 NM, Santa Fe 5/19/98 45, F, W Recovered Bubonic (axillary) Bite (veterinarian) 23 CO, Archuleta 6/10/98 39, F, W Recovered Bubonic (axillary) Cared for sick cat NOTE. W, white; H, Hispanic; NA, Native American. a Bilateral axillary lymphadenopathy. 2 criteria: development of clinical plague within 10 days after a cat considered to be infected with Y. pestis was handled (22 cases) or after a scratch by a cat believed to have had viable Y. pestis on its claws (case 7) and epidemiological evidence suggesting that other exposures were unlikely [12]. All case records were reviewed to identify dates of onset of illness, the patient s age, sex, ethnicity, the primary clinical presentation, secondary complications and other clinical manifestations, the outcome of the illness (recovered or fatal), the likely mode of transmission, and information on exposure sites for patients and the cats they had handled. Additional data were gathered from reports of on-site environmental investigations, including whether plague epizootics (outbreaks in susceptible species of rodents) had occurred at the exposure sites. Plague epizootics were considered to have occurred when Y. pestis infection was identified in typical epizootic hosts (ground squirrels, prairie dogs, chipmunks, or wood rats) or their fleas or when observers noted inactive burrows or nests of these animals [1]. Certain information for 7 of the 23 cases has been published previously in isolated case reports or has been discussed briefly by other investigators [7, 12 16]. We include here selected data from the CDC records on these 7 cases for the sake of completeness and to present information absent from the published reports. Results Evidence for Y. pestis in cats associated with cases of human plague. Twenty-two of the 23 cats associated with the human cases of plague described in this review were considered to have had plague: Y. pestis was isolated from 5 cats (human cases 1, 2, 3, 4, and 19); 5 culture-negative cats were strongly seropositive by passive hemagglutination assays (titers to the F1 surface antigen of Y. pestis 1024; 4 cats were positive for Y. pestis by fluorescent antibody assays that used a rabbit polyclonal antibody that was specific for F1 antigen and conjugated to fluroescein isothiocyanate; and 8 cats had disappeared or had been killed before the arrival of investigators (human cases 9, 10, 11, 13, 14, 15, 16, and 21). Each of the 8 missing animals, however, had exhibited signs and symptoms of illness strongly suggestive of plague. The remaining cat (human case 7) did not exhibit evidence of Y. pestis infection but was thought to have had Y. pestis on its claws as a result of fighting with a cat that had a Y. pestis positive cervical abscess [12]. Case Summaries The 23 cat-associated cases of human plague identified in the US are summarized in table 1. These cases occurred in 8 western states, including New Mexico (7 cases), Colorado (6), California (4), Arizona (2), Nevada (1), Oregon (1), Utah (1), and Wyoming (1). Illness onset was in every month except January and February; there was no clear increase in incidence during the summer months of June, July, and August. The median age of the patients was 28 years (range, 6 77 years) (table 1). Eighteen patients were white, 3 were Native American, and 2 were Hispanic; 12 (52.2%) of the 23 were male. Seventeen patients
3 CID 2000;30 (June) Cats and Human Plague 895 (73.9%) presented with manifestations of bubonic plague; 16 of these patients had axillary buboes, and 1 had a cervical bubo. Only 1 patient (4.3%) presented with primary septicemic plague. Five patients (21.7%) developed primary pneumonic plague, apparently as a result of inhaling infectious materials from cats with pneumonic plague or oral lesions (table 1). Of the 18 patients presenting with bubonic or septicemic plague, 8 had incurred antecedent scratches or bites, including 1 patient (case 7) who was scratched near her left nipple by a cat while intervening in a fight between this animal and another cat that had a Y. pestis infected abscess on its neck (table 1) [12]. The remaining 10 patients who developed either septicemic plague (1 case) or bubonic plague (9 cases) had potential exposures while examining or caring for ill cats, sleeping with an ill cat, or burying a dead cat (table 1). We compared the 23 patients with cat-associated human plague with the remaining 274 patients whose cases occurred during the same period ( ) with respect to the following data: the patients sex and ethnicity and the state of exposure, initial clinical presentation, outcome (fatal or recovered), bubo location (if present), and likely route of exposure (inhalation, direct contact, or not determined). Probable modes of transmission were determined for 228 of the 274 cases without known infective cat exposures. Only 2 of these 228 patients were thought to have acquired plague through inhalation of infectious materials (unknown sources for these 2 cases), compared with 5 of the 23 patients with cat-associated cases (P!.0001; Fisher s exact test). As expected, the proportion of patients who developed primary pneumonic plague also was higher among patients with cat-associated cases than among other patients (5 of 23 vs. 2 of 274, respectively; P!.0001; 2- tailed Fisher s exact test). Axillary buboes were observed much more frequently in cat-associated cases of bubonic plague (16 of 17 cases) than in other cases (76 of 210 cases with identified bubo locations; P!.0001; x 2 test). None of the cat-associated cases included femoral or inguinal buboes, which occurred in most patients (52.9%) who acquired plague from other sources. Significant differences could not be demonstrated between catassociated cases and other cases with respect to the relative proportion of cases of bubonic plague and septicemic plague, gender, ethnicity, state of exposure, and outcomes ( P 1.05; x 2 or Fisher s exact tests, as appropriate). Incubation periods for cat-associated cases were similar in range (2 10 days) to those that have been reported for plague cases in general [17], but these cases could not be compared directly with most cases in our files because of the difficulties in determining precise exposure times for cases associated with flea bites or the lack of sufficiently detailed information in the case files. Ecological Investigations The methods used in ecological investigations and the intensity of sampling at sites where cats were believed to have acquired infection varied between cases. However, in all but 2 cases (14 and 18), blood samples for serology were obtained from pet dogs or cats, rodents, rabbits, or wild carnivores, and attempts were made to collect fleas from trapped rodents or rabbits and from rodent burrows or nests. At 6 sites, Y. pestis infection was identified in various highly susceptible epizootic rodent hosts (or their fleas), including ground squirrels, prairie dogs, chipmunks, or wood rats (cases 4, 7, 8, 10, 16, and 18). Inactive rodent burrows suggestive of epizootics were noted at another 5 sites (cases 6, 9, 11, 14, and 20). On the basis of these observations, plague epizootics were thought to have occurred at only 11 (47.8%) of the 23 sites where cats became infected with Y. pestis before passing their infections on to humans. By comparison, similar evidence of epizootic activity was observed at 188 (68.6%) of the 274 exposure sites for human cases that were not associated with cats but had occurred during the same period ( ; P=.0417; x 2 test). Y. pestis infection also was identified at 2 sites in native mice (Peromyscus species), which often are considered to be partially resistant to plague (enzootic or maintenance hosts), and in a Peromyscus-infesting flea (Aetheca wagneri) at another site [1]. In addition to infected rodents, seropositive samples were obtained at 7 sites from dogs and at 2 sites from coyotes. Description of Selected Cases Because there is not space here to describe each of the 23 cases of cat-associated human plague in detail, we have selected 4 recent cases as instructive examples. Three of these patients presented with bubonic plague (cases 17, 21, and 23), and 1 presented with primary pneumonic plague (case 18). Case 17: bubonic plague with axillary lymphadenopathy. This case occurred in a patient whose cat was treated by a veterinarian for an ulcerative oral lesion. Plague was not suspected, and the cat was treated with an oral tetracycline formulation. On the following day, the patient, who had undergone splenectomy, was scratched on the left arm while attempting to medicate the cat. Three days later, he had fatigue, prostration, diffuse myalgia, low-grade fever, nausea, and an episode of vomiting. The patient initially attributed these symptoms to chemotherapy (vinblastine sulfate and daily doses of prednisone) for idiopathic thrombocytopenic purpura. He also developed redness, induration, and sensitivity to touch in his upper left arm. Two days after the onset of illness, he presented to an emergency department with fever, an enlarged left axillary lymph node (1 cm in diameter), and swelling of the distal epitrochlear node. Laboratory tests indicated an elevated leukocyte count (18,300/mm 3 ) with a high proportion of immature neutrophils (27% band forms). The initial diagnosis was cellulitis with lymphangitis caused by a cat scratch and a possible relapse of idiopathic thrombocytopenic purpura. He was hospitalized and treated with ceftriaxone, an increased dosage of prednisone,
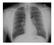
4 896 Gage et al. CID 2000;30 (June) and a second dose of vinblastine sulfate. Four days after the onset of illness, his temperature had returned to normal, and cellulitis with lymphangitis had begun to resolve. On the following day, culture of blood obtained at admission yielded Y. pestis. Because ceftriaxone and other b-lactam antibiotics are not considered effective against Y. pestis in vivo, the patient s treatment was changed to intravenous gentamicin and oral doxycycline. He was released from the hospital after 6 days and recovered without complications. The patient s mobile home was situated among other widely spaced ( m) homesites in a semiarid grassland habitat containing scattered juniper trees, cholla, and 4-winged saltbush. His homesite had numerous piles of lumber, scrap metal, and other potential rodent harborage, as did other nearby residences. None of the live rodents captured at the site was seropositive, and fleas infesting these animals were negative for Y. pestis by mouse inoculation assay. Y. pestis was, however, identified in 2 white-throated wood rat carcasses collected within 100 m of the patient s home. A flea (Orchopeas sexdentatus) recovered from 1 of these carcasses was positive for Y. pestis by mouse inoculation assay. Investigators also recovered 3 species of fleas from a wood rat nest located!10 m from the patient s home. A pool of 1 of these fleas (Megarthroglossus divisus) was found to be infected with Y. pestis by mouse inoculation assay. These findings indicated that an epizootic had occurred among the local wood rats, and it is likely that the cat became infected through contact with these animals. Abandoned rock squirrel burrows (20 25 burrows) were noted in an arroyo located about 100 m from the patient s house, which suggests that these animals also were affected by the epizootic. Case 21: bubonic plague with axillary lymphadenopathy. This case occurred in a veterinarian who visited a ranch where he was scratched while examining a moribund cat that had a submandibular abscess, rectal temperature of 35.0 C, and dehydration. Two days later, the veterinarian developed fever (temperature, 40.0 C) and a swollen and tender left axillary lymph node (5 cm in diameter). Upon consulting a physician, he was noted to have hypotension (blood pressure, 80/40 mm Hg; pulse rate, 48) and leukocytosis (leukocyte count, 31,000/ mm 3 ); he was hospitalized and treated with intravenous ceftriaxone. Tetracycline therapy was begun 2 days after admission and continued after his discharge on the same day. Although this patient recovered completely, he was dissatisfied with the unspecified diagnosis and consulted a second physician who obtained a serum sample about 3 weeks after the onset of illness. The sample was forwarded to the California Department of Health Service s Microbial Diseases Laboratory (Berkeley) and the CDC, where it was reported to be positive for Y. pestis at a titer of 256 in passive hemagglutination assays. Six weeks later, the patient s titer had increased 4-fold to 1024, which confirmed that his illness was plague [18]. Several weeks later, investigators visited the site where the cat had lived but found no evidence of epizootic activity. Residents did indicate, however, that another cat from this site had become ill and died with symptoms resembling those displayed by the cat that scratched the patient. The carcasses of both cats had been disposed of many weeks earlier and were unavailable for analysis. Case 23: bubonic plague with axillary lymphadenopathy. This case occurred in a patient who probably became infected while cleaning a draining abscess on the head of her cat. The cat, which had been ill for many days, was taken to a veterinary clinic where it died. The veterinarian considered plague as a possible diagnosis but decided against it because the abscess was located on the top of the cat s head rather than in the submandibular lymph nodes, which is more typical for cats infected with Y. pestis. Two days after last contact with the cat, the patient developed aching and throbbing in the left arm. Over the next 3 