Multidrug-resistant enterococci in the hospital environment: detection of novel vancomycin-resistant E. faecium clone ST910
|
|
|
- Mildred Barnett
- 5 years ago
- Views:
Transcription
1 Original Article Multidrug-resistant enterococci in the hospital environment: detection of novel vancomycin-resistant E. faecium clone ST910 Raoudha Dziri 1, Carmen Lozano 2, Leila Ben Said 1, Ridha Bellaaj 3, Abdellatif Boudabous 1, Karim Ben Slama 1,4, Carmen Torres 2, Naouel Klibi 1 1 Laboratory of Microorganisms and Active Biomolecules, Faculty of Sciences of Tunis, University of Tunis El Manar, Tunis, Tunisia 2 Area of Biochemistry and Molecular Biology, University of La Rioja, Logroño, Spain 3 Hospital Hygiene and Environmental Protection Service, Military Hospital of Tunis HMPIT Tunis, Tunisia 4 High Institute of Applied Biological Sciences of Tunis, University of Tunis El Manar, Tunis, Tunisia Abstract Introduction: The role of the hospital environment as a reservoir of resistant bacteria in Tunisia has been poorly investigated; however, it could be responsible for the transmission of multidrug-resistant bacteria. The objective was to study the prevalence of Enterococcus in the environment of a Tunisian hospital and the antibiotic resistance phenotype/genotype in recovered isolates, with special reference to vancomycin resistance. Methodology: A total of 300 samples were taken (March June, 2013) and inoculated in Slanetz-Bartley agar plates supplemented or not supplemented with 8 µg/ml of vancomycin. Antibiotic resistance genes were tested by polymerase chain reaction (PCR). The clonal relatedness of the vana isolates was assessed using pulsed-field gel electrophoresis (PFGE) and multilocus sequence testing (MLST). Results: Enterococci were recovered in 33.3% of tested samples inoculated in SB medium. E faecium was the most prevalent species, followed by E. faecalis and E. casseliflavus. Antimicrobial resistance genes detected were as follows (number of isolates): erm(b) (71), tet(m) (18), aph(3 )-IIIa (27), ant(6)-ia (15), cat(a) (4), and van(c2) (6). Vancomycin-resistant-enterococci (VRE) were recovered from 14 samples (4.7%), when tested in SB-VAN. The 14 VRE (one per positive sample) were identified as E. faecium and contained the van(a),erm(b), tet(m), ant(6)- Ia, and aph(3 )-IIIa genes. Thirteen of the VRE strains were ascribed by PFGE and MLST to a novel clone (new ST910), and only one VRE strain was typed as ST80 included in CC17. Conclusions: The emergence and spread of new clones of VRE, especially in the hospital environment in this country, could become particularly problematic. Key words: Enterococcus; hospital environment; VRE; ST910; cross-contamination; Tunisia. J Infect Dev Ctries 2016; 10(8): doi: /jidc.8014 (Received 16 December 2015 Accepted 27 March 2016) Copyright 2016 Dziri et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Introduction Enterococci are recognized now as important causes of human nosocomial infections, especially in immunocompromised patients. These microorganisms are able to cause multiple pathologies such as urinary tract infections, bacteremia, and endocarditis, among others [1]. Furthermore, the capacity of enterococci to survive outside the human body for prolonged periods of time contributes to cross-contamination through the hands of healthcare workers, leading to outbreaks [1,2]. The hospital environment can be a reservoir of resistant bacteria, and the role of a possible dissemination of these bacteria, especially vancomycin-resistant enterococci (VRE) via inanimate surfaces and equipment, has been documented by several researchers [3-6]. Moreover, the cross-transmission, between patients via contaminated hands and surfaces, of multidrug-resistant microorganisms increases length of stays in hospitals, healthcare costs, and mortality [7,8]. Over the past 20 years, epidemiological data collected have shown the emergence of enterococci, especially E. faecium, as pathogens responsible for nosocomial infections. The expansion of E. faecium isolates belonging to clonal complex (CC17) is remarkable [9]. This epidemic clonal complex is characterized by ampicillin and vancomycin resistance, as well as by the presence of virulence determinants such as hyl Efm (encoding a hyaluronidase-like protein) and a putative pathogenicity island that includes esp Efm (enterococcal surface protein in E. faecium), and is responsible for severe nosocomial infections and hospital outbreaks [10,11]. The resistance to
2 vancomycin in enterococci is still uncommon in Tunisia, and there are only a few previous reports of detection of vana-containing enterococci in hospitals in that country [6,12].In a previous study performed by our group in the Military Hospital of Tunis in 2012 [6], the presence of vana-containing E. faecium isolates was detected in clinical samples as well as in 5% of environmental samples of this hospital. The objective of the present work was to continue the epidemiological study on environmental samples in the hospital during 2013, to determine the prevalence of Enterococcus and the phenotypes and genotypes of antibiotic resistance in recovered isolates, with special reference to vancomycin resistance, as well as to analyze the evolution of environmental VRE in terms of frequency and type of detected clones. Methodology Samples and bacterial isolates A total of 300 environmental samples were obtained from 17 different services of the Military Hospital of Tunis, during March June The samples were taken with sterile cotton swabs from inanimate surfaces (beds, treatment tables, toilets, faucets, wrists, sinks) after routine cleaning (n = 250), and from the hands of patients (n = 23) and staff (n = 27). Informed consent was obtained from all individuals who participated in the study. Samples were pre-enriched in brain-heart infusion broth for 24 hours at 37 C. Next, 0.1 ml of the suspension was seeded in plates of Slanetz-Bartley (Biolife, Milano, Italy) not supplemented (SB) and supplemented with 8 μg/ml of vancomycin (BioRad, Marnes-la-Coquette, France) (SB-VAN). The samples were then incubated for 48 hours at 37ºC for the recovery of enterococci and VRE. A colony was selected of each positive sample and was identified by species-specific polymerase chain reactions (PCRs) [13]. Antimicrobial susceptibility testing and detection of resistance genes Antibiotic susceptibility was tested for 12 antimicrobial agents by the disk-diffusion method. The agents tested were the following (μg/disk): vancomycin (30), teicoplanin (30), tetracycline (30), chloramphenicol (30), erythromycin (15), trimethoprim-sulfamethoxazole (1.25/23.75), ciprofloxacin (5) and ampicillin (10) (BioRad, Marnesla-Coquette, France) [14]. Detection of pristinamycin resistance (15) and high-level resistance to gentamicin (500), kanamycin (1,000), and streptomycin (500) was performed according to the Antibiogram Committee of the French Society for Microbiology [15]. Minimum inhibitory concentrations (MICs) for vancomycin and teicoplanin were determined using the agar dilution method [14]. Resistance genes were sought by PCR in enterococcal isolates that showed resistance to glycopeptides (van(a), van(b), van(c1), and van(c2)) [16], tetracycline [tet(m), tet(l)], macrolides [erm(a), erm(b), erm(c)], and aminoglycosides [aph(3 )-IIIa, aac(6 )-aph(2 ), ant(6)-ia] using primers and conditions previously reported [13]. The presence of the insertion element IS16 (usually found in hospital-associated E. faecium isolates implicated in outbreaks) was performed by PCR in all the vana-containing isolates [17]. Detection of virulence genes The presence of esp (enterococcal surface protein) and hyl (encoding a hyaluronidase-like protein) virulence genes was detected by PCR in the vanacontaining E. faecium isolates, and the esp gene was also tested in the vancomycin-susceptible enterococci [18]. Pulsed-field gel electrophoresis (PFGE) The clonal relatedness of the vana-containing isolates was assessed using PFGE of the genomic DNA digested with SmaI enzyme [13]. Interpretation of banding patterns was performed according to the criteria specified by Tenover et al. [19]. The dendrogram of PFGE patterns of SmaI digested DNA of 14 VRE was prepared by FPQuest (BioRad, Hercules, USA) software using UPGMA algorithm. Multilocus sequence typing (MLST) Internal fragments of seven housekeeping genes (atpa, ddl, gdh, purk, gyd, psts, and adk) of our vanapositive E. faecium isolates were amplified and sequenced. The sequences obtained were analyzed and compared with those included in the website database ( in order to assign a specific sequence type (ST). STs were grouped into clonal complexes using the program eburst ( [20]. The type of purk allele was analyzed in the esp-positive vancomycin-susceptible E. faecium isolates to determine if they could have the purk1 allele (CC17-associated). 800