days, her symptoms increased, and she had increasingly severe left axillary pain, fever (temperature as high as 40.0 C), chills, and myalgia. On day 4 after the onset of illness, she presented to an emergency department complaining of severe left axillary, chest, and back pain, an enlarged left axillary lymph node (2 cm in diameter), fever, chills, diffuse myalgias, and persistent nausea and vomiting without diarrhea. Her temperature was 38.2 C, and her chest radiogram was clear. Blood was collected for culture, and seropurulent fluid, which contained some WBCs but no obvious microorganisms, was aspirated from the left axillary region. Her WBC count was 8930/mm 3 with a left shift. The patient mentioned the veterinarian s concern that her cat s illness might have been plague, and she was treated with ampicillin and gentamicin, along with supportive therapy. On day 6 after the onset of illness, the patient was transferred to a larger health care facility with a suspected diagnosis of Staphylococcus-related cellulitis and necrotizing fasciitis; treatment with gentamicin was discontinued, and streptomycin and tobramycin therapy was initiated. Culture of blood obtained on day 4 after the onset of illness subsequently yielded a gramnegative rod that was confirmed to be Y. pestis. The patient was discharged after 3 days and recovered completely. She lived in a mobile home park in a semirural area. Her cat reportedly hunted small rodents and was allowed to roam freely in a rocky canyon and other nearby natural areas. An ecological investigation did not identify signs of epizootic plague activity. Although numerous pet cats roamed the area, only the patient s cat was reported to have been ill. Tissue samples obtained from the exhumed carcass of this animal were positive for Y. pestis by fluorescent antibody assay. Case 18: primary pneumonic plague. This case occurred in a veterinarian who examined a severely ill cat that had a small lesion on its lower lip and was febrile and dehydrated. Plague was not considered as a possible diagnosis for the animal, which died several hours after the examination. Four days later, the veterinarian developed fever, myalgia, and malaise. On the following day, her condition worsened, and she sought emergency
5 CID 2000;30 (June) Cats and Human Plague 897 Table 2. Case Summary of data on fatal cases of cat-associated human plague. Clinical presentation 3 Septicemic (primary) 4 Pneumonic (primary) 11 Bubonic (axillary) 16 Pneumonic (primary) 20 Bubonic (axillary) NOTE. Initial diagnosis (place and time of diagnosis) Asthmatic attack, chronic obstructive pulmonary disease, sepsis (ER) Urinary tract infection (outpatient, day 1 after onset); pneumonia (ER, day 3 after onset) Cellulitis Gastroenteritis (outpatient day 1 after onset); cyanosis and septic shock (ER, day 3 after onset) Injury due to a fall (outpatient, day 3 after onset); septicemia, DIC, ARDS, and possible meningitis (ER, day 5 after onset) Method of diagnosis of Yersinia pestis infection (time of diagnosis) Antimicrobial therapy recommended Interval between start of appropriate therapy and diagnosis Bacterial culture (PM) Gentamicin 8 h before death and PM diagnosis Bacterial culture (PM) Tetracycline 3 d before death and PM diagnosis (during outpatient visit); might not have received proper doses because of vomiting Bacterial culture (day 11 after onset; day 5 of hospitalization) Tetracycline, gentamicin; streptomycin, chloramphenicol 2 d before correct diagnosis (tetracycline and gentamicin); day of correct diagnosis (streptomycin and chloramphenicol) Complications, reference Respiratory failure, acidosis, hypoxemia, and irreversible shock complicated by chronic obstructive pulmonary disease Severe hypoxemia, metabolic acidosis, and heart failure [10] Thrombocytopenia, gastrointestinal bleeding, lactic acid acidosis, ARDS, and cardiac arrest Bacterial culture (PM) None Died before correct diagnosis Cyanosis, septic shock, and right upper lobe pneumonia [11] Bacterial culture (PM) None Died before correct diagnosis DIC, ARDS, and possible meningitis [12] ARDS, acute respiratory distress syndrome; DIC, disseminated intravascular coagulation; ER, emergency room; PM, postmortem. medical care, complaining of vomiting as well as persistent fever (temperature, 40.6 C), myalgia, and malaise. Her leukocyte count was 15,600/mm 3 with a left shift, and her blood pressure was 90/50 mm Hg. A chest radiogram revealed right lower lobe pneumonia. She was hospitalized, and treatment with cephalosporin antibiotics and supportive therapy were started; 7 hours later, she became hypotensive and hypoxic, requiring intubation, mechanical ventilation, and vasopressor therapy. On day 2 after the onset of illness, a diagnosis of plague was considered, and antibiotic therapy was changed to include gentamicin and doxycycline. On day 3, a bronchoscopic washing was positive for Y. pestis by fluorescent antibody assay, as was a sputum sample obtained on day 4. The patient remained intubated for 10 days; however, her condition gradually improved, and she was discharged from the hospital 17 days after the onset of illness. The right-sided pulmonary infiltrate was still present at discharge but resolved within the following 3 weeks, and the patient made a full recovery. Eighty-five members of the hospital staff and 18 family members or friends were identified as having had significant contact with the patient; these contacts underwent either fever surveillance or prophylactic antibiotic treatment. Most of the contacts received prophylaxis with doxycycline, which is recommended for such use [8, 19]. Three of the contacts developed fever within 6 days of exposure to the patient, including the physician who had performed the intubation and the patient s 2 children. For postexposure prophylaxis, the physician received doxycycline, and the 2 children were given trimethoprim-sulfamethoxazole; all were negative for antibodies to the F1 surface antigen of Y. pestis when tested 6 7 months after exposure. A bone marrow sample from the exhumed cat s carcass was positive for Y. pestis by fluorescent antibody assay. The carcass was not evaluated for evidence of pneumonia, but the veterinarian who acquired plague from this animal reported that it did not exhibit coughing or other symptoms of respiratory illness. An inspection of the site where the cat lived did not reveal any obvious signs of epizootic plague activity. Fatal Cases of Cat-Associated Human Plague Five (21.7%) of the 23 cases of cat-associated human plague were fatal (table 2). Two of these patients had primary pneumonic plague (cases 4 and 16), 1 had septicemic plague (case 3), and 2 had bubonic plague with axillary buboes (cases 11 and 20). Each of these patients initially was considered to have an illness other than plague (table 2). Four of the 5 decedents consulted physicians 1 3 days before being admitted to a hospital and were prescribed the following: tetracycline (case 4), an antibiotic considered effective for treatment of plague; antibiotics other than those recommended for treating human plague (dicloxacillin [case 11] and ciprofloxacin [case 16]); medications to relieve nausea and vomiting (case 4); and analgesics for relief of symptoms thought to be related to injury (case 20) [13 15, 19]. Although 1 patient (case 4) was prescribed treatment with an appropriate antibiotic (tetracycline) on the day after the onset of illness, her condition rapidly deteriorated,
6 898 Gage et al. CID 2000;30 (June) and she died within 4.5 hours after being admitted to a hospital on day 2 after the onset of illness. Reportedly, she took only 2 of the 4 prescribed doses of tetracycline on her first day of illness, and these doses might not have been retained because of vomiting [13]. The admitting diagnosis for this patient was pneumonia, and she was treated with penicillin (not recommended as therapy for plague). Only 1 of the patients who died (case 3) was admitted to a hospital during the first attempt to obtain medical assistance. This individual complained of difficulty in breathing and flulike symptoms on her second day of illness and was taken by ambulance to an emergency department. Physical examination revealed that in addition to dyspnea the patient had peripheral cyanosis and a WBC count of 21,800 cells/mm 3. At admission, she was given supportive respiratory therapy, as well as a single dose of gentamicin for treatment of undiagnosed septicemia. Shortly before her death (8 hours after admission), the hospital laboratory reported observing bacilli on a peripheral blood smear, which is considered a grave prognostic sign [17]. Case 11 was the only fatal case to be correctly diagnosed before the patient died. During his first 2 days of hospitalization, the patient was treated with nafcillin, which is not recommended for treatment of plague. On the third day of hospitalization (day 9 after the onset of illness), gram-negative rods were noted in sputum samples from the patient, and he was treated with tetracycline, gentamicin, and cefotaxime. Two days later, a diagnosis of Y. pestis infection was confirmed by bacterial culture (day 11 after the onset of illness), resulting in treatment with streptomycin and chloramphenicol. Secondary complications associated with these cases included various sequelae of the systemic inflammatory response syndrome, such as metabolic acidosis, acute respiratory distress syndrome, disseminated intravascular coagulation and bleeding, hypoxemia, and cardiac arrest (table 2). Discussion Physicians in areas in the West where plague is enzootic should be informed of the risks of cat-associated human plague and consider plague as a possible diagnosis for persons who have handled sick or dead cats and present with fever and chills, myalgias, arthralgias, headache, or prostration. The presence of inguinal, femoral, axillary, or cervical lymphadenopathy should further increase the suspicion of plague, as should the identification of septicemia or pneumonia. Persons who acquire plague as a result of handling infected cats are most likely to develop axillary or cervical lymphadenopathy (table 1), while those infected through flea bites typically develop inguinal or femoral lymphadenopathy. Misdiagnoses and delays in treatment often lead to fatalities, as demonstrated by the 5 cases summarized in table 2. As soon as plague is suspected, diagnostic samples should be obtained, and patients should receive appropriate antimicrobial therapy. The drug of choice is streptomycin, although gentamicin is an acceptable alternative [19]. Tetracyclines and chloramphenicol also are considered to be effective. Because of its ability to penetrate tissues, chloramphenicol is recommended for cases involving plague meningitis, pleuritis, endophthalmitis, and myocarditis. Appropriate diagnostic samples include blood and other potentially infectious fluids or tissues. Isolation of Y. pestis from such samples by culture typically requires 2 days, but a presumptive diagnosis often can be made by using a fluorescent antibody assay to detect the F1 surface antigen of Y. pestis. Samples also should be obtained for serology, but detectable titers might not develop until many days after the onset of illness. Since 1977, cases of cat-associated human plague have occurred almost every year in the western US, which emphasizes the importance of considering plague as a possible diagnosis for persons who have compatible symptoms and a history of exposure to cats. This diagnosis should especially be considered in the 4 states (Arizona, California, Colorado, and New Mexico) that reported 19 of the cat-associated cases and 89% of all cases occurring in the US from 1977 through 1998 [8, 20]. Populations in these states are increasing, and housing developments continue to expand into rural areas containing active plague foci. Furthermore, cat owners in these high-risk areas often allow their animals to roam freely, greatly increasing the likelihood that cats will encounter infectious fleas or eat Y. pestis infected animals, particularly deer mice or other small rodents [21 23]. These factors suggest that human exposures to Y. pestis infected cats in western states will increase. The occurrences of vector-borne diseases are often highly seasonal because of annual variations in vector or host activity patterns that affect the likelihood of pathogen transmission. In the western US, most persons acquire plague through the bites of infectious fleas, and the risks of such exposures increase markedly during the summer months when epizootic transmission among rodents and their fleas also is at a peak [1, 20]. It is interesting that the occurrence of cases of cat-associated human plague did not appear to increase significantly during the summer months. Cat-associated cases also were less likely than other cases to be associated with epizootic activity. These observations are perhaps not surprising, as cats are likely to become infected as a result of eating native mice or voles (Peromyscus species or Microtus species), which have been proposed to be partially resistant to plague and are presumed to serve, along with their fleas, as reservoirs (maintenance or enzootic hosts) of Y. pestis infection during interepizootic periods. Cats that consume these animals might, therefore, become infected with Y. pestis at any time of year and then expose humans to plague. These observations suggest that a diagnosis of plague should be considered, regardless of season or levels of local epizootic plague activity, for persons who have compatible symptoms and a history of exposure to a sick or dead cat in the western US.
7 CID 2000;30 (June) Cats and Human Plague 899 Persons who handle Y. pestis infected cats can acquire plague through a variety of means (table 1). All patients in our study had a history of close physical contact with cats (handling), which resulted in these persons being bitten or scratched by teeth or claws believed to be contaminated with Y. pestis (8 cases), being exposed to presumably infectious body fluids while caring for, examining, or burying sick cats (10), or being exposed to presumably infectious respiratory droplets or other airborne infectious materials during face-to-face contact with the cats (5). The epidemiological investigations did not suggest that these persons acquired plague through infectious flea bites, although we cannot exclude the possibility that patients were exposed to wild rodent fleas as a result of handling flea-infested cats. If flea exposures did occur, it is highly unlikely that they were associated with the bites of cat fleas (Ctenocephalides felis), which infest cats in some regions of the US but are extremely rare or absent in the primarily semiarid regions where the cases in our study occurred. Repeated experimental studies also have demonstrated that cat fleas are very poor vectors of plague [24]. Cats that hunt are also likely to bring infected rodents into or near human habitations, which could pose a risk for persons who dispose of, or otherwise come into direct contact with, these animals. The timely diagnosis of primary pneumonic plague in persons who have handled potentially infectious cats is extremely important because this form of the disease is associated with a high fatality rate and has potential for person-to-person spread [1, 17, 25]. Of the 23 patients described in our review, 5 developed primary pneumonic plague as a result of inhaling infectious respiratory droplets or airborne oral secretions during face-to-face contact with infected cats (table 1). These 5 cases are noteworthy because only 2 other cases (7 total) of primary pneumonic plague have been identified in the US since Sources of infection for the latter 2 cases could not be determined, but contact with infected cats cannot be ruled out, particularly for a fatal case that occurred in a California veterinarian in 1977 [26]. Persons who have close contact (!2 m) with infected cats or human patients with symptoms of pneumonic plague should be advised of the risk of airborne exposure to plague and given antibiotic prophylaxis. For prophylaxis, doxycycline or tetracycline is recommended for persons aged 9 years, and trimethoprim-sulfamethoxazole is preferred for children aged!9 years [8, 19]. Plague vaccine is no longer commercially available in the US and is not suitable for postexposure prophylaxis. Vaccination is recommended only for persons working in microbiological research laboratories or for certain individuals, such as mammalogists, who routinely handle the rodent hosts or flea vectors of plague [8]. Human exposures to Y. pestis infected cats frequently occur during care for an ill cat (table 1). Infected cats have a spectrum of illness similar to that for people, with high rates of mortality among untreated animals (38% in 1 experimental study) [21, 22, 27, 28]. These animals typically present with fever, lethargy, anorexia, and an enlarged lymph node or bubo (frequently submandibular) that may be abscessed [22]. Pneumonic plague was described in 10% of Y. pestis infected cats in a New Mexico study [22]. Because of the severity of cat plague, ill animals are often taken to a veterinary clinic for care, placing veterinarians and their staff at risk of exposure. Six (26.1%) of the 23 cases reported in this article occurred in veterinarians (4 cases) or their staff (2 cases). Two of these 6 patients developed primary pneumonic plague, and 4 had axillary buboes (which suggests inoculation of the hand or arm); none of these 6 cases was fatal. Kaufmann et al. [7] reported another likely case (culture confirmed; nonfatal) that occurred in 1936 in a California veterinarian who developed fever, chills, headache, myalgia, and an apparent axillary bubo 3 days after examining a cat with a submandibular abscess of unknown etiology. Veterinarians often fail to consider plague as a diagnosis for cats because they are not aware that plague exists in the regions where they practice. The likelihood of misdiagnosis also is increased by the fact that most veterinarians lack experience in diagnosing Y. pestis infections in cats. Submandibular buboes in cats are easily confused with similar looking abscesses that commonly arise from various causes, including fights with other animals. Under such circumstances, veterinarians may fail to take appropriate precautionary measures and are likely to prescribe antibiotics, such as penicillins, that are useful for treating cat fight abscesses but are ineffective against Y. pestis. Misdiagnosis of Y. pestis infected cats also can pose a further risk to their owners when they are sent home for additional care. Persons working in veterinary practices where plague is enzootic should maintain an awareness that plague can occur in cats. In general, veterinary staff who follow standard operating procedures for handling potentially infectious animals or materials should be adequately protected from Y. pestis infection. Such procedures should include wearing a well-fitted surgical mask, gloves, and eye protection. Exudates should be considered infectious, and any material used for treating suspect cats should be disinfected, autoclaved, or incinerated. Veterinary clinic personnel should be advised of these risks and encouraged to consult their physician and local or state health department in the event of possible exposure to an infected cat. Vaccination is not recommended for veterinarians or their staff (none of the veterinary personnel with plague who were described in this article had received plague vaccine) [8]. In conclusion, Y. pestis infected cats are expected to pose a continuing risk for persons living in those areas of the western US where Y. pestis exists in native rodent and flea populations. Prevention of cat-associated cases will require enhanced plague awareness among pet owners, veterinarians, health care providers, and the general public. These persons should be provided with appropriate information on the symptoms of plague in humans and cats, personal protective measures, and use of insecticidal agents to kill fleas on cats. Cat owners also should
8 900 Gage et al. CID 2000;30 (June) prevent their cats from roaming freely in plague-affected areas [1, 8]. General preventive measures should be taken in areas where cats or other animals are found to be infected. These measures, which are designed to reduce the likelihood that humans will be exposed to infectious fleas or rodents, include avoiding sick or dead animals, avoiding areas where recent plague epizootics have occurred, modifying human environments to reduce the amount of food and shelter available to rodents, and, in certain situations, having trained personnel apply insecticides to control fleas or, rarely, rodenticides to reduce rodent populations. Finally, state and local health departments in areas where plague is enzootic in the western US should maintain adequate plague surveillance programs to detect increased human risks associated with plague epizootics in rodents and cases of plague in domestic cats. References 1. Gage KL. Plague. In: Collier L, Balows A, Sussman M, Hausler WJ, eds. Topley and Wilson s microbiology and microbial infections. 9th ed. London: Arnold, 1998: Link VB9. A history of plague in the US of America [public health monograph no. 26]. Washington, DC: US Department of Health, Education, and Welfare, 1955: Barnes AM. Surveillance and control of bubonic plague in the US. In: Edwards MA, McDonnel U, eds. Animal disease in relation to conservation. New York: Academic Press, 1982: Poland JD, Barnes AM, Herman JJ. Human bubonic plague from exposure to a naturally infected wild coyote. Am J Epidemiol 1973;97: von Reyn CF, Barnes AM, Weber NS, Hodgin UG. Bubonic plague from exposure to a rabbit: a documented case, and a review of rabbit-associated plague cases in the US. Am J Epidemiol 1976;104: von Reyn CF, Barnes AM, Weber NS, Quan T, Dean WJ. Bubonic plague from direct exposure to a naturally infected wild coyote. Am J Trop Med Hyg 1976;25: Kaufmann AF, Mann JM, Gardiner TM, et al. Public health implications of plague in domestic cats. J Am Vet Med Assoc 1981;179: Centers for Disease Control and Prevention. Prevention of plague. Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep 1996;45(RR-14): Thornton DJ, Tustin RC, Piennar WN, Bubb JD. Cat bite transmission of Yersinia pestis infection to man. J S Afr Vet Assoc 1975;46: Simpson WJ. A treatise on plague. Cambridge, England: Cambridge University Press, 1905:104 15, Mall AA, O Leary SB. Plague in the Americas [publication no. 225]. Washington, DC: Pan American Sanitary Bureau, 1945:45, 65, Weniger BG, Warren AJ, Forseth V, et al. Human bubonic plague transmitted by a domestic cat scratch. JAMA 1984;251: Werner SB, Weidmer CE, Nelson BC, Nygaard GS, Goethals RM, Poland JD. Primary plague pneumonia contracted from a domestic cat at South Lake Tahoe, Calif. JAMA 1984;251: Doll JM, Zeitz PS, Ettestad P, Bucholtz AL, Davis T, Gage K. Cat-transmitted fatal pneumonic plague in a person who traveled from Colorado to Arizona. Am J Trop Med Hyg 1994;51: Centers for Disease Control and Prevention. Fatal human plague: Arizona and Colorado, MMWR Morb Mortal Wkly Rep 1997; 46: Cramer C, Christensen B. Pneumonic plague in a 15-year-old Utah girl. J Emerg Nurs 1995;21: Poland JD, Barnes AM. Plague. In: Steele JH, Stoenner H, Kaplan W, Torten M, eds. CRC handbook series in zoonoses. I. Boca Raton, FL: CRC Press, 1979: Centers for Disease Control and Prevention. Case definitions for infectious conditions under public health surveillance. MMWR Morb Mortal Wkly Rep 1997;46(RR-10): Campbell GL, Dennis DT. Plague and other yersinial infections. In: Fauci AS, Braunwald E, Isselbacher KJ, et al, eds. Harrison s principles of internal medicine. New York: McGraw-Hill, 1995: Craven RB, Maupin GO, Beard ML, Quan TJ, Barnes AM. Reported cases of human plague in the US, J Med Entomol 1993;30: Eidson M, Tierney LA, Rollag OJ, Becker T, Brown T, Hull HF. Feline plague in New Mexico: risk factors and transmission to humans. Am J Public Health 1988;78: Eidson M, Thilsted JP, Rollag OJ. Clinical, clinicopathologic, and pathologic features of plague in cats: 119 cases ( ). J Am Vet Med Assoc 1991;199: Gage KL, Ostfeld RS, Olson JG. Nonviral vector-borne zoonoses associated with mammals in the US. J Mammal 1995;76: Pollitzer R. Plague. Geneva: World Health Organization, 1954: Burmeister RW, Tigertt WD, Overholt EL. Laboratory-acquired pneumonic plague, report of a case and review of previous cases. Ann Intern Med 1962;56: Madon MB, Hitchcock JC, Davis RM, et al. An overview of plague in the US and a report of investigations of two human cases in Kern County, California, J Vector Ecol 1997;22: Rust JH, Cavanaugh DC, O Shita R, Marshall JD Jr. The role of domestic animals in the epidemiology of plague. I. Experimental infection of dogs and cats. J Infect Dis 1971;124: Gasper PW, Barnes AM, Quan TJ, et al. Plague (Yersinia pestis) in cats: description of experimentally induced disease. J Med Entomol 1993;30: 20 6.