3 Results Prevalence and characterization of enterococci isolated in SB agar plates not supplemented with antibiotics Enterococci were recovered from 100 of the 300 tested samples (33.3%) when SB agar plates were used, and one enterococci per sample was further characterized. These positive samples corresponded to 80/250 (32%) inanimate surface samples, and 20/50 (40%) of hand samples (Table 1). E. faecium was the most prevalent species (86%), followed by E. faecalis (8%) and E. casseliflavus (6%) (Table 1). Antimicrobial resistance distributed by species of enterococcal strains isolated in SB medium not supplemented with vancomycin is shown intable 2. All enterococci, including E. faecium isolates, were susceptible to ampicillin and to a high dose of gentamicin. The antimicrobial resistance genes detected in these isolates by PCR are shown in Table 3. The majority of the erythromycin-resistant enterococci (71 of 73 isolates, 97.3%) harbored the erm(b) gene. All the 18 tetracycline-resistant enterococci (12 E. faecium, 4 E. faecalis, and 2 E. casseliflavus) carried the tet(m) gene. High-level resistance to streptomycin and kanamycin was also detected in some of the isolates; 15 of 27 streptomycinresistant strains carried the ant(6)-ia gene, and 27 of 28 kanamycin-resistant strains carried the aph(3 )-IIIa gene. The cat(a) gene was found in four chloramphenicol-resistant E. faecium isolates. No acquired vancomycin resistance was demonstrated among the enterococci recovered in SB agar plates not supplemented with vancomycin, although 6 E. casseliflavus isolates were obtained (with intrinsic lowlevel vancomycin resistance,with the vanc2gene). The esp gene, usually associated with nosocomial isolates, was detected by PCR in only 5 strains isolated from inanimate surfaces, all of them identified as E. faecium. These 5 esp-positive E. faecium strains had the purk17 allele, indicating that they were not ascribed to clonal complex CC17. Prevalence and characterization of VRE strains isolated in SB-VAN agar plates To determine the potential carriage of VRE in the 300 samples taken from the environment of the hospital, the samples were inoculated in SB-VAN agar plates (supplemented with 8 μg/ml of vancomycin). Table 1. Prevalence of Enterococcus strains in the hospital environment (isolated in Slanetz-Bartley medium not supplemented with antibiotics). Number of Number of isolates of the species Number of samples Origin of samples samples containing E. faecium E. faecalis E. casseliflavus enterococci Inanimate surfaces Hands of patient Hands of personnel Total Table 2. Antimicrobial resistance of enterococci strains isolated in Slanetz-Bartley medium not supplemented with antibiotics. Number of enterococci resistant to antimicrobial agents of the species Antimicrobial agent E. faecium (n = 86) E. faecalis (n = 8) E. casseliflavus (n = 6) Total (n = 100) Vancomycin * 6 Teicoplanin Ampicillin Streptomycin Gentamicin Kanamycin Chloramphenicol Tetracycline Erythromycin Trimethoprim-sulfamethoxazole Ciprofloxacin Pristanamycin * 6 E. casseliflavus isolated in Slanetz-Bartley not supplemented with vancomycin showed a low level resistance to vancomycin (intrinsic resistance conferred by the vanc2 gene). 801
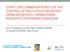
4 VRE were recovered from 14 of these 300 samples (4.7%). The 14 VRE strains (one per positive sample) were identified as E. faecium and carried the van(a)gene, showing high-level vancomycin and teicoplanin resistance (MIC > 256 μg/ml). The characteristics of the 14 vana-containing E. faecium isolates are shown in Table 4. These VRE isolates were recovered from different origins (12 from inanimate surfaces, 1 from the hand of a patient, and 1 from the hand of a healthcare worker), and from 6 different services of the hospital (Table 4). All the VRE isolates showed the same multiresistance phenotype to erythromycin and tetracycline as well as high-level resistance to streptomycin and kanamycin, and harbored erm(b), tet(m), ant(6)-ia, and aph(3 )- IIIa genes. The VRE isolates showed resistance to ampicillin and ciprofloxacin, but they were susceptible to high dose of gentamicin. All of them carried the IS16 element. Table 3. Resistance genes among enterococci isolated in Slanetz-Bartley medium not supplemented with antibiotics. Antibiotic Species Number of resistant strains Resistance genes detected (number of strains) Vancomycin E. casseliflavus 6 vanc2 (6) Erythromycin E. faecium 62 erm(b) (60) E. faecalis 6 erm(b) (6) E. casseliflavus 5 erm(b) (5) Tetracycline E. faecium 12 tet(m) (12) E. faecalis 4 tet(m) (4) E. casseliflavus 2 tet(m) (2) Chloramphenicol E. faecium 7 cat(a) (4) E. faecalis 1 - E. casseliflavus 2 - Kanamycin E. faecium 20 aph(3 )-IIIa (19) E. faecalis 5 aph(3 )-IIIa (5) E. casseliflavus 3 aph(3 )-IIIa (3) Streptomycin E. faecium 17 ant(6)-ia (10) E. faecalis 7 ant(6)-ia (3) E. casseliflavus 3 ant(6)-ia (2) Figure 1. Dendrogram prepared by FPQuest software using UPGMA algorithm (optimization 0.50%; tolerance 1.00%) of PFGE patterns of SmaI digested DNA from 14 vancomycin-resistant Enterococcus faecium strains isolated from the hospital environment. 802
5 Dziri et al. Multiresistant enterococci in hospital environment J Infect Dev Ctries 2016; 10(8): Table 4. Vancomycin-resistant E. faecium isolates recovered in Slanetz-Bartley medium supplemented with 8 µg/ml of vancomycin. Strains Origin Service Phenotype of resistance Genotype of resistance MIC (µg/ml) VA/TEC C7150 Treatment room Cardiothoracic surgery VA, TEC, TE, SXT, AM, KAN, STR, E, Cp vana, tet(m), aph (3 )-IIIa, ant (6)-Ia, erm(b) > 256 S New ST910 C7151 Treatment room Cardiothoracic surgery VA, TEC, TE, SXT, AM, KAN, STR, E, Cp vana, tet(m), aph (3 )-IIIa, ant (6)-Ia, erm(b) > 256 S New ST910 C7152 Handle door Hemodialysis VA, TEC, TE, SXT, AM, KAN, STR, E, Cp vana, tet(m), aph (3 )-IIIa, ant (6)-Ia, erm(b) > 256 S New ST910 C7153 Sink Ophtalmology VA, TEC, TE, SXT, AM, KAN, STR, E, Cp vana, tet(m), aph (3 )-IIIa, ant (6)-Ia, erm(b) > 256 S New ST910 C7154 Television Ophtalmology VA, TEC, TE, SXT, AM, KAN, STR, E, Cp vana, tet(m), aph (3 )-IIIa, ant (6)-Ia, erm(b) > 256 S New ST910 C7155 C7156 C7157 C7158 Handle door Hand of patient Bedlinen Sink Orthopedics and traumatology surgery Orthopedics and traumatology surgery Orthopedics and traumatology surgery Orthopedics and traumatology surgery PFGE ST/CC VA, TEC, TE, SXT, AM, KAN, STR, E, Cp vana, tet(m), aph (3 )-IIIa, ant (6)-Ia, erm(b) > 256 S New ST910 VA, TEC, TE, SXT, AM, KAN, STR, E, Cp vana, tet(m), aph (3 )-IIIa, ant (6)-Ia, erm(b) > 256 S New ST910 VA, TEC, TE, SXT, AM, KAN, STR, E, Cp vana, tet(m), aph (3 )-IIIa, ant (6)-Ia, erm(b) > 256 A ST80/CC17 VA, TEC, TE, SXT, AM, KAN, STR, E, Cp vana, tet(m), aph (3 )-IIIa, ant (6)-Ia, erm(b) > 256 S New ST910 C7159 Treatment table Pneumonology VA, TEC, TE, SXT, AM, KAN, STR, E, Cp vana, tet(m), aph (3 )-IIIa, ant (6)-Ia, erm(b) > 256 S New ST910 C7160 Handle door Pneumonology VA, TEC, TE, SXT, AM, KAN, STR, E, Cp vana, tet(m), aph (3 )-IIIa, ant (6)-Ia, erm(b) > 256 S New ST910 C7161 Hand of emloyee Pneumonology VA, TEC, TE, SXT, AM, KAN, STR, E, Cp vana, tet(m), aph (3 )-IIIa, ant (6)-Ia, erm(b) > 256 S New ST910 C7162 Medical apparatus ICU VA, TEC, TE, SXT, AM, KAN, STR, E, Cp vana, tet(m), aph (3 )-IIIa, ant (6)-Ia, erm(b) > 256 S New ST910 C7163 Chair ICU VA, TEC, TE, SXT, AM, KAN, STR, E, Cp vana, tet(m), aph (3 )-IIIa, ant (6)-Ia, erm(b) > 256 S New ST910 MIC (VAN/TEC): minimum inhibitory concentration of Vancomycin and Teicoplanin; PFGE: pulsed-field gel elecrophoresis ; ST: sequence type; CC: clonal complex VA: vancomycin; TEC: teicoplanin; TE: tetracycline; SXT: trimetoprim-sulfamethoxazole; AM: ampicillin; KAN: kanamycin; STR: streptomycin; E: erythromycin; Cp: ciprofloxacin.