Clinical Manifestations and Treatment of Plague Dr. Jacky Chan. Associate Consultant Infectious Disease Centre, PMH
 Clinical Manifestations and Treatment of Plague Dr. Jacky Chan Associate Consultant Infectious Disease Centre, PMH Update of plague outbreak situation in Madagascar A large outbreak since 1 Aug 2017 As
Clinical Manifestations and Treatment of Plague Dr. Jacky Chan Associate Consultant Infectious Disease Centre, PMH Update of plague outbreak situation in Madagascar A large outbreak since 1 Aug 2017 As
Running head: PLAGUE: WHAT EVERY NURSE NEEDS TO KNOW 1
 Running head: PLAGUE: WHAT EVERY NURSE NEEDS TO KNOW 1 Plague: What every nurse needs to know Nathon Kelley Ferris State University PLAGUE: WHAT EVERY NURSE NEEDS TO KNOW 2 Abstract Plague is not just
Running head: PLAGUE: WHAT EVERY NURSE NEEDS TO KNOW 1 Plague: What every nurse needs to know Nathon Kelley Ferris State University PLAGUE: WHAT EVERY NURSE NEEDS TO KNOW 2 Abstract Plague is not just
Feline zoonoses. Institutional Animal Care and Use Committee 12/09
 Feline zoonoses Institutional Animal Care and Use Committee 12/09 Cat scratch disease Bacterial infection caused by Bartonella henselae Associated with a cat bite or scratch Infection at point of injury,
Feline zoonoses Institutional Animal Care and Use Committee 12/09 Cat scratch disease Bacterial infection caused by Bartonella henselae Associated with a cat bite or scratch Infection at point of injury,
3 TREATMENT OF PLAGUE
 3 TREATMENT OF PLAGUE Dr Jack D. Poland and Dr D. T. Dennis Case management: therapy and prevention of spread When a diagnosis of human plague is suspected on clinical and epidemiological grounds, appropriate
3 TREATMENT OF PLAGUE Dr Jack D. Poland and Dr D. T. Dennis Case management: therapy and prevention of spread When a diagnosis of human plague is suspected on clinical and epidemiological grounds, appropriate
Zoonoses in West Texas. Ken Waldrup, DVM, PhD Texas Department of State Health Services
 Zoonoses in West Texas Ken Waldrup, DVM, PhD Texas Department of State Health Services Notifiable Zoonotic Diseases Arboviruses* Anthrax Brucellosis Bovine Tuberculosis Creutzfeldt-Jacob disease (variant)
Zoonoses in West Texas Ken Waldrup, DVM, PhD Texas Department of State Health Services Notifiable Zoonotic Diseases Arboviruses* Anthrax Brucellosis Bovine Tuberculosis Creutzfeldt-Jacob disease (variant)
Tularemia. Information for Health Care Providers. Physicians D Nurses D Laboratory Personnel D Infection Control Practitioners
 Tularemia Information for Health Care Providers Physicians D Nurses D Laboratory Personnel D Infection Control Practitioners Tularemia Caused by Francisella tularensis, a small, pleomorphic, gram-negative
Tularemia Information for Health Care Providers Physicians D Nurses D Laboratory Personnel D Infection Control Practitioners Tularemia Caused by Francisella tularensis, a small, pleomorphic, gram-negative
Welcome to Pathogen Group 9
 Welcome to Pathogen Group 9 Yersinia pestis Francisella tularensis Borrelia burgdorferi Rickettsia rickettsii Rickettsia prowazekii Acinetobacter baumannii Yersinia pestis: Plague gram negative oval bacillus,
Welcome to Pathogen Group 9 Yersinia pestis Francisella tularensis Borrelia burgdorferi Rickettsia rickettsii Rickettsia prowazekii Acinetobacter baumannii Yersinia pestis: Plague gram negative oval bacillus,
Biology and Control of Insects and Rodents Workshop Vector Borne Diseases of Public Health Importance
 Vector-Borne Diseases of Public Health Importance Rudy Bueno, Jr., Ph.D. Director Components in the Disease Transmission Cycle Pathogen Agent that is responsible for disease Vector An arthropod that transmits
Vector-Borne Diseases of Public Health Importance Rudy Bueno, Jr., Ph.D. Director Components in the Disease Transmission Cycle Pathogen Agent that is responsible for disease Vector An arthropod that transmits
RABIES CONTROL INTRODUCTION
 RABIES CONTROL INTRODUCTION Throughout human history, few illnesses have provoked as much anxiety as has rabies. Known as a distinct entity since at least 500 B.C., rabies has been the subject of myths
RABIES CONTROL INTRODUCTION Throughout human history, few illnesses have provoked as much anxiety as has rabies. Known as a distinct entity since at least 500 B.C., rabies has been the subject of myths
Medical Bacteriology- Lecture 14. Gram negative coccobacilli. Zoonosis. Brucella. Yersinia. Francesiella
 Medical Bacteriology- Lecture 14 Gram negative coccobacilli Zoonosis Brucella Yersinia Francesiella 1 Zoonosis: A disease, primarily of animals, which is transmitted to humans as a result of direct or
Medical Bacteriology- Lecture 14 Gram negative coccobacilli Zoonosis Brucella Yersinia Francesiella 1 Zoonosis: A disease, primarily of animals, which is transmitted to humans as a result of direct or
THE BUBONIC PLAGUE. Miss Hansen. 10th Grade World History
 THE BUBONIC PLAGUE Miss Hansen 10th Grade World History WAIT WHAT? The Bubonic Plague The Black Death The Plague THE BUBONIC PLAGUE Later named the Black Death Marks the outbreak from 1346 to 1352 Not
THE BUBONIC PLAGUE Miss Hansen 10th Grade World History WAIT WHAT? The Bubonic Plague The Black Death The Plague THE BUBONIC PLAGUE Later named the Black Death Marks the outbreak from 1346 to 1352 Not
Gentamicin and Tetracyclines for the Treatment of Human Plague: Review of 75 Cases in New Mexico,
 MAJOR ARTICLE Gentamicin and Tetracyclines for the Treatment of Human Plague: Review of 75 Cases in New Mexico, 1985 1999 L. Lucy Boulanger, 1,5 Paul Ettestad, 3 John D. Fogarty, 2,5 David T. Dennis, 6
MAJOR ARTICLE Gentamicin and Tetracyclines for the Treatment of Human Plague: Review of 75 Cases in New Mexico, 1985 1999 L. Lucy Boulanger, 1,5 Paul Ettestad, 3 John D. Fogarty, 2,5 David T. Dennis, 6
CLINICAL USE OF BETA-LACTAMS
 CLINICAL USE OF BETA-LACTAMS Douglas Black, Pharm.D. Associate Professor School of Pharmacy University of Washington dblack@u.washington.edu WHY IS INFECTIOUS DISEASE PHARMACOTHERAPY SO CONFUSING? Microbial
CLINICAL USE OF BETA-LACTAMS Douglas Black, Pharm.D. Associate Professor School of Pharmacy University of Washington dblack@u.washington.edu WHY IS INFECTIOUS DISEASE PHARMACOTHERAPY SO CONFUSING? Microbial
New Jersey Department of Health Rabies Background and Technical Information
 New Jersey Department of Health Rabies Background and Technical Information The History of Rabies Rabies is an ancient disease. It is well described in writings by Egyptians dating back to 2300 B.C. Rabies
New Jersey Department of Health Rabies Background and Technical Information The History of Rabies Rabies is an ancient disease. It is well described in writings by Egyptians dating back to 2300 B.C. Rabies
Canine and Feline Distemper. Description. The following chart indicates the animals which are susceptible to infection by canine and feline distemp
 Canine and Feline Distemper Description Canine and feline distemper are diseases affecting many wild and domestic carnivo The following chart indicates the animals which are susceptible to infection by
Canine and Feline Distemper Description Canine and feline distemper are diseases affecting many wild and domestic carnivo The following chart indicates the animals which are susceptible to infection by
Subacute Adenitis. Ann M. Loeffler, MD
 Subacute Adenitis Ann M. Loeffler, MD Lymphadenitis Swelling and hyperplasia of sinusoidal lining cells Infiltration of leukocytes +/- abscess formation Granulomatous or non-granulomatous Pyogenic adenitis
Subacute Adenitis Ann M. Loeffler, MD Lymphadenitis Swelling and hyperplasia of sinusoidal lining cells Infiltration of leukocytes +/- abscess formation Granulomatous or non-granulomatous Pyogenic adenitis
Outlines. Introduction Prevalence Resistance Clinical presentation Diagnosis Management Prevention Case presentation Achievements
 Amal Meas Al-Anizi, PharmD Candidate KSU, Infectious Disease Rotation 2014 Outlines Introduction Prevalence Resistance Clinical presentation Diagnosis Management Prevention Case presentation Achievements
Amal Meas Al-Anizi, PharmD Candidate KSU, Infectious Disease Rotation 2014 Outlines Introduction Prevalence Resistance Clinical presentation Diagnosis Management Prevention Case presentation Achievements
2014 Update of the odd Zoonotic Diseases on Navajo
 2014 Update of the odd Zoonotic Diseases on Navajo Dr. Scott Bender, DVM Tribal Veterinarian Navajo Nation Veterinary Program Navajo Nation Dept. of Agriculture Navajo Nation OR THE ANSWERS ARE: Flu like
2014 Update of the odd Zoonotic Diseases on Navajo Dr. Scott Bender, DVM Tribal Veterinarian Navajo Nation Veterinary Program Navajo Nation Dept. of Agriculture Navajo Nation OR THE ANSWERS ARE: Flu like
BRUCELLOSIS. Morning report 7/11/05 Andy Bomback
 BRUCELLOSIS Morning report 7/11/05 Andy Bomback Also called undulant, Mediterranean, or Mata fever, brucellosis is an acute and chronic infection of the reticuloendothelial system gram negative facultative
BRUCELLOSIS Morning report 7/11/05 Andy Bomback Also called undulant, Mediterranean, or Mata fever, brucellosis is an acute and chronic infection of the reticuloendothelial system gram negative facultative
Standard Operating Procedure for Rabies. November Key facts
 Standard Operating Procedure for Rabies November 2011 Key facts Rabies occurs in more than 150 countries and territories. Dogs are the source of 99% of human rabies deaths. Worldwide, more than 55 000
Standard Operating Procedure for Rabies November 2011 Key facts Rabies occurs in more than 150 countries and territories. Dogs are the source of 99% of human rabies deaths. Worldwide, more than 55 000
To describe step-wise procedures for the collection of live fleas from prairie dog burrows.