6 The gene encoding esp was detected by PCR screening in all VRE isolates, but none of them contained the hyl Efmvirulence gene (encoding a hyaluronidase-like protein). Analysis of clonality of vana-containing E. faecium isolates The clonal relatedness of isolates was assessed using PFGE and MLST. The macrorestriction analysis of genomic DNA in the 14 vana-containing E. faecium isolates revealed two different profiles: 13 isolates showed the profile S and 1 isolate the profile A (Table 4) (Figure 1). Two different sequences types were identified among the vana isolates: the strain with PFGE-pattern A was ascribed to sequence type ST80 and clonal complex CC17, and the 13 vana strains with PFGEpattern S showed a new sequence type ST910 with the following allele combination: atpa (9), ddl (1), gdh (1), purk (1), gyd (16), psts (1), and adk (18) (Table 4). Discussion In this study, VRE isolates were detected in 14 of the 300 (4.7%) environmental samples at the hospital setting studied. All VRE isolates obtained were E. faecium and contained the vancomycin resistance vana gene. In Tunisia, the presence of VRE in hospitals is scarcely reported and there are only two previous reports of detection of vana-containing enterococci [6,12]. VRE was first isolated in this country in 2007 [12], and it still appears to be uncommon [6]. Nevertheless, since VRE could be an emerging pathogen, further studies of VRE infection and colonization are important. Nowadays, acquired vanatype vancomycin-resistant E. faecium are widely prevalent in hospitals around the world [21],and this situation could be similar in the future in Tunisia if appropriate control measures are not adopted. According to previous studies, the E. faecium species was the most prevalent among VRE clinical isolates [22,23]. The gene vana has also been detected in E. faecalis by other authors [18,24].Thirteen of our vana-positive isolates were ascribed by PFGE and MLST to a novel clone (new ST910) and only one strain was typed as ST80 and belonged to CC17. In a previous study performed by our group, three different PFGEtypes (A-C) and two sequence types (ST18 and ST80) were identified, a clone PFGE-A/ST80 being detected both among clinical and environmental vana E. faecium isolates during 2012 in the same hospital [6]. It is of interest that profile A detected in one of our isolates was indistinguishable from the profile A previously detected (with the same ST80/CC17) [6], indicating that this clone is still present in the environment of the hospital. All remaining vana-positive isolates belonged to a new clone, ST910/PFGE-S. These strains were vancomycin and ampicillin resistant, had purk (1), the esp gene, and all of them contained the insertion element IS16. All these characteristics are markers of nosocomial clones belonging to CC17. However, according to e-burst, this ST did not belong to CC17, but was a double locus variant of ST80, from which it could be derived. The appearance of this new clone in the studied hospital could be an alert of the emergence and rapid evolution and of these clinically important resistant bacteria and the necessity for active surveillance in other hospitals and regions of the country. In several recent studies carried out in different countries around the world (including Tunisia), VRE outbreaks seem to be related to monoclonal spread [6,25,26]. The homogeneity detected among our acquired vancomycin-resistant E. faecium isolates could be explained by the endemic dissemination of these VRE strains in the hospital. We found VRE in six different services of the hospital. The dissemination of vana-containing E. faecium strains in different areas of the hospital might be due to direct contact with colonized patients or even to indirect contact via the hands of healthcare workers [4,27]. The esp expression on the surface of E. faecium seems to play a role in adherence and biofilm formation [28]. This gene could have a great role in the exchanging genetic mobile elements and could also be implicated in the adaptation of these bacteria to the hospital environment [29,30].Remarkably, our vana E. faecium isolates presented a multiresistance phenotype, being resistant, in addition to glycopeptides, to erythromycin, tetracycline, ciprofloxacin, ampicillin, kanamycin, streptomycin, pristinamycin, and trimethoprimsulfamethoxazole. In the case of resistance genes erm(b) and tet(m), it is interesting to mention that they have been previously detected in the same conjugative plasmid as the van(a) gene, which indicates that coselection processes might have happened [31]. Our VRE strains were still susceptible to high dose of gentamicin, similar to what was found in a previous study reported in Tunisia showing the emergence of epidemic CC17 clones of vana E. faecium that were also sensitive to high dose of gentamicin [6]. On the other hand, the detection of enterococcal species (in non-supplemented media) in one-third of samples tested (inanimate surfaces of hospital and in the intact skin hand of some patients and healthcare workers) can be explained by the fact that these microorganisms are 804
7 able to survive in the environment outside the host for a long period of time and are able to resist routine cleaning [32]. The species more frequently detected, E. faecium and E. faecalis, are implicated in most enterococcal infections [33,34]. In fact, environmental contamination may contribute to transmission of pathogens by contact of medical equipment or patients with contaminated surfaces [35]. The wide dissemination of antibiotic resistance among our enterococci isolates could be partly due to the horizontal transfer of mobile genetic elements between strains and, on the other hand, to crosstransmission of multiresistant enterococci between patients, staff, and the environment [5]. Conclusions The dissemination of multi-resistant bacteria, including VRE of genotype vana, in the hospital has become particularly problematic. An active surveillance program should be carefully prepared to stop this uncontrollable evolution and emergence of these resistant bacteria. Further studies are needed and strict infection control strategies should be adopted in hospital environments to reduce potential infections by these microorganisms. Acknowledgements We thank the patients and staff members of the Military hospital of Tunis for their cooperation. This work was supported by the Tunisian Ministry of Higher Education and Scientific Research and by the Ministerio de Economía y Competitividad of Spain (project SAF ). C. Lozano has a contract associated with Project SAF Authors contributions RD and RB designed the sampling protocol and the bacteriological analyses; RD, LBS, CL, performed the molecular testing (all the PCR, MLST and PFGE); RD, NK and CL performed the analysis and the interpretation of all the data; RD, NK and CTO were involved in drafting the manuscript and revising it critically. BSK performed the analysis of the PFGE; NK, CTO and AB participated in its design and coordination and helped to draft and correct the manuscript. All authors contributed to and approved the submitted manuscript. References 1. Sood S, Malhotra M, Das B, Kapil A (2008) Enterococcal infections and antimicrobial resistance. Indian J Med Res 128: Lee BY, Yilmaz SL, Wong KF, Bartsch SM, Eubank S, Song Y, Avery TR, Christie R, Brown ST, Epstein JM, Parker JI, Huang SS (2013) Modeling the regional spread and control of vancomycin-resistant enterococci. Am J Infect Contro 41: Martinez J, Ruthazer R, Karen H, Barefoot L, Syndman D (2003) Role of environmental contamination as a risk factor for acquisition of vancomycin-resistant enterococci in patients treated in a medical intensive care unit. Arch Inter Med 163: Drees M, Snydman D, Schmid C, Barefoot L, Hansjosten K, Vue PM, Cronin M, Nasraway S, Golan Y (2008) Prior environmental contamination increases the risk of acquisition of vancomycin-resistant enterococci. Clin Infect Dis 46: Hayden M, Blom D, Lyle E, Moore C, Weistein R (2008) Risk of hand or glove contamination after contact with vancomycinresistant Enterococcus or the colonized patients environment. Infect Control Hosp Epidemiol 29: Elhani D, Klibi N, Dziri R, Ben Hassan M, Asli Mohamed S, Ben Said L, Aouini M, Ben Slama K, Jemli B, Bellaj R, Barguellil F, Torres C (2014) vana-containing E. faecium isolates of clonal complex CC17 in clinical and environmental samples in a Tunisian hospital. Diag Microbiol Infect Dis 79: Siegel J, Rhinehart E, Jackson M, Chiarello L (2006) Healthcare Infection Control Practices Advisory Committee. Management of multidrug-resistant organisms in healthcare setting. Atlanta: Centers for Disease Control and Prevention. 8. Lloyd-Smith P, Younger J, Lloyd-Smith E, Green H, Leung V, Romney M (2013) Economic analysis of vancomycin-resistant enterococci at a Canadian hospital: assessing attributable cost and length of stay. J Hosp Infect 85: Top J, Willems R, Bonten M (2008) Emergence of CC17 Enterococcus faecium: from commensal to hospital-adapted pathogen. FEMS Immunol Med Microbiol52: Hendrickx A, van Wamel W, Posthuma G, Bonten M, Willems R (2007) Five genes encoding surface-exposed LPXTG proteins are enriched in hospital-adapted Enterococcus faecium clonal complex 17 isolates. J Bacteriol 189: Willems RJ, Top J, van Santen M, Robinson DA, Coque TM, Baquero F, Grundmann H, Bonten MJ (2005) Global spread of vancomycin-resistant Enterococcus faecium from distinct nosocomial genetic complex. Emerg Infect Dis 11: Abbassi M, Znazen A, Mahjoubi F, Hammami A, Ben Hassen A (2007) Emergence of vancomycin-resistant Enterococus faecium in Sfax: clinical features and molecular typing. Med Mal Infect 37: Klibi N, Gharbi S, Masmoudi A, Ben Slama K, Poeta P, Zarazaga M, Fendri C, Boudabous A, Torres C (2006) Antibiotic resistance and mechanisms implicated in clinical enterococci in a Tunisian hospital. J Chemother 18: Clinical and Laboratory Standards Institute (2015) Performance standards for antimicrobial susceptibility testing. Twenty-fifth informational supplement. CLSI document M100-S Société Française de Microbiologie(SFM) (2013) Antibiogram Committee of the French Society for Microbiology. 805