 Kaufman, V20170619 TITLE Collection of fleas from prairie dog burrows 1. PURPOSE To describe step-wise procedures for the collection of live fleas from prairie dog burrows. 2. SCOPE This SOP will cover
Kaufman, V20170619 TITLE Collection of fleas from prairie dog burrows 1. PURPOSE To describe step-wise procedures for the collection of live fleas from prairie dog burrows. 2. SCOPE This SOP will cover
General Approach to Infectious Diseases
 General Approach to Infectious Diseases 2 The pharmacotherapy of infectious diseases is unique. To treat most diseases with drugs, we give drugs that have some desired pharmacologic action at some receptor
General Approach to Infectious Diseases 2 The pharmacotherapy of infectious diseases is unique. To treat most diseases with drugs, we give drugs that have some desired pharmacologic action at some receptor
Guidelines for the Initiation of Empirical Antibiotic therapy in Respiratory Disease (Adults)
 Guidelines for the Initiation of Empirical Antibiotic therapy in Respiratory Disease (Adults) Community Acquired Community Acquired 1) Is it pneumonia? ie new symptoms and signs of a lower respiratory
Guidelines for the Initiation of Empirical Antibiotic therapy in Respiratory Disease (Adults) Community Acquired Community Acquired 1) Is it pneumonia? ie new symptoms and signs of a lower respiratory
ANTHRAX. INHALATION, INTESTINAL and CUTANEOUS ANTHRAX
 INHALATION, INTESTINAL and CUTANEOUS ANTHRAX CPMP/4048/01, rev. 3 1/7 General points on treatment Anthrax is an acute infectious disease caused by Bacillus anthracis, that may be infecting man via cutaneous
INHALATION, INTESTINAL and CUTANEOUS ANTHRAX CPMP/4048/01, rev. 3 1/7 General points on treatment Anthrax is an acute infectious disease caused by Bacillus anthracis, that may be infecting man via cutaneous
What s Your Diagnosis? By Sohaila Jafarian, Class of 2018
 Signalment: Greeley, 3 yo MC DSH Presenting Complaint: ADR History: What s Your Diagnosis? By Sohaila Jafarian, Class of 2018 Patient is an indoor/outdoor cat. Previously healthy and up to date on vaccines
Signalment: Greeley, 3 yo MC DSH Presenting Complaint: ADR History: What s Your Diagnosis? By Sohaila Jafarian, Class of 2018 Patient is an indoor/outdoor cat. Previously healthy and up to date on vaccines
Control emergence of drug-resistant. Reduce costs
 ...PRESENTATIONS... Guidelines for the Management of Community-Acquired Pneumonia Richard E. Chaisson, MD Presentation Summary Guidelines for the treatment of community-acquired pneumonia (CAP) have been
...PRESENTATIONS... Guidelines for the Management of Community-Acquired Pneumonia Richard E. Chaisson, MD Presentation Summary Guidelines for the treatment of community-acquired pneumonia (CAP) have been
General Surgery Small Group Activity (Facilitator Notes) Curriculum for Antimicrobial Stewardship
 General Surgery Small Group Activity (Facilitator Notes) Curriculum for Antimicrobial Stewardship Facilitator instructions: Read through the facilitator notes and make note of discussion points for each
General Surgery Small Group Activity (Facilitator Notes) Curriculum for Antimicrobial Stewardship Facilitator instructions: Read through the facilitator notes and make note of discussion points for each
Hand washing, Asepsis, Precautions and Infection Control
 Hand washing, Asepsis, Precautions and Infection Control FN Ch 12, NICS Ch4 Week 2 Lesa McArdle, MSN, RN Objectives Hand washing, Asepsis, Precautions & Infection Control Explain the chain of infection
Hand washing, Asepsis, Precautions and Infection Control FN Ch 12, NICS Ch4 Week 2 Lesa McArdle, MSN, RN Objectives Hand washing, Asepsis, Precautions & Infection Control Explain the chain of infection
DATA COLLECTION SECTION BY FRONTLINE TEAM. Patient Identifier/ Medical Record number (for facility use only)
 Assessment of Appropriateness of ICU Antibiotics (Patient Level Sheet) **Note this is intended for internal purposes only. Please do not return to PQC.** For this assessment, inappropriate antibiotic use
Assessment of Appropriateness of ICU Antibiotics (Patient Level Sheet) **Note this is intended for internal purposes only. Please do not return to PQC.** For this assessment, inappropriate antibiotic use
Suitability of Antibiotic Treatment for CAP (CAPTIME) The duration of antibiotic treatment in community acquired pneumonia (CAP)
 STUDY PROTOCOL Suitability of Antibiotic Treatment for CAP (CAPTIME) Purpose The duration of antibiotic treatment in community acquired pneumonia (CAP) lasts about 9 10 days, and is determined empirically.
STUDY PROTOCOL Suitability of Antibiotic Treatment for CAP (CAPTIME) Purpose The duration of antibiotic treatment in community acquired pneumonia (CAP) lasts about 9 10 days, and is determined empirically.
Breastfeeding Challenges - Mastitis & Breast Abscess -
 CLINICAL PRACTICE GUIDELINE Breastfeeding Challenges - Mastitis & Breast Abscess - SCOPE (Area): Maternity Unit, Emergency Department, Paediatrics SCOPE (Staff): Medical, Midwifery & Nursing DESIRED OUTCOME/OBJECTIVE
CLINICAL PRACTICE GUIDELINE Breastfeeding Challenges - Mastitis & Breast Abscess - SCOPE (Area): Maternity Unit, Emergency Department, Paediatrics SCOPE (Staff): Medical, Midwifery & Nursing DESIRED OUTCOME/OBJECTIVE
Ecology of RMSF on Arizona Tribal Lands
 Ecology of RMSF on Arizona Tribal Lands Tribal Vector Borne Disease Meeting M. L. Levin Ph.D. Medical Entomology Laboratory Centers for Disease Control mlevin@cdc.gov Rocky Mountain Spotted Fever Disease
Ecology of RMSF on Arizona Tribal Lands Tribal Vector Borne Disease Meeting M. L. Levin Ph.D. Medical Entomology Laboratory Centers for Disease Control mlevin@cdc.gov Rocky Mountain Spotted Fever Disease
RABIES PROPHYLAXIS REQUIREMENT FOR DVM STUDENTS
 RABIES PROPHYLAXIS REQUIREMENT FOR DVM STUDENTS In 2007 there was an increase in the number of terrestrial mammals with rabies in the state of Colorado. Rabies is often a fatal disease and our students
RABIES PROPHYLAXIS REQUIREMENT FOR DVM STUDENTS In 2007 there was an increase in the number of terrestrial mammals with rabies in the state of Colorado. Rabies is often a fatal disease and our students
Clinical Practice Standard
 Clinical Practice Standard 1-20-6-1-010 TITLE: INTRAVENOUS TO ORAL CONVERSION FOR ANTIMICROBIALS A printed copy of this document may not reflect the current, electronic version on OurNH. APPLICABILITY:
Clinical Practice Standard 1-20-6-1-010 TITLE: INTRAVENOUS TO ORAL CONVERSION FOR ANTIMICROBIALS A printed copy of this document may not reflect the current, electronic version on OurNH. APPLICABILITY:
Invasive Group A Streptococcus (GAS)
 Invasive Group A Streptococcus (GAS) Cause caused by a bacterium commonly found on the skin and in the throat transmitted by direct, indirect or droplet contact with secretions from the nose, and throat
Invasive Group A Streptococcus (GAS) Cause caused by a bacterium commonly found on the skin and in the throat transmitted by direct, indirect or droplet contact with secretions from the nose, and throat
Infection Comments First Line Agents Penicillin Allergy History of multiresistant. line treatment: persist for >7 days they may be
 Gastrointestinal Infections Infection Comments First Line Agents Penicillin Allergy History of multiresistant Campylobacter Antibiotics not recommended. Erythromycin 250mg PO 6 Alternative to first N/A
Gastrointestinal Infections Infection Comments First Line Agents Penicillin Allergy History of multiresistant Campylobacter Antibiotics not recommended. Erythromycin 250mg PO 6 Alternative to first N/A
GUIDELINES FOR THE MANAGEMENT OF COMMUNITY-ACQUIRED PNEUMONIA IN ADULTS
 Version 3.1 GUIDELINES FOR THE MANAGEMENT OF COMMUNITY-ACQUIRED PNEUMONIA IN ADULTS Date ratified June 2008 Updated March 2009 Review date June 2010 Ratified by Authors Consultation Evidence base Changes
Version 3.1 GUIDELINES FOR THE MANAGEMENT OF COMMUNITY-ACQUIRED PNEUMONIA IN ADULTS Date ratified June 2008 Updated March 2009 Review date June 2010 Ratified by Authors Consultation Evidence base Changes
Source: Portland State University Population Research Center (
 Methicillin Resistant Staphylococcus aureus (MRSA) Surveillance Report 2010 Oregon Active Bacterial Core Surveillance (ABCs) Office of Disease Prevention & Epidemiology Oregon Health Authority Updated:
Methicillin Resistant Staphylococcus aureus (MRSA) Surveillance Report 2010 Oregon Active Bacterial Core Surveillance (ABCs) Office of Disease Prevention & Epidemiology Oregon Health Authority Updated:
Guidelines for the Initiation of Empirical Antibiotic therapy in Respiratory Disease (Adults)
 Guidelines for the Initiation of Empirical Antibiotic therapy in Respiratory Disease (Adults) Community Acquired Pneumonia Community Acquired Pneumonia 1) Is it pneumonia? ie new symptoms and signs of
Guidelines for the Initiation of Empirical Antibiotic therapy in Respiratory Disease (Adults) Community Acquired Pneumonia Community Acquired Pneumonia 1) Is it pneumonia? ie new symptoms and signs of
Pneumonia considerations Galia Rahav Infectious diseases unit Sheba medical center
 Pneumonia considerations 2017 Galia Rahav Infectious diseases unit Sheba medical center Sir William Osler (1849 1919) "Father of modern medicine Pneumonia: The old man's friend The captain of the men of
Pneumonia considerations 2017 Galia Rahav Infectious diseases unit Sheba medical center Sir William Osler (1849 1919) "Father of modern medicine Pneumonia: The old man's friend The captain of the men of
March 2017 Animal Care Conference, Sacramento, CA
 Rabies Bats and Parvo Clusters: Helping the Community through Animal Disease Surveillance Emily Beeler DVM MPH, Veterinary Public Health Program, Los Angeles County Department of Public Health ebeeler@ph.lacounty.gov
Rabies Bats and Parvo Clusters: Helping the Community through Animal Disease Surveillance Emily Beeler DVM MPH, Veterinary Public Health Program, Los Angeles County Department of Public Health ebeeler@ph.lacounty.gov
Brucellosis in Kyrgyzstan
 Centers for Disease Control and Prevention Case Studies in Applied Epidemiology No. 053-D11 Brucellosis in Kyrgyzstan Participant's Guide Learning Objectives After completing this case study, the participant
Centers for Disease Control and Prevention Case Studies in Applied Epidemiology No. 053-D11 Brucellosis in Kyrgyzstan Participant's Guide Learning Objectives After completing this case study, the participant
Volume. December Infection. Notes. length of. cases as 90% 1 week. tonsillitis. First Line. sore throat / daily for 5 days. quinsy >4000.