8 Recommendations, Available: Accessed: 20 June Torres C, Tenorio C, Portillo A, García M, Martínez C, Del Campo R, Ruiz-larrea F, Zarazaga M (2003) Intestinal colonization by vana or vanb2-containing enterococcal isolates of healthy animals in Spain. Microb Drug Resist 9: Werner G, Fleige C, Geringer U, Van Schaik W, Klare I, Wolfgang W (2011) IS element IS 16 as a molecular screening tool to identify hospital-associated strains of Enterococcus faecium. BMC Infect Dis 11: Lopez M, Cercenado E, Tenorio C, Ruiz-Larrea F, Torres C (2012) Diversity of clones and genotypes among vancomycinresistant clinical Enterococcus isolates recovered in a Spanish hospital. Microb Drug Resist 18: Tenover F, Arbeit R, Goering R, Mickelsen P, Murray B, Persing D, Swaminathan B (1995) Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: criteria for bacterial strain typing. J Clin Microbiol33: Homan W, Tribe D, Poznanski S, Li M, Hogg M, Spalburg E,Van Embden J, Willems R (2002) Multilocus Sequence Typing scheme for Enterococcus faecium. J Clin Microbiol 40: Cattoir V, Leclercq R (2013) Twenty-five years of shared life with vancomycin-resistant enterococci: is it time to divorce? J Antimicrob Chemother 68: Panesso D, Reyes J, Rincón S, Díaz L, Galloway-Peña J, Zurita J, Carrillo C, Merentes A, Guzman M, Adachi JA, Murray BE, Arias CA (2010) Molecular epidemiology of vancomycinresistant Enterococcus faecium: a prospective, multicenter study in South American hospitals. J Clin Microbiol 48: Sun H, Wang H, Xu Y, Jones RN, Costello AJ, Liu Y, Li G, Chen M, Mendes RE (2012) Molecular characterization of vancomycin-resistant Enterococcus spp. clinical isolates recovered from hospitalized patients among several medical institutions in China. Diagn Microbiol Infect Dis 74: Pourakbari B, Aghdam MK, Mahmoudi S, Ashtiani MT, Sabouni F, Movahedi Z, Alyari AE, Sadeghi RH, Mamishi S (2012) High frequency of vancomycin-resistant Enterococcus faecalis in an Iranian referral children medical hospital. Maedica (Buchar) 7: D'Azevedo PA, Furtado GH, Medeiros EA, Santiago KA, Silbert S, Pignatari AC (2008) Molecular characterization of vancomycin-resistant Enterococci strains eight years apart from its first isolation in São Paulo, Brazil. Rev Inst Med Trop Sao Paulo 50: Valdezate S, Miranda C, Navarro A, Freitas AR, Cabrera JJ, Carrasco G, Coque TM, Jiménez-Romano E, Saéz-Nieto JA (2012) Clonal outbreak of ST17 multidrug-resistant Enterococcus faecium harbouring an Inc18-like: Tn1546 plasmid in a haemo-oncology ward of a Spanish hospital. J Antimicrob Chemother67: Ruzon F, De paula S, Kanoshiki R, Pereira-Santos J, Kerbauy G, Kobayashi R, Yamauchi L, Perugini M, Ogatta S (2010) Virulence determinants in vancomycin-resistant Enterococcus faeciumvana isolated from different sources at University hospital of Londrina, Parana Brazil.J Microbiol48: Van Wamel W, Hendrix A, Bonten M, Top J, Posthuma G, Willems R (2007) Growth condition-dependent Esp expression by Enterococcus faecium affects initial adherence and biofilm formation. Infect Immun 75: Kee S, Park C, Lee J, Kwon Y, Pyo H, Kim W, Cheong H (2012) Healthcare-associated risk factors of vancomycinresistant enterococci colonization among outpatients undergoing hemodialysis. J Infect Dis 65: Resende M, Caierão J, Gil Prates J, Narvaez G, Gomes Dias C, Alves d Azevedo P (2014) Emergence of vana vancomycinresistant Enterococcus faecium in a hospital in Porto Alegre, South Brazil. J Infect Dev Ctries8: doi: /jidc López M, Sáenz Y, Alvarez-Martínez MJ, Marco F, Robredo B, Rojo-Bezares B, Ruiz-Larrea F, Zarazaga M, Torres C (2010) Tn1546 structures and multilocus sequence typing of vana-containing enterococci of animal, human and food origin. J Antimicrob Chemother 65: Dancer S (1999) Mapping up hospital infection J Hosp Infect 43: Fisher K, Phillips C (2009) The ecology, epidemiology and virulence of Enterococcus. Microbiology 155: Orsi G, Ciorba V (2013) Vancomycin resistant enterococci healthcare associated infections. Ann Ig 25: Boyce J (2007) Environmental contamination makes an important contribution to hospital infection. J Hosp Infect 65: Corresponding author Naouel Klibi, PhD Laboratory of Microorganisms and Active Biomolecules, Faculty of Sciences of Tunis, University of Tunis El Manar, Campus Universitaire, ManarII 2092, Tunis, Tunisia Phone: , Fax: n_klibi@yahoo.fr Conflict of interests:no conflict of interests is declared. 806
Decrease of vancomycin resistance in Enterococcus faecium from bloodstream infections in
 AAC Accepted Manuscript Posted Online 30 March 2015 Antimicrob. Agents Chemother. doi:10.1128/aac.00513-15 Copyright 2015, American Society for Microbiology. All Rights Reserved. 1 2 Decrease of vancomycin
AAC Accepted Manuscript Posted Online 30 March 2015 Antimicrob. Agents Chemother. doi:10.1128/aac.00513-15 Copyright 2015, American Society for Microbiology. All Rights Reserved. 1 2 Decrease of vancomycin
ESCMID Online Lecture Library. by author
 ESCMID Postgraduate Technical Workshop Antimicrobial susceptibility testing and surveillance of resistance in Gram-positive cocci: laboratory to clinic Current epidemiology of invasive enterococci in Europe
ESCMID Postgraduate Technical Workshop Antimicrobial susceptibility testing and surveillance of resistance in Gram-positive cocci: laboratory to clinic Current epidemiology of invasive enterococci in Europe
ANTIMICROBIAL SUSCEPTIBILITY VANCOMYCIN RESISTANCE IN AN UNCOMMON ENTEROCOCCAL SPECIES
 ENTEROCOCCAL SPECIES Sample ES-02 was a simulated blood culture isolate from a patient with symptoms of sepsis. Participants were asked to identify any potential pathogen and to perform susceptibility
ENTEROCOCCAL SPECIES Sample ES-02 was a simulated blood culture isolate from a patient with symptoms of sepsis. Participants were asked to identify any potential pathogen and to perform susceptibility
Glycopeptide Resistant Enterococci (GRE) Policy IC/292/10
 BASINGSTOKE AND NORTH HAMPSHIRE NHS FOUNDATION TRUST Glycopeptide Resistant Enterococci (GRE) Policy IC/292/10 Supersedes: IC/292/07 Owner Name Dr Nicki Hutchinson Job Title Consultant Microbiologist,
BASINGSTOKE AND NORTH HAMPSHIRE NHS FOUNDATION TRUST Glycopeptide Resistant Enterococci (GRE) Policy IC/292/10 Supersedes: IC/292/07 Owner Name Dr Nicki Hutchinson Job Title Consultant Microbiologist,
EDUCATIONAL COMMENTARY - Methicillin-Resistant Staphylococcus aureus: An Update
 EDUCATIONAL COMMENTARY - Methicillin-Resistant Staphylococcus aureus: An Update Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain
EDUCATIONAL COMMENTARY - Methicillin-Resistant Staphylococcus aureus: An Update Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain
Presence of extended spectrum β-lactamase producing Escherichia coli in
 1 2 Presence of extended spectrum β-lactamase producing Escherichia coli in wild geese 3 4 5 A. Garmyn* 1, F. Haesebrouck 1, T. Hellebuyck 1, A. Smet 1, F. Pasmans 1, P. Butaye 2, A. Martel 1 6 7 8 9 10
1 2 Presence of extended spectrum β-lactamase producing Escherichia coli in wild geese 3 4 5 A. Garmyn* 1, F. Haesebrouck 1, T. Hellebuyck 1, A. Smet 1, F. Pasmans 1, P. Butaye 2, A. Martel 1 6 7 8 9 10
MRSA surveillance 2014: Poultry
 Vicky Jasson MRSA surveillance 2014: Poultry 1. Introduction In the framework of the FASFC surveillance, a surveillance of MRSA in poultry has been executed in order to determine the prevalence and diversity
Vicky Jasson MRSA surveillance 2014: Poultry 1. Introduction In the framework of the FASFC surveillance, a surveillance of MRSA in poultry has been executed in order to determine the prevalence and diversity
ORIGINAL ARTICLE /j x
 ORIGINAL ARTICLE 10.1111/j.1469-0691.2006.01533.x Genetic and phenotypic differences among Enterococcus faecalis clones from intestinal colonisation and invasive disease P. Ruiz-Garbajosa 1, R. Cantón
ORIGINAL ARTICLE 10.1111/j.1469-0691.2006.01533.x Genetic and phenotypic differences among Enterococcus faecalis clones from intestinal colonisation and invasive disease P. Ruiz-Garbajosa 1, R. Cantón
Consequences of Antimicrobial Resistant Bacteria. Antimicrobial Resistance. Molecular Genetics of Antimicrobial Resistance. Topics to be Covered
 Antimicrobial Resistance Consequences of Antimicrobial Resistant Bacteria Change in the approach to the administration of empiric antimicrobial therapy Increased number of hospitalizations Increased length
Antimicrobial Resistance Consequences of Antimicrobial Resistant Bacteria Change in the approach to the administration of empiric antimicrobial therapy Increased number of hospitalizations Increased length
MID 23. Antimicrobial Resistance. Consequences of Antimicrobial Resistant Bacteria. Molecular Genetics of Antimicrobial Resistance
 Antimicrobial Resistance Molecular Genetics of Antimicrobial Resistance Micro evolutionary change - point mutations Beta-lactamase mutation extends spectrum of the enzyme rpob gene (RNA polymerase) mutation
Antimicrobial Resistance Molecular Genetics of Antimicrobial Resistance Micro evolutionary change - point mutations Beta-lactamase mutation extends spectrum of the enzyme rpob gene (RNA polymerase) mutation
Antimicrobial Resistance
 Antimicrobial Resistance Consequences of Antimicrobial Resistant Bacteria Change in the approach to the administration of empiric antimicrobial therapy Increased number of hospitalizations Increased length
Antimicrobial Resistance Consequences of Antimicrobial Resistant Bacteria Change in the approach to the administration of empiric antimicrobial therapy Increased number of hospitalizations Increased length
Antimicrobial Resistance Acquisition of Foreign DNA
 Antimicrobial Resistance Acquisition of Foreign DNA Levy, Scientific American Horizontal gene transfer is common, even between Gram positive and negative bacteria Plasmid - transfer of single or multiple
Antimicrobial Resistance Acquisition of Foreign DNA Levy, Scientific American Horizontal gene transfer is common, even between Gram positive and negative bacteria Plasmid - transfer of single or multiple
Antimicrobial Resistance and Molecular Epidemiology of Staphylococcus aureus in Ghana
 Antimicrobial Resistance and Molecular Epidemiology of Staphylococcus aureus in Ghana Beverly Egyir, PhD Noguchi Memorial Institute for Medical Research Bacteriology Department, University of Ghana Background
Antimicrobial Resistance and Molecular Epidemiology of Staphylococcus aureus in Ghana Beverly Egyir, PhD Noguchi Memorial Institute for Medical Research Bacteriology Department, University of Ghana Background
Safe Patient Care Keeping our Residents Safe Use Standard Precautions for ALL Residents at ALL times
 Safe Patient Care Keeping our Residents Safe 2016 Use Standard Precautions for ALL Residents at ALL times #safepatientcare Do bugs need drugs? Dr Deirdre O Brien Consultant Microbiologist Mercy University
Safe Patient Care Keeping our Residents Safe 2016 Use Standard Precautions for ALL Residents at ALL times #safepatientcare Do bugs need drugs? Dr Deirdre O Brien Consultant Microbiologist Mercy University
Please distribute a copy of this information to each provider in your organization.