 Volume 8; Number 22 LINCOLNSHIRE GUIDELINES FOR THE TREATMENT OF COMMONLYY OCCURRING INFECTIONS IN PRIMARY CARE: WINTER 2014/15 In this issue of the PACE Bulletin we present an update of our Guidelines
Volume 8; Number 22 LINCOLNSHIRE GUIDELINES FOR THE TREATMENT OF COMMONLYY OCCURRING INFECTIONS IN PRIMARY CARE: WINTER 2014/15 In this issue of the PACE Bulletin we present an update of our Guidelines
Disclosures. Consider This Case. Objectives. Consequences of Bites. Animal Bites: What to Do and What to Avoid. Animal Bites: Epidemiology
 Animal Bites: What to Do and What to Avoid Meg Fisher, MD Medical Director Disclosures I have no disclosures I do not plan to discuss off label uses of drugs Objectives Manage a child who is bitten Discuss
Animal Bites: What to Do and What to Avoid Meg Fisher, MD Medical Director Disclosures I have no disclosures I do not plan to discuss off label uses of drugs Objectives Manage a child who is bitten Discuss
The Law of Unintended Consequences
 The Law of Unintended Consequences Tests I Wish You d Never Ordered (Choosing Wisely ) G Blackburn DO, MACOI Moderator, (Infectious Disease) Annual WVOMA CME Conference November, 2017 White Sulphur Springs,
The Law of Unintended Consequences Tests I Wish You d Never Ordered (Choosing Wisely ) G Blackburn DO, MACOI Moderator, (Infectious Disease) Annual WVOMA CME Conference November, 2017 White Sulphur Springs,
TOC INDEX. Salmonellosis in Feedlot Cattle. Jane Pritchard. Take Home Message. Introduction
 TOC INDEX Salmonellosis in Feedlot Cattle Jane Pritchard Take Home Message Salmonellosis in feedlot cattle is an important but uncommon disease. The disease has been recognized only recently as a significant
TOC INDEX Salmonellosis in Feedlot Cattle Jane Pritchard Take Home Message Salmonellosis in feedlot cattle is an important but uncommon disease. The disease has been recognized only recently as a significant
Questions and Answers about Rabies
 Pets 1. Q: How can I protect my pet from rabies? A: There are several things you can do to protect your pet from rabies. First, visit your veterinarian with your pet on a regular basis and keep rabies
Pets 1. Q: How can I protect my pet from rabies? A: There are several things you can do to protect your pet from rabies. First, visit your veterinarian with your pet on a regular basis and keep rabies
Accidental Exposure to Cattle Brucellosis Vaccines in Wyoming, Montana, and Idaho Veterinarians
 Accidental Exposure to Cattle Brucellosis Vaccines in Wyoming, Montana, and Idaho Veterinarians Kerry Pride, DVM, MPH, DACVPM Brucellosis Meeting April 3, 2013 Veterinary Occupational Exposure 1 needle
Accidental Exposure to Cattle Brucellosis Vaccines in Wyoming, Montana, and Idaho Veterinarians Kerry Pride, DVM, MPH, DACVPM Brucellosis Meeting April 3, 2013 Veterinary Occupational Exposure 1 needle
Does history-taking help predict rabies diagnosis in dogs?
 Asian Biomedicine Vol. 4 No. 5 October 2010; 811-815 Brief communication (original) Does history-taking help predict rabies diagnosis in dogs? Veera Tepsumethanon, Boonlert Lumlertdacha, Channarong Mitmoonpitak
Asian Biomedicine Vol. 4 No. 5 October 2010; 811-815 Brief communication (original) Does history-taking help predict rabies diagnosis in dogs? Veera Tepsumethanon, Boonlert Lumlertdacha, Channarong Mitmoonpitak
Guideline for Prevention of Brucellosis in Meat Packing Plant Workers
 Guideline for Prevention of Brucellosis in Meat Packing Plant Workers Introduction Brucellosis is a disease which may spread from animals to man. There is no evidence for person to person transmission.
Guideline for Prevention of Brucellosis in Meat Packing Plant Workers Introduction Brucellosis is a disease which may spread from animals to man. There is no evidence for person to person transmission.
Preventing Multi-Drug Resistant Organism (MDRO) Infections. For National Patient Safety Goal
 Preventing Multi-Drug Resistant Organism (MDRO) Infections For National Patient Safety Goal 07.03.01 2009 Methicillin Resistant Staphlococcus aureus (MRSA) About 3-8% of the population at large is a carrier
Preventing Multi-Drug Resistant Organism (MDRO) Infections For National Patient Safety Goal 07.03.01 2009 Methicillin Resistant Staphlococcus aureus (MRSA) About 3-8% of the population at large is a carrier
Case 2 Synergy satellite event: Good morning pharmacists! Case studies on antimicrobial resistance
 Case 2 Synergy satellite event: Good morning pharmacists! Case studies on antimicrobial resistance 22nd Congress of the EAHP "Hospital pharmacists catalysts for change", 22-24 March 2017, Cannes Disclosure
Case 2 Synergy satellite event: Good morning pharmacists! Case studies on antimicrobial resistance 22nd Congress of the EAHP "Hospital pharmacists catalysts for change", 22-24 March 2017, Cannes Disclosure
GP Small Group education April/May 2015 Antibiotics Resistance is futile
 GP Small Group education April/May 2015 Antibiotics Resistance is futile Acknowledgements This material was prepared by the Clinical Quality and Education team with help gratefully received from: Topic
GP Small Group education April/May 2015 Antibiotics Resistance is futile Acknowledgements This material was prepared by the Clinical Quality and Education team with help gratefully received from: Topic
Safety of an Out-Patient Intravenous Antibiotics Programme
 Safety of an Out-Patient Intravenous Antibiotics Programme Chan VL, Tang ESK, Leung WS, Wong L, Cheung PS, Chu CM Department of Medicine & Geriatrics United Christian Hospital Outpatient Parental Antimicrobial
Safety of an Out-Patient Intravenous Antibiotics Programme Chan VL, Tang ESK, Leung WS, Wong L, Cheung PS, Chu CM Department of Medicine & Geriatrics United Christian Hospital Outpatient Parental Antimicrobial
Community-Associated C. difficile Infection: Think Outside the Hospital. Maria Bye, MPH Epidemiologist May 1, 2018
 Community-Associated C. difficile Infection: Think Outside the Hospital Maria Bye, MPH Epidemiologist Maria.Bye@state.mn.us 651-201-4085 May 1, 2018 Clostridium difficile Clostridium difficile Clostridium
Community-Associated C. difficile Infection: Think Outside the Hospital Maria Bye, MPH Epidemiologist Maria.Bye@state.mn.us 651-201-4085 May 1, 2018 Clostridium difficile Clostridium difficile Clostridium
Appropriate Management of Common Pediatric Infections. Blaise L. Congeni M.D. Akron Children s Hospital Division of Pediatric Infectious Diseases
 Appropriate Management of Common Pediatric Infections Blaise L. Congeni M.D. Akron Children s Hospital Division of Pediatric Infectious Diseases It s all about the microorganism The common pathogens Viruses
Appropriate Management of Common Pediatric Infections Blaise L. Congeni M.D. Akron Children s Hospital Division of Pediatric Infectious Diseases It s all about the microorganism The common pathogens Viruses
Above: life cycle of toxoplasma gondii. Below: transmission of this infection.
 Toxoplasmosis PDF This article is based on a paid for research paper dated 1972 of similar title and authored by J.K.Frenkel and J.P. Dubey. It was published by The Journal of Infectious Diseases Vol.
Toxoplasmosis PDF This article is based on a paid for research paper dated 1972 of similar title and authored by J.K.Frenkel and J.P. Dubey. It was published by The Journal of Infectious Diseases Vol.
Prescribing Guidelines for Outpatient Antimicrobials in Otherwise Healthy Children
 Prescribing Guidelines for Outpatient Antimicrobials in Otherwise Healthy Children Prescribing Antimicrobials for Common Illnesses When treating common illnesses such as ear infections and strep throat,
Prescribing Guidelines for Outpatient Antimicrobials in Otherwise Healthy Children Prescribing Antimicrobials for Common Illnesses When treating common illnesses such as ear infections and strep throat,
Blood protozoan: Plasmodium
 Blood protozoan: Plasmodium Dr. Hala Al Daghistani The causative agent of including Plasmodium vivax P. falciparum P. malariae P. ovale. malaria in humans: four species are associated The Plasmodium spp.
Blood protozoan: Plasmodium Dr. Hala Al Daghistani The causative agent of including Plasmodium vivax P. falciparum P. malariae P. ovale. malaria in humans: four species are associated The Plasmodium spp.
Treatment of septic peritonitis
 Vet Times The website for the veterinary profession https://www.vettimes.co.uk Treatment of septic peritonitis Author : Andrew Linklater Categories : Companion animal, Vets Date : November 2, 2016 Septic
Vet Times The website for the veterinary profession https://www.vettimes.co.uk Treatment of septic peritonitis Author : Andrew Linklater Categories : Companion animal, Vets Date : November 2, 2016 Septic
مادة االدوية المرحلة الثالثة م. غدير حاتم محمد
 م. مادة االدوية المرحلة الثالثة م. غدير حاتم محمد 2017-2016 ANTIMICROBIAL DRUGS Antimicrobial drugs Lecture 1 Antimicrobial Drugs Chemotherapy: The use of drugs to treat a disease. Antimicrobial drugs:
م. مادة االدوية المرحلة الثالثة م. غدير حاتم محمد 2017-2016 ANTIMICROBIAL DRUGS Antimicrobial drugs Lecture 1 Antimicrobial Drugs Chemotherapy: The use of drugs to treat a disease. Antimicrobial drugs:
Australian and New Zealand College of Veterinary Scientists. Membership Examination. Veterinary Emergency and Critical Care Paper 1
 Australian and New Zealand College of Veterinary Scientists Membership Examination June 2014 Veterinary Emergency and Critical Care Paper 1 Perusal time: Fifteen (15) minutes Time allowed: Two (2) hours
Australian and New Zealand College of Veterinary Scientists Membership Examination June 2014 Veterinary Emergency and Critical Care Paper 1 Perusal time: Fifteen (15) minutes Time allowed: Two (2) hours
Clostridium difficile Colitis
 Update on Clostridium difficile Colitis Fredrick M. Abrahamian, D.O., FACEP Associate Professor of Medicine UCLA School of Medicine Director of Education Department of Emergency Medicine Olive View-UCLA
Update on Clostridium difficile Colitis Fredrick M. Abrahamian, D.O., FACEP Associate Professor of Medicine UCLA School of Medicine Director of Education Department of Emergency Medicine Olive View-UCLA
Surveillance of animal brucellosis
 Surveillance of animal brucellosis Assoc.Prof.Dr. Theera Rukkwamsuk Department of large Animal and Wildlife Clinical Science Faculty of Veterinary Medicine Kasetsart University Review of the epidemiology
Surveillance of animal brucellosis Assoc.Prof.Dr. Theera Rukkwamsuk Department of large Animal and Wildlife Clinical Science Faculty of Veterinary Medicine Kasetsart University Review of the epidemiology
Antibiotic Stewardship in the Neonatal Intensive Care Unit. Objectives. Background 4/20/2017. Natasha Nakra, MD April 28, 2017
 Antibiotic Stewardship in the Neonatal Intensive Care Unit Natasha Nakra, MD April 28, 2017 Objectives 1. Describe antibiotic use in the NICU 2. Explain the role of antibiotic stewardship in the NICU 3.
Antibiotic Stewardship in the Neonatal Intensive Care Unit Natasha Nakra, MD April 28, 2017 Objectives 1. Describe antibiotic use in the NICU 2. Explain the role of antibiotic stewardship in the NICU 3.
Antibiotic stewardship in long term care
 Antibiotic stewardship in long term care Shira Doron, MD Associate Professor of Medicine Division of Geographic Medicine and Infectious Diseases Tufts Medical Center Boston, MA Consultant to Massachusetts
Antibiotic stewardship in long term care Shira Doron, MD Associate Professor of Medicine Division of Geographic Medicine and Infectious Diseases Tufts Medical Center Boston, MA Consultant to Massachusetts
Let me clear my throat: empiric antibiotics in
 Let me clear my throat: empiric antibiotics in respiratory tract infections Alexander John Langley, MD MS MPH Goals of this talk Overuse of antibiotics is a major issue, as a result many specialist medical
Let me clear my throat: empiric antibiotics in respiratory tract infections Alexander John Langley, MD MS MPH Goals of this talk Overuse of antibiotics is a major issue, as a result many specialist medical
Biological Threat Fact Sheets
 Biological Threat Fact Sheets Anthrax Agent: Bacillus anthracis There are three clinical forms of B. anthracis which are determined by route of entry: Pulmonary or Inhalation BT implications Cutaneous
Biological Threat Fact Sheets Anthrax Agent: Bacillus anthracis There are three clinical forms of B. anthracis which are determined by route of entry: Pulmonary or Inhalation BT implications Cutaneous
Guidelines on prescribing antibiotics. For physicians and others in Denmark
 Guidelines on prescribing antibiotics 2013 For physicians and others in Denmark Guidelines on prescribing antibiotics For physicians and others in Denmark 2013 by the Danish Health and Medicines Authority.
Guidelines on prescribing antibiotics 2013 For physicians and others in Denmark Guidelines on prescribing antibiotics For physicians and others in Denmark 2013 by the Danish Health and Medicines Authority.
11-ID-10. Committee: Infectious Disease. Title: Creation of a National Campylobacteriosis Case Definition
 11-ID-10 Committee: Infectious Disease Title: Creation of a National Campylobacteriosis Case Definition I. Statement of the Problem Although campylobacteriosis is not nationally-notifiable, it is a disease
11-ID-10 Committee: Infectious Disease Title: Creation of a National Campylobacteriosis Case Definition I. Statement of the Problem Although campylobacteriosis is not nationally-notifiable, it is a disease
High Risk Behavior for Wild Sheep: Contact with Domestic Sheep and Goats
 High Risk Behavior for Wild Sheep: Contact with Domestic Sheep and Goats Introduction The impact of disease on wild sheep populations was brought to the forefront in the winter of 2009-10 due to all age
High Risk Behavior for Wild Sheep: Contact with Domestic Sheep and Goats Introduction The impact of disease on wild sheep populations was brought to the forefront in the winter of 2009-10 due to all age
Antimicrobial Susceptibility Patterns of Salmonella Typhi From Kigali,
 In the name of God Shiraz E-Medical Journal Vol. 11, No. 3, July 2010 http://semj.sums.ac.ir/vol11/jul2010/88030.htm Antimicrobial Susceptibility Patterns of Salmonella Typhi From Kigali, Rwanda. Ashok
In the name of God Shiraz E-Medical Journal Vol. 11, No. 3, July 2010 http://semj.sums.ac.ir/vol11/jul2010/88030.htm Antimicrobial Susceptibility Patterns of Salmonella Typhi From Kigali, Rwanda. Ashok
Blood protozoan: Plasmodium
 Blood protozoan: Plasmodium The causative agent of including Plasmodium vivax P. falciparum P. malariae P. ovale. malaria in humans:four species are associated The Plasmodium spp. life cycle can be divided
Blood protozoan: Plasmodium The causative agent of including Plasmodium vivax P. falciparum P. malariae P. ovale. malaria in humans:four species are associated The Plasmodium spp. life cycle can be divided
Healthcare-associated Infections and Antimicrobial Use Prevalence Survey
 Healthcare-associated Infections and Antimicrobial Use Prevalence Survey Shamima Sharmin, M.B.B.S., MSc, MPH Emerging Infections Program New Mexico Department of Health Agenda Recognize healthcare-associated
Healthcare-associated Infections and Antimicrobial Use Prevalence Survey Shamima Sharmin, M.B.B.S., MSc, MPH Emerging Infections Program New Mexico Department of Health Agenda Recognize healthcare-associated
Care and Handling of Pets
 Communicable Disease Outreach Program 3020 Rucker Avenue, Suite 300 Everett, WA 98201-3900 425.339.5278 Care and Handling of Pets Name of facility: WIWS Pet restrictions 1. Pets will be inaccessible to
Communicable Disease Outreach Program 3020 Rucker Avenue, Suite 300 Everett, WA 98201-3900 425.339.5278 Care and Handling of Pets Name of facility: WIWS Pet restrictions 1. Pets will be inaccessible to
Overview of Infection Control and Prevention
 Overview of Infection Control and Prevention Review of the Cesarean-section Antibiotic Prophylaxis Program in Jordan and Workshop on Rational Medicine Use and Infection Control Terry Green and Salah Gammouh
Overview of Infection Control and Prevention Review of the Cesarean-section Antibiotic Prophylaxis Program in Jordan and Workshop on Rational Medicine Use and Infection Control Terry Green and Salah Gammouh
Pneumonia Antibiotic Guidance for Adults PAGL Inclusion Approved at January 2017 PGC
 Pneumonia Antibiotic Guidance for Adults PAGL Inclusion Approved at January 2017 PGC APPROVED BY: Policy and Guidelines Committee TRUST REFERENCE: B9/2009 AWP Ref: AWP61 Date (approved): July 2008 REVIEW
Pneumonia Antibiotic Guidance for Adults PAGL Inclusion Approved at January 2017 PGC APPROVED BY: Policy and Guidelines Committee TRUST REFERENCE: B9/2009 AWP Ref: AWP61 Date (approved): July 2008 REVIEW
Acute Pyelonephritis POAC Guideline
 Acute Pyelonephritis POAC Guideline Refer full regional pathway http://aucklandregion.healthpathways.org.nz/33444 EXCLUSION CRITERIA: COMPLICATED PYELONEPHRITIS Discuss with relevant specialist for advice
Acute Pyelonephritis POAC Guideline Refer full regional pathway http://aucklandregion.healthpathways.org.nz/33444 EXCLUSION CRITERIA: COMPLICATED PYELONEPHRITIS Discuss with relevant specialist for advice
Hendra virus: Important information for all horse owners. An update on Hendra virus The Hendra vaccine
 Hendra virus: Important information for all horse owners An update on Hendra virus The Hendra vaccine HENDRA VIRUS Welcome to the Hendra virus information update The aim of this update is to provide information
Hendra virus: Important information for all horse owners An update on Hendra virus The Hendra vaccine HENDRA VIRUS Welcome to the Hendra virus information update The aim of this update is to provide information
PIGEON FEVER (Corynebacterium pseudotuberculosis Infection)
 PIGEON FEVER (Corynebacterium pseudotuberculosis Infection) Definition Clinical Signs Incubation Period Risk Factors Transmission Diagnostic Sampling, Testing and Handling Environmental Persistence Specific
PIGEON FEVER (Corynebacterium pseudotuberculosis Infection) Definition Clinical Signs Incubation Period Risk Factors Transmission Diagnostic Sampling, Testing and Handling Environmental Persistence Specific
Perichondritis: Source: UpToDate Ciprofloxacin 10 mg/kg/dose PO (max 500 mg/dose) BID Inpatient: Ceftazidime 50 mg/kg/dose q8 hours IV
 Empiric Antibiotics for Pediatric Infections Seen in ED NOTE: Choice of empiric antibiotic therapy must take into account local pathogen frequency and resistance patterns, individual patient characteristics,
Empiric Antibiotics for Pediatric Infections Seen in ED NOTE: Choice of empiric antibiotic therapy must take into account local pathogen frequency and resistance patterns, individual patient characteristics,
Zoonotic Diseases. Risks of working with wildlife. Maria Baron Palamar, Wildlife Veterinarian
 Zoonotic Diseases Risks of working with wildlife www.cdc.gov Definition Zoonoses: infectious diseases of vertebrate animals that can be naturally transmitted to humans Health vs. Disease Transmission -
Zoonotic Diseases Risks of working with wildlife www.cdc.gov Definition Zoonoses: infectious diseases of vertebrate animals that can be naturally transmitted to humans Health vs. Disease Transmission -
Fungal Disease. What is a fungus?