 HEALTH ADVISORY TO: Physicians and other Healthcare Providers Please distribute a copy of this information to each provider in your organization. Questions regarding this information may be directed to
HEALTH ADVISORY TO: Physicians and other Healthcare Providers Please distribute a copy of this information to each provider in your organization. Questions regarding this information may be directed to
Int.J.Curr.Microbiol.App.Sci (2018) 7(8):
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 7 Number 08 (2018) Journal homepage: http://www.ijcmas.com Original Research Article https://doi.org/10.20546/ijcmas.2018.708.378
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 7 Number 08 (2018) Journal homepage: http://www.ijcmas.com Original Research Article https://doi.org/10.20546/ijcmas.2018.708.378
Preventing Multi-Drug Resistant Organism (MDRO) Infections. For National Patient Safety Goal
 Preventing Multi-Drug Resistant Organism (MDRO) Infections For National Patient Safety Goal 07.03.01 2009 Methicillin Resistant Staphlococcus aureus (MRSA) About 3-8% of the population at large is a carrier
Preventing Multi-Drug Resistant Organism (MDRO) Infections For National Patient Safety Goal 07.03.01 2009 Methicillin Resistant Staphlococcus aureus (MRSA) About 3-8% of the population at large is a carrier
ENTEROCOCCI. April Abbott Deaconess Health System Evansville, IN
 ENTEROCOCCI April Abbott Deaconess Health System Evansville, IN OBJECTIVES Discuss basic antimicrobial susceptibility principles and resistance mechanisms for Enterococcus Describe issues surrounding AST
ENTEROCOCCI April Abbott Deaconess Health System Evansville, IN OBJECTIVES Discuss basic antimicrobial susceptibility principles and resistance mechanisms for Enterococcus Describe issues surrounding AST
Drug resistance & virulence determinants in clinical isolates of Enterococcus species
 Student IJMR Indian J Med Res 137, May 2013, pp 981-985 Drug resistance & virulence determinants in clinical isolates of Enterococcus species Sanal C. Fernandes & B. Dhanashree * M.B.B.S. Third year student,
Student IJMR Indian J Med Res 137, May 2013, pp 981-985 Drug resistance & virulence determinants in clinical isolates of Enterococcus species Sanal C. Fernandes & B. Dhanashree * M.B.B.S. Third year student,
Microbiological Surveillance of Methicillin Resistant Staphylococcus aureus (MRSA) in Belgian Hospitals in 2003
 Microbiological Surveillance of Methicillin Resistant Staphylococcus aureus (MRSA) in Belgian Hospitals in 3 Final report Olivier Denis and Marc J. Struelens Reference Laboratory for Staphylococci Department
Microbiological Surveillance of Methicillin Resistant Staphylococcus aureus (MRSA) in Belgian Hospitals in 3 Final report Olivier Denis and Marc J. Struelens Reference Laboratory for Staphylococci Department
Project Summary. Impact of Feeding Neomycin on the Emergence of Antibiotic Resistance in E. coli O157:H7 and Commensal Organisms
 Project Summary Impact of Feeding Neomycin on the Emergence of Antibiotic Resistance in E. coli O157:H7 and Commensal Organisms Principal Investigators: Mindy Brashears, Ph.D., Texas Tech University Guy
Project Summary Impact of Feeding Neomycin on the Emergence of Antibiotic Resistance in E. coli O157:H7 and Commensal Organisms Principal Investigators: Mindy Brashears, Ph.D., Texas Tech University Guy
Healthcare-associated vancomycin resistant Enterococcus faecium infections in the Mansoura University Hospitals intensive care units, Egypt
 Brazilian Journal of Microbiology 46, 3, 777-783 (2015) ISSN 1678-4405 DOI: http://dx.doi.org/10.1590/s1517-838246320140403 Copyright 2015, Sociedade Brasileira de Microbiologia www.sbmicrobiologia.org.br
Brazilian Journal of Microbiology 46, 3, 777-783 (2015) ISSN 1678-4405 DOI: http://dx.doi.org/10.1590/s1517-838246320140403 Copyright 2015, Sociedade Brasileira de Microbiologia www.sbmicrobiologia.org.br
High frequency distribution of heterogeneous vancomycin resistant Enterococcous faecium (VREfm) in Iranian hospitals
 Shokoohizadeh et al. Diagnostic Pathology 2013, 8:163 RESEARCH Open Access High frequency distribution of heterogeneous vancomycin resistant Enterococcous faecium (VREfm) in Iranian hospitals Leili Shokoohizadeh
Shokoohizadeh et al. Diagnostic Pathology 2013, 8:163 RESEARCH Open Access High frequency distribution of heterogeneous vancomycin resistant Enterococcous faecium (VREfm) in Iranian hospitals Leili Shokoohizadeh
Background and Plan of Analysis
 ENTEROCOCCI Background and Plan of Analysis UR-11 (2017) was sent to API participants as a simulated urine culture for recognition of a significant pathogen colony count, to perform the identification
ENTEROCOCCI Background and Plan of Analysis UR-11 (2017) was sent to API participants as a simulated urine culture for recognition of a significant pathogen colony count, to perform the identification
Origins of Resistance and Resistance Transfer: Food-Producing Animals.
 Origins of Resistance and Resistance Transfer: Food-Producing Animals. Chris Teale, AHVLA. Origins of Resistance. Mutation Brachyspira hyodysenteriae and macrolide and pleuromutilin resistance. Campylobacter
Origins of Resistance and Resistance Transfer: Food-Producing Animals. Chris Teale, AHVLA. Origins of Resistance. Mutation Brachyspira hyodysenteriae and macrolide and pleuromutilin resistance. Campylobacter
Is biocide resistance already a clinical problem?
 Is biocide resistance already a clinical problem? Stephan Harbarth, MD MS University of Geneva Hospitals and Faculty of Medicine, Geneva, Switzerland Important points Biocide resistance exists Antibiotic
Is biocide resistance already a clinical problem? Stephan Harbarth, MD MS University of Geneva Hospitals and Faculty of Medicine, Geneva, Switzerland Important points Biocide resistance exists Antibiotic
Antimicrobial Resistance
 Antimicrobial Resistance Consequences of Antimicrobial Resistant Bacteria Change in the approach to the administration of Change in the approach to the administration of empiric antimicrobial therapy Increased
Antimicrobial Resistance Consequences of Antimicrobial Resistant Bacteria Change in the approach to the administration of Change in the approach to the administration of empiric antimicrobial therapy Increased
Antimicrobial Resistance and Multilocus Sequence Typing of Vancomycin-Resistant Enterococcus faecium Isolated from the Chungcheong Area
 Korean J Clin Microbiol Vol., No., June, 0 DOI: 0./KJCM.0...0 Antimicrobial esistance and Multilocus Sequence Typing of Vancomycin-esistant Enterococcus faecium Isolated from the Chungcheong Area Hye Hyun
Korean J Clin Microbiol Vol., No., June, 0 DOI: 0./KJCM.0...0 Antimicrobial esistance and Multilocus Sequence Typing of Vancomycin-esistant Enterococcus faecium Isolated from the Chungcheong Area Hye Hyun
Prevalence of Metallo-Beta-Lactamase Producing Pseudomonas aeruginosa and its antibiogram in a tertiary care centre
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 4 Number 9 (2015) pp. 952-956 http://www.ijcmas.com Original Research Article Prevalence of Metallo-Beta-Lactamase
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 4 Number 9 (2015) pp. 952-956 http://www.ijcmas.com Original Research Article Prevalence of Metallo-Beta-Lactamase
Methicillin-Resistant Staphylococcus aureus
 Methicillin-Resistant Staphylococcus aureus By Karla Givens Means of Transmission and Usual Reservoirs Staphylococcus aureus is part of normal flora and can be found on the skin and in the noses of one
Methicillin-Resistant Staphylococcus aureus By Karla Givens Means of Transmission and Usual Reservoirs Staphylococcus aureus is part of normal flora and can be found on the skin and in the noses of one
Other Enterobacteriaceae
 GUIDE TO INFECTION CONTROL IN THE HOSPITAL CHAPTER NUMBER 50: Other Enterobacteriaceae Author Kalisvar Marimuthu, MD Chapter Editor Michelle Doll, MD, MPH Topic Outline Topic outline - Key Issues Known
GUIDE TO INFECTION CONTROL IN THE HOSPITAL CHAPTER NUMBER 50: Other Enterobacteriaceae Author Kalisvar Marimuthu, MD Chapter Editor Michelle Doll, MD, MPH Topic Outline Topic outline - Key Issues Known
ESBL Producers An Increasing Problem: An Overview Of An Underrated Threat
 ESBL Producers An Increasing Problem: An Overview Of An Underrated Threat Hicham Ezzat Professor of Microbiology and Immunology Cairo University Introduction 1 Since the 1980s there have been dramatic
ESBL Producers An Increasing Problem: An Overview Of An Underrated Threat Hicham Ezzat Professor of Microbiology and Immunology Cairo University Introduction 1 Since the 1980s there have been dramatic
Antimicrobial Resistance Strains
 Antimicrobial Resistance Strains Microbiologics offers a wide range of strains with characterized antimicrobial resistance mechanisms including: Extended-Spectrum β-lactamases (ESBLs) Carbapenamases Vancomycin-Resistant
Antimicrobial Resistance Strains Microbiologics offers a wide range of strains with characterized antimicrobial resistance mechanisms including: Extended-Spectrum β-lactamases (ESBLs) Carbapenamases Vancomycin-Resistant
Monitoring of antimicrobial resistance in Campylobacter EURL AR activities in framework of the new EU regulation Lina Cavaco
 Monitoring of antimicrobial resistance in Campylobacter EURL AR activities in framework of the new EU regulation Lina Cavaco licav@food.dtu.dk 1 DTU Food, Technical University of Denmark Outline EURL-AR
Monitoring of antimicrobial resistance in Campylobacter EURL AR activities in framework of the new EU regulation Lina Cavaco licav@food.dtu.dk 1 DTU Food, Technical University of Denmark Outline EURL-AR
Frank Møller Aarestrup
 Danish Veterinary Laboratory Bacterial populations and resistance development: Intestinal tract of meat animals Frank Møller Aarestrup 12 Antibiotic production 10 Mill. Kg 8 6 4 2 0 50 52 54 56 58 60 62
Danish Veterinary Laboratory Bacterial populations and resistance development: Intestinal tract of meat animals Frank Møller Aarestrup 12 Antibiotic production 10 Mill. Kg 8 6 4 2 0 50 52 54 56 58 60 62
ANTIMICROBIAL SUSCEPTIBILITY CONTEMPORARY SUSCEPTIBILITY TESTS AND TREATMENTS FOR VRE INFECTIONS
 TREATMENTS FOR VRE INFECTIONS Sample ES-01 (2015) was a simulated blood culture isolate from a patient with associated clinical symptoms (pure culture). Participants were requested to identify any potential
TREATMENTS FOR VRE INFECTIONS Sample ES-01 (2015) was a simulated blood culture isolate from a patient with associated clinical symptoms (pure culture). Participants were requested to identify any potential
MRSA Control : Belgian policy
 MRSA Control : Belgian policy PEN ERY CLI DOT GEN KAN SXT CIP MIN RIF FUC MUP OXA Marc Struelens Service de microbiologie & unité d épidémiologie des maladies infectieuses Université Libre de Bruxelles
MRSA Control : Belgian policy PEN ERY CLI DOT GEN KAN SXT CIP MIN RIF FUC MUP OXA Marc Struelens Service de microbiologie & unité d épidémiologie des maladies infectieuses Université Libre de Bruxelles
Iberian Lynx (Lynx pardinus) from the captive breeding program as reservoir of antimicrobial resistant enterococci and Escherichia coli isolates
 JOURNAL OF INTEGRATED OMICS Journal of Integrated Omics A METHODOLOGICAL JOURNAL HTTP://WWW.JIOMICS.COM ORIGINAL ARTICLE DOI: 10.5584/jiomics.v3i2.142 Iberian Lynx (Lynx pardinus) from the captive breeding
JOURNAL OF INTEGRATED OMICS Journal of Integrated Omics A METHODOLOGICAL JOURNAL HTTP://WWW.JIOMICS.COM ORIGINAL ARTICLE DOI: 10.5584/jiomics.v3i2.142 Iberian Lynx (Lynx pardinus) from the captive breeding
β-lactams resistance among Enterobacteriaceae in Morocco 1 st ICREID Addis Ababa March 2018
 β-lactams resistance among Enterobacteriaceae in Morocco 1 st ICREID Addis Ababa 12-14 March 2018 Antibiotic resistance center Institut Pasteur du Maroc Enterobacteriaceae (E. coli, Salmonella, ) S. aureus
β-lactams resistance among Enterobacteriaceae in Morocco 1 st ICREID Addis Ababa 12-14 March 2018 Antibiotic resistance center Institut Pasteur du Maroc Enterobacteriaceae (E. coli, Salmonella, ) S. aureus
GUIDE TO INFECTION CONTROL IN THE HOSPITAL. Enterococcal Species
 GUIDE TO INFECTION CONTROL IN THE HOSPITAL CHAPTER 44 Enterococcal Species Authors Jacob Pierce, MD, Michael Edmond, MD, MPH, MPA Michael P. Stevens, MD, MPH Chapter Editor Victor D. Rosenthal, MD, CIC,
GUIDE TO INFECTION CONTROL IN THE HOSPITAL CHAPTER 44 Enterococcal Species Authors Jacob Pierce, MD, Michael Edmond, MD, MPH, MPA Michael P. Stevens, MD, MPH Chapter Editor Victor D. Rosenthal, MD, CIC,
Understanding and prevention of transmission of antibiotic resistance between bacterial populations and One Health reservoirs
 Priority Topic D - Transmission Understanding and prevention of transmission of antibiotic resistance between bacterial populations and One Health reservoirs The overarching goal of this priority topic
Priority Topic D - Transmission Understanding and prevention of transmission of antibiotic resistance between bacterial populations and One Health reservoirs The overarching goal of this priority topic
Principles of Antimicrobial Therapy
 Principles of Antimicrobial Therapy Doo Ryeon Chung, MD, PhD Professor of Medicine, Division of Infectious Diseases Director, Infection Control Office SUNGKYUNKWAN UNIVERSITY SCHOOL OF MEDICINE CASE 1
Principles of Antimicrobial Therapy Doo Ryeon Chung, MD, PhD Professor of Medicine, Division of Infectious Diseases Director, Infection Control Office SUNGKYUNKWAN UNIVERSITY SCHOOL OF MEDICINE CASE 1
Research Article Risk Factors Associated with Vancomycin-Resistant Enterococcus in Intensive Care Unit Settings in Saudi Arabia
 Interdisciplinary Perspectives on Infectious Diseases Volume 2013, Article ID 369674, 4 pages http://dx.doi.org/10.1155/2013/369674 Research Article Risk Factors Associated with Vancomycin-Resistant Enterococcus
Interdisciplinary Perspectives on Infectious Diseases Volume 2013, Article ID 369674, 4 pages http://dx.doi.org/10.1155/2013/369674 Research Article Risk Factors Associated with Vancomycin-Resistant Enterococcus
PILOT STUDY OF THE ANTIMICROBIAL SUSCEPTIBILITY OF SHIGELLA IN NEW ZEALAND IN 1996
 PILOT STUDY OF THE ANTIMICROBIAL SUSCEPTIBILITY OF SHIGELLA IN NEW ZEALAND IN 996 November 996 by Maggie Brett Antibiotic Reference Laboratory ESR Communicable Disease Centre Porirua CONTENTS Page SUMMARY
PILOT STUDY OF THE ANTIMICROBIAL SUSCEPTIBILITY OF SHIGELLA IN NEW ZEALAND IN 996 November 996 by Maggie Brett Antibiotic Reference Laboratory ESR Communicable Disease Centre Porirua CONTENTS Page SUMMARY
Detection and Quantitation of the Etiologic Agents of Ventilator Associated Pneumonia in Endotracheal Tube Aspirates From Patients in Iran
 Letter to the Editor Detection and Quantitation of the Etiologic Agents of Ventilator Associated Pneumonia in Endotracheal Tube Aspirates From Patients in Iran Mohammad Rahbar, PhD; Massoud Hajia, PhD
Letter to the Editor Detection and Quantitation of the Etiologic Agents of Ventilator Associated Pneumonia in Endotracheal Tube Aspirates From Patients in Iran Mohammad Rahbar, PhD; Massoud Hajia, PhD
Molecular epidemiology of community-acquired methicillin-resistant Staphylococcus aureus bacteremia in a teaching hospital
 Epidemiology J Microbiol Immunol of MRSA Infect. bacteremia 2007;40:310-316 Molecular epidemiology of community-acquired methicillin-resistant Staphylococcus aureus bacteremia in a teaching hospital Chih-Yu
Epidemiology J Microbiol Immunol of MRSA Infect. bacteremia 2007;40:310-316 Molecular epidemiology of community-acquired methicillin-resistant Staphylococcus aureus bacteremia in a teaching hospital Chih-Yu
Mechanisms and Pathways of AMR in the environment
 FMM/RAS/298: Strengthening capacities, policies and national action plans on prudent and responsible use of antimicrobials in fisheries Final Workshop in cooperation with AVA Singapore and INFOFISH 12-14
FMM/RAS/298: Strengthening capacities, policies and national action plans on prudent and responsible use of antimicrobials in fisheries Final Workshop in cooperation with AVA Singapore and INFOFISH 12-14
Multiple drug resistance pattern in Urinary Tract Infection patients in Aligarh
 Multiple drug resistance pattern in Urinary Tract Infection patients in Aligarh Author(s): Asad U Khan and Mohd S Zaman Vol. 17, No. 3 (2006-09 - 2006-12) Biomedical Research 2006; 17 (3): 179-181 Asad
Multiple drug resistance pattern in Urinary Tract Infection patients in Aligarh Author(s): Asad U Khan and Mohd S Zaman Vol. 17, No. 3 (2006-09 - 2006-12) Biomedical Research 2006; 17 (3): 179-181 Asad
ESCHERICHIA COLI RESISTANCE AND GUT MICROBIOTA PROFILE IN PIGS RAISED WITH DIFFERENT ANTIMICROBIAL ADMINISTRATION IN FEED
 ESCHERICHIA COLI RESISTANCE AND GUT MICROBIOTA PROFILE IN PIGS RAISED WITH DIFFERENT ANTIMICROBIAL ADMINISTRATION IN FEED Caroline Pissetti 1, Jalusa Deon Kich 2, Heather K. Allen 3, Claudia Navarrete
ESCHERICHIA COLI RESISTANCE AND GUT MICROBIOTA PROFILE IN PIGS RAISED WITH DIFFERENT ANTIMICROBIAL ADMINISTRATION IN FEED Caroline Pissetti 1, Jalusa Deon Kich 2, Heather K. Allen 3, Claudia Navarrete
Antimicrobial Resistance: Do we know everything? Dr. Sid Thakur Assistant Professor Swine Health & Production CVM, NCSU
 Antimicrobial Resistance: Do we know everything? Dr. Sid Thakur Assistant Professor Swine Health & Production CVM, NCSU Research Focus Antimicrobial Resistance On farm, Slaughter, Retail, Human Sample
Antimicrobial Resistance: Do we know everything? Dr. Sid Thakur Assistant Professor Swine Health & Production CVM, NCSU Research Focus Antimicrobial Resistance On farm, Slaughter, Retail, Human Sample
Evolution of antibiotic resistance. October 10, 2005
 Evolution of antibiotic resistance October 10, 2005 Causes of death, 2001: USA 6. Population: 6,122,210,000 Deaths: 56,554,000 1. Infectious and parasitic diseases: 14.9 million 1. 2. 3. 4. 5. 2. Heart
Evolution of antibiotic resistance October 10, 2005 Causes of death, 2001: USA 6. Population: 6,122,210,000 Deaths: 56,554,000 1. Infectious and parasitic diseases: 14.9 million 1. 2. 3. 4. 5. 2. Heart
SUPPLEMENT ARTICLE. Donald E. Low, 1 Nathan Keller, 2 Alfonso Barth, 3 and Ronald N. Jones 4
 SUPPLEMENT ARTICLE Clinical Prevalence, Antimicrobial Susceptibility, and Geographic Resistance Patterns of Enterococci: Results from the SENTRY Antimicrobial Surveillance Program, 1997 1999 Donald E.