 Fungal Disease What is a fungus? A fungus is a living organism. It goes through a complicated life cycle and is able to spread in the environment by producing large numbers of spores that are easily dispersed
Fungal Disease What is a fungus? A fungus is a living organism. It goes through a complicated life cycle and is able to spread in the environment by producing large numbers of spores that are easily dispersed
Infection Control and Standard Precautions
 Home Care Aide Training Guide Infection Control and Standard Precautions Pre-Service Training Course #1 Home Care Aide Orientation Training Manual: Infection Control & Standard Precautions Page 2 Table
Home Care Aide Training Guide Infection Control and Standard Precautions Pre-Service Training Course #1 Home Care Aide Orientation Training Manual: Infection Control & Standard Precautions Page 2 Table
Detection and Quantitation of the Etiologic Agents of Ventilator Associated Pneumonia in Endotracheal Tube Aspirates From Patients in Iran
 Letter to the Editor Detection and Quantitation of the Etiologic Agents of Ventilator Associated Pneumonia in Endotracheal Tube Aspirates From Patients in Iran Mohammad Rahbar, PhD; Massoud Hajia, PhD
Letter to the Editor Detection and Quantitation of the Etiologic Agents of Ventilator Associated Pneumonia in Endotracheal Tube Aspirates From Patients in Iran Mohammad Rahbar, PhD; Massoud Hajia, PhD
Humane Society of West Michigan
 Humane Society of West Michigan Health Concerns & Medical Treatment Feline Upper Respiratory Infections Your cat may have a cold when you get him home. Cats are subject to airborne virus disease that is
Humane Society of West Michigan Health Concerns & Medical Treatment Feline Upper Respiratory Infections Your cat may have a cold when you get him home. Cats are subject to airborne virus disease that is
NUMBER: /2005
 Purpose PAGE 1 OF 7 The purpose of this policy is to describe the procedures for keeping and maintaining animal medical records. This procedure is approved by the Creighton University Institutional Animal
Purpose PAGE 1 OF 7 The purpose of this policy is to describe the procedures for keeping and maintaining animal medical records. This procedure is approved by the Creighton University Institutional Animal
Canine Anaplasmosis Anaplasma phagocytophilum Anaplasma platys
 Canine Anaplasmosis Anaplasma phagocytophilum Anaplasma platys It takes just hours for an infected tick to transmit Anaplasma organisms to a dog. What is canine anaplasmosis? Canine anaplasmosis is a disease
Canine Anaplasmosis Anaplasma phagocytophilum Anaplasma platys It takes just hours for an infected tick to transmit Anaplasma organisms to a dog. What is canine anaplasmosis? Canine anaplasmosis is a disease
ANTIMICROBIAL STEWARDSHIP: THE ROLE OF THE CLINICIAN SAM GUREVITZ PHARM D, CGP BUTLER UNIVERSITY COLLEGE OF PHARMACY AND HEALTH SCIENCES
 ANTIMICROBIAL STEWARDSHIP: THE ROLE OF THE CLINICIAN SAM GUREVITZ PHARM D, CGP BUTLER UNIVERSITY COLLEGE OF PHARMACY AND HEALTH SCIENCES 1 Crisis: Antibiotic Resistance Success Strategy 2 OBJECTIVES Discuss
ANTIMICROBIAL STEWARDSHIP: THE ROLE OF THE CLINICIAN SAM GUREVITZ PHARM D, CGP BUTLER UNIVERSITY COLLEGE OF PHARMACY AND HEALTH SCIENCES 1 Crisis: Antibiotic Resistance Success Strategy 2 OBJECTIVES Discuss
NUMBER: R&C-ARF-10.0
 1. PURPOSE PAGE 1 OF 6 This policy describes the procedures for keeping and maintaining animal medical records. This procedure is approved by the Creighton University Institutional Animal Care and Use
1. PURPOSE PAGE 1 OF 6 This policy describes the procedures for keeping and maintaining animal medical records. This procedure is approved by the Creighton University Institutional Animal Care and Use
IACUC Policy on Humane Endpoints in Animal Use Proposals
 IACUC Policy on Humane Endpoints in Animal Use Proposals Definitions: moribund \MOR-uh-bund\, adjective: In a dying state; dying; at the point of death. morbid\ MOR-bid\, adjective: pertaining to, affected
IACUC Policy on Humane Endpoints in Animal Use Proposals Definitions: moribund \MOR-uh-bund\, adjective: In a dying state; dying; at the point of death. morbid\ MOR-bid\, adjective: pertaining to, affected
Rational management of community acquired infections
 Rational management of community acquired infections Dr Tanu Singhal MD, MSc Consultant Pediatrics and Infectious Disease Kokilaben Dhirubhai Ambani Hospital, Mumbai Why is rational management needed?
Rational management of community acquired infections Dr Tanu Singhal MD, MSc Consultant Pediatrics and Infectious Disease Kokilaben Dhirubhai Ambani Hospital, Mumbai Why is rational management needed?
Pre- and Post -Surgery Information
 Pre- and Post -Surgery Information Preparing For Anesthetic Procedures or Surgery Preparing your pet: If you notice your pet is coughing or sneezing, vomiting, or has diarrhea, please call to speak with
Pre- and Post -Surgery Information Preparing For Anesthetic Procedures or Surgery Preparing your pet: If you notice your pet is coughing or sneezing, vomiting, or has diarrhea, please call to speak with
Capnocytophaga canimorsus
 Capnocytophaga canimorsus infection caused by dog/cat-bites/scratches Michio SUZUKI C. canimorsus Capnocytophaga canimorsus 200 C. canimorsus 30 C. canimorsus Capnocytophaga canimorsus 30 200 2 90 470
Capnocytophaga canimorsus infection caused by dog/cat-bites/scratches Michio SUZUKI C. canimorsus Capnocytophaga canimorsus 200 C. canimorsus 30 C. canimorsus Capnocytophaga canimorsus 30 200 2 90 470
TREAT Steward. Antimicrobial Stewardship software with personalized decision support
 TREAT Steward TM Antimicrobial Stewardship software with personalized decision support ANTIMICROBIAL STEWARDSHIP - Interdisciplinary actions to improve patient care Quality Assurance The aim of antimicrobial
TREAT Steward TM Antimicrobial Stewardship software with personalized decision support ANTIMICROBIAL STEWARDSHIP - Interdisciplinary actions to improve patient care Quality Assurance The aim of antimicrobial
Introduction- Rickettsia felis
 Cat flea-borne spotted fever in humans is the dog to blame? Rebecca J Traub Assoc. Prof. in Parasitology Faculty of Veterinary and Agricultural Sciences Introduction- Rickettsia felis Emerging zoonoses
Cat flea-borne spotted fever in humans is the dog to blame? Rebecca J Traub Assoc. Prof. in Parasitology Faculty of Veterinary and Agricultural Sciences Introduction- Rickettsia felis Emerging zoonoses
Methicillin-Resistant Staphylococcus aureus
 Methicillin-Resistant Staphylococcus aureus By Karla Givens Means of Transmission and Usual Reservoirs Staphylococcus aureus is part of normal flora and can be found on the skin and in the noses of one
Methicillin-Resistant Staphylococcus aureus By Karla Givens Means of Transmission and Usual Reservoirs Staphylococcus aureus is part of normal flora and can be found on the skin and in the noses of one
Septicaemia Definitions 1
 Septicaemia Definitions 1 Term Definition Bacteraemia Systemic Inflammatory response (SIRS) Sepsis Bacteria that can be cultured from the blood stream The systemic response to a wide range of stresses.
Septicaemia Definitions 1 Term Definition Bacteraemia Systemic Inflammatory response (SIRS) Sepsis Bacteria that can be cultured from the blood stream The systemic response to a wide range of stresses.
THE COMPLEXITIES AT THE INTERFACE AMONG DOMESTIC/WILD RODENTS, FLEAS, PETS, AND MAN IN URBAN PLAGUE ECOLOGY IN LOS ANGELES, COUNTY, CALIFORNIA
 University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Proceedings of the Twelfth Vertebrate Pest Conference (1986) Vertebrate Pest Conference Proceedings collection March 1986
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Proceedings of the Twelfth Vertebrate Pest Conference (1986) Vertebrate Pest Conference Proceedings collection March 1986
LEPTOSPIROSIS. Understanding the risk to your dog
 LEPTOSPIROSIS Understanding the risk to your dog What is leptospirosis? Leptospirosis is an infectious disease that causes serious illness in dogs, other animals, and people throughout the US and around
LEPTOSPIROSIS Understanding the risk to your dog What is leptospirosis? Leptospirosis is an infectious disease that causes serious illness in dogs, other animals, and people throughout the US and around