SUPPLEMENT ARTICLE Clinical Prevalence, Antimicrobial Susceptibility, and Geographic Resistance Patterns of Enterococci: Results from the SENTRY Antimicrobial Surveillance Program, 1997 1999 Donald E.
Tel: Fax:
 CONCISE COMMUNICATION Bactericidal activity and synergy studies of BAL,a novel pyrrolidinone--ylidenemethyl cephem,tested against streptococci, enterococci and methicillin-resistant staphylococci L. M.
CONCISE COMMUNICATION Bactericidal activity and synergy studies of BAL,a novel pyrrolidinone--ylidenemethyl cephem,tested against streptococci, enterococci and methicillin-resistant staphylococci L. M.
The Basics: Using CLSI Antimicrobial Susceptibility Testing Standards
 The Basics: Using CLSI Antimicrobial Susceptibility Testing Standards Janet A. Hindler, MCLS, MT(ASCP) UCLA Health System Los Angeles, California, USA jhindler@ucla.edu 1 Learning Objectives Describe information
The Basics: Using CLSI Antimicrobial Susceptibility Testing Standards Janet A. Hindler, MCLS, MT(ASCP) UCLA Health System Los Angeles, California, USA jhindler@ucla.edu 1 Learning Objectives Describe information
Virulence and Antimicrobial Resistance in Enterococci Isolated from Urinary Tract Infections
 Advanced Pharmaceutical Bulletin, 2013, 3(1), 197-201 doi: http://dx.doi.org/10.5681/apb.2013.032 http://apb.tbzmed.ac.ir/ Virulence and Antimicrobial Resistance in Enterococci Isolated from Urinary Tract
Advanced Pharmaceutical Bulletin, 2013, 3(1), 197-201 doi: http://dx.doi.org/10.5681/apb.2013.032 http://apb.tbzmed.ac.ir/ Virulence and Antimicrobial Resistance in Enterococci Isolated from Urinary Tract
A Study on Bacterial Flora on the Finger printing Surface of the Biometric Devices at a Tertiary Care Hospital
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 5 Number 9 (2016) pp. 441-446 Journal homepage: http://www.ijcmas.com Original Research Article http://dx.doi.org/10.20546/ijcmas.2016.509.047
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 5 Number 9 (2016) pp. 441-446 Journal homepage: http://www.ijcmas.com Original Research Article http://dx.doi.org/10.20546/ijcmas.2016.509.047
Antimicrobial Activity of Linezolid Against Gram-Positive Cocci Isolated in Brazil
 BJID 2001; 5 (August) 171 Antimicrobial Activity of Linezolid Against Gram-Positive Cocci Isolated in Brazil Helio S. Sader, Ana C. Gales and Ronald N. Jones Special Clinical Microbiology Laboratory, Division
BJID 2001; 5 (August) 171 Antimicrobial Activity of Linezolid Against Gram-Positive Cocci Isolated in Brazil Helio S. Sader, Ana C. Gales and Ronald N. Jones Special Clinical Microbiology Laboratory, Division
Enterococci of animal origin and their significance for public health
 REVIEW 10.1111/j.1469-0691.2012.03829.x Enterococci of animal origin and their significance for public health A. M. Hammerum Department of Microbiological Surveillance and Research, Statens Serum Institut,
REVIEW 10.1111/j.1469-0691.2012.03829.x Enterococci of animal origin and their significance for public health A. M. Hammerum Department of Microbiological Surveillance and Research, Statens Serum Institut,
Antibiotic Reference Laboratory, Institute of Environmental Science and Research Limited (ESR); August 2017
 Antimicrobial susceptibility of Shigella, 2015 and 2016 Helen Heffernan and Rosemary Woodhouse Antibiotic Reference Laboratory, Institute of Environmental Science and Research Limited (ESR); August 2017
Antimicrobial susceptibility of Shigella, 2015 and 2016 Helen Heffernan and Rosemary Woodhouse Antibiotic Reference Laboratory, Institute of Environmental Science and Research Limited (ESR); August 2017
MICRONAUT MICRONAUT-S Detection of Resistance Mechanisms. Innovation with Integrity BMD MIC
 MICRONAUT Detection of Resistance Mechanisms Innovation with Integrity BMD MIC Automated and Customized Susceptibility Testing For detection of resistance mechanisms and specific resistances of clinical
MICRONAUT Detection of Resistance Mechanisms Innovation with Integrity BMD MIC Automated and Customized Susceptibility Testing For detection of resistance mechanisms and specific resistances of clinical
Drd. OBADĂ MIHAI DORU. PhD THESIS ABSTRACT
 UNIVERSITY OF AGRICULTURAL SCIENCES AND VETERINARY MEDICINE ION IONESCU DE LA BRAD IAŞI FACULTY OF VETERINARY MEDICINE SPECIALIZATION MICROBIOLOGY- IMUNOLOGY Drd. OBADĂ MIHAI DORU PhD THESIS ABSTRACT RESEARCHES
UNIVERSITY OF AGRICULTURAL SCIENCES AND VETERINARY MEDICINE ION IONESCU DE LA BRAD IAŞI FACULTY OF VETERINARY MEDICINE SPECIALIZATION MICROBIOLOGY- IMUNOLOGY Drd. OBADĂ MIHAI DORU PhD THESIS ABSTRACT RESEARCHES
6. STORAGE INSTRUCTIONS
 VRESelect 63751 A selective and differential chromogenic medium for the qualitative detection of gastrointestinal colonization of vancomycin-resistant Enterococcus faecium () and vancomycin-resistant Enterococcus
VRESelect 63751 A selective and differential chromogenic medium for the qualitative detection of gastrointestinal colonization of vancomycin-resistant Enterococcus faecium () and vancomycin-resistant Enterococcus
SURVIVABILITY OF HIGH RISK, MULTIRESISTANT BACTERIA ON COTTON TREATED WITH COMMERCIALLY AVAILABLE ANTIMICROBIAL AGENTS
 SURVIVABILITY OF HIGH RISK, MULTIRESISTANT BACTERIA ON COTTON TREATED WITH COMMERCIALLY AVAILABLE ANTIMICROBIAL AGENTS Adrienn Hanczvikkel 1, András Vígh 2, Ákos Tóth 3,4 1 Óbuda University, Budapest,
SURVIVABILITY OF HIGH RISK, MULTIRESISTANT BACTERIA ON COTTON TREATED WITH COMMERCIALLY AVAILABLE ANTIMICROBIAL AGENTS Adrienn Hanczvikkel 1, András Vígh 2, Ákos Tóth 3,4 1 Óbuda University, Budapest,
Detection of Methicillin Resistant Strains of Staphylococcus aureus Using Phenotypic and Genotypic Methods in a Tertiary Care Hospital
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 6 Number 7 (2017) pp. 4008-4014 Journal homepage: http://www.ijcmas.com Original Research Article https://doi.org/10.20546/ijcmas.2017.607.415
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 6 Number 7 (2017) pp. 4008-4014 Journal homepage: http://www.ijcmas.com Original Research Article https://doi.org/10.20546/ijcmas.2017.607.415
Driving forces of vancomycin-resistant E. faecium and E. faecalis blood-stream infections in children
 Di Pentima et al. Antimicrobial Resistance and Infection Control 2014, 3:29 RESEARCH Open Access Driving forces of vancomycin-resistant E. faecium and E. faecalis blood-stream infections in children Maria
Di Pentima et al. Antimicrobial Resistance and Infection Control 2014, 3:29 RESEARCH Open Access Driving forces of vancomycin-resistant E. faecium and E. faecalis blood-stream infections in children Maria
Typing of Enterococcus spp. strains in 4 hospitals in the Małopolska region in Poland
 Original papers Typing of Enterococcus spp. in 4 hospitals in the Małopolska region in Poland Katarzyna Talaga 1, A F, Dalma Odrowąż-Konduracka 2, B, F, Beata Paradowska 3, B, F, Barbara Jagiencarz-Starzec
Original papers Typing of Enterococcus spp. in 4 hospitals in the Małopolska region in Poland Katarzyna Talaga 1, A F, Dalma Odrowąż-Konduracka 2, B, F, Beata Paradowska 3, B, F, Barbara Jagiencarz-Starzec
MRCoNS : .Duplex-PCR.
 - ( ) - * (MRCoNS) : Vancomycin Resistant Coagulase Negative ) VRCoNS. (Vancomycin Intermediate Coagulase Negative Staphylococci) VICoNS (Staphylococci Methicillin-Resistant Coagulase ) MRCoNS.. VRCoNS
- ( ) - * (MRCoNS) : Vancomycin Resistant Coagulase Negative ) VRCoNS. (Vancomycin Intermediate Coagulase Negative Staphylococci) VICoNS (Staphylococci Methicillin-Resistant Coagulase ) MRCoNS.. VRCoNS
Does Screening for MRSA Colonization Have A Role In Healthcare-Associated Infection Prevention Programs?
 Does Screening for MRSA Colonization Have A Role In Healthcare-Associated Infection Prevention Programs? John A. Jernigan, MD, MS Division of Healthcare Quality Promotion Centers for Disease Control and
Does Screening for MRSA Colonization Have A Role In Healthcare-Associated Infection Prevention Programs? John A. Jernigan, MD, MS Division of Healthcare Quality Promotion Centers for Disease Control and
WHY IS THIS IMPORTANT?
 CHAPTER 20 ANTIBIOTIC RESISTANCE WHY IS THIS IMPORTANT? The most important problem associated with infectious disease today is the rapid development of resistance to antibiotics It will force us to change
CHAPTER 20 ANTIBIOTIC RESISTANCE WHY IS THIS IMPORTANT? The most important problem associated with infectious disease today is the rapid development of resistance to antibiotics It will force us to change
Antimicrobial Stewardship Strategy: Antibiograms
 Antimicrobial Stewardship Strategy: Antibiograms A summary of the cumulative susceptibility of bacterial isolates to formulary antibiotics in a given institution or region. Its main functions are to guide
Antimicrobial Stewardship Strategy: Antibiograms A summary of the cumulative susceptibility of bacterial isolates to formulary antibiotics in a given institution or region. Its main functions are to guide
Co-transfer of bla NDM-5 and mcr-1 by an IncX3 X4 hybrid plasmid in Escherichia coli 4
 SUPPLEMENTARY INFORMATION ARTICLE NUMBER: 16176 DOI: 10.1038/NMICROBIOL.2016.176 Co-transfer of bla NDM-5 and mcr-1 by an IncX3 X4 hybrid plasmid in Escherichia coli 4 5 6 7 8 9 10 11 12 13 14 15 16 17
SUPPLEMENTARY INFORMATION ARTICLE NUMBER: 16176 DOI: 10.1038/NMICROBIOL.2016.176 Co-transfer of bla NDM-5 and mcr-1 by an IncX3 X4 hybrid plasmid in Escherichia coli 4 5 6 7 8 9 10 11 12 13 14 15 16 17
Informing Public Policy on Agricultural Use of Antimicrobials in the United States: Strategies Developed by an NGO
 Informing Public Policy on Agricultural Use of Antimicrobials in the United States: Strategies Developed by an NGO Stephen J. DeVincent, DVM, MA Director, Ecology Program Alliance for the Prudent Use of
Informing Public Policy on Agricultural Use of Antimicrobials in the United States: Strategies Developed by an NGO Stephen J. DeVincent, DVM, MA Director, Ecology Program Alliance for the Prudent Use of
Antibiotic resistance of bacteria along the food chain: A global challenge for food safety
 GREASE Annual Scientific Seminar. NIVR, 17-18th March 2014. Hanoi-Vietnam Antibiotic resistance of bacteria along the food chain: A global challenge for food safety Samira SARTER CIRAD-UMR Qualisud Le
GREASE Annual Scientific Seminar. NIVR, 17-18th March 2014. Hanoi-Vietnam Antibiotic resistance of bacteria along the food chain: A global challenge for food safety Samira SARTER CIRAD-UMR Qualisud Le
Q1. (a) Clostridium difficile is a bacterium that is present in the gut of up to 3% of healthy adults and 66% of healthy infants.
 Q1. (a) Clostridium difficile is a bacterium that is present in the gut of up to 3% of healthy adults and 66% of healthy infants. C. difficile rarely causes problems, either in healthy adults or in infants.
Q1. (a) Clostridium difficile is a bacterium that is present in the gut of up to 3% of healthy adults and 66% of healthy infants. C. difficile rarely causes problems, either in healthy adults or in infants.
MDR Acinetobacter baumannii. Has the post antibiotic era arrived? Dr. Michael A. Borg Infection Control Dept Mater Dei Hospital Malta
 MDR Acinetobacter baumannii Has the post antibiotic era arrived? Dr. Michael A. Borg Infection Control Dept Mater Dei Hospital Malta 1 The Armageddon recipe Transmissible organism with prolonged environmental
MDR Acinetobacter baumannii Has the post antibiotic era arrived? Dr. Michael A. Borg Infection Control Dept Mater Dei Hospital Malta 1 The Armageddon recipe Transmissible organism with prolonged environmental
Characterization of Vancomycin-Resistant Enterococcus faecium Isolates from Broiler Poultry and Pig Farms in England and Wales
 JOURNAL OF CLINICAL MICROBIOLOGY, July 2005, p. 3283 3289 Vol. 43, No. 7 0095-1137/05/$08.00 0 doi:10.1128/jcm.43.7.3283 3289.2005 Characterization of Vancomycin-Resistant Enterococcus faecium Isolates
JOURNAL OF CLINICAL MICROBIOLOGY, July 2005, p. 3283 3289 Vol. 43, No. 7 0095-1137/05/$08.00 0 doi:10.1128/jcm.43.7.3283 3289.2005 Characterization of Vancomycin-Resistant Enterococcus faecium Isolates
Vancomycin resistant Enterococcus spp (VRE): Follow
 http://dx.doi.org/10.4322/2357-9730.50891 Original Article Vancomycin resistant Enterococcus spp (VRE): Follow up during 9 years in a Tertiary Teaching Hospital in Southern Brazil Ana Maria Sandri 1, Geórgia
http://dx.doi.org/10.4322/2357-9730.50891 Original Article Vancomycin resistant Enterococcus spp (VRE): Follow up during 9 years in a Tertiary Teaching Hospital in Southern Brazil Ana Maria Sandri 1, Geórgia
Animal Antibiotic Use and Public Health
 A data table from Nov 2017 Animal Antibiotic Use and Public Health The selected studies below were excerpted from Pew s peer-reviewed 2017 article Antimicrobial Drug Use in Food-Producing Animals and Associated
A data table from Nov 2017 Animal Antibiotic Use and Public Health The selected studies below were excerpted from Pew s peer-reviewed 2017 article Antimicrobial Drug Use in Food-Producing Animals and Associated
Antimicrobial Resistance and Prescribing
 Antimicrobial Resistance and Prescribing John Ferguson, Microbiology & Infectious Diseases, John Hunter Hospital, University of Newcastle, NSW, Australia M Med Part 1 updates UPNG 2017 Tw @mdjkf http://idmic.net
Antimicrobial Resistance and Prescribing John Ferguson, Microbiology & Infectious Diseases, John Hunter Hospital, University of Newcastle, NSW, Australia M Med Part 1 updates UPNG 2017 Tw @mdjkf http://idmic.net
DR. MICHAEL A. BORG DIRECTOR OF INFECTION PREVENTION & CONTROL MATER DEI HOSPITAL - MALTA
 DR. MICHAEL A. BORG DIRECTOR OF INFECTION PREVENTION & CONTROL MATER DEI HOSPITAL - MALTA The good old days The dread (of) infections that used to rage through the whole communities is muted Their retreat
DR. MICHAEL A. BORG DIRECTOR OF INFECTION PREVENTION & CONTROL MATER DEI HOSPITAL - MALTA The good old days The dread (of) infections that used to rage through the whole communities is muted Their retreat
Twenty Years of the National Antimicrobial Resistance Monitoring System (NARMS) Where Are We And What Is Next?
 Twenty Years of the National Antimicrobial Resistance Monitoring System (NARMS) Where Are We And What Is Next? Patrick McDermott, Ph.D. Director, NARMS Food & Drug Administration Center for Veterinary
Twenty Years of the National Antimicrobial Resistance Monitoring System (NARMS) Where Are We And What Is Next? Patrick McDermott, Ph.D. Director, NARMS Food & Drug Administration Center for Veterinary
What is antimicrobial resistance?
 What is antimicrobial resistance? Gérard MOULIN gerard.moulin@anses.fr French agency for food, environmental and occupationnal safety National agency for veterinary Medicinal Products BP 90203-35302 FOUGERES
What is antimicrobial resistance? Gérard MOULIN gerard.moulin@anses.fr French agency for food, environmental and occupationnal safety National agency for veterinary Medicinal Products BP 90203-35302 FOUGERES
(DRAFT) RECOMMENDATIONS FOR THE CONTROL OF MULTI-DRUG RESISTANT GRAM-NEGATIVES: CARBAPENEM RESISTANT ENTEROBACTERIACEAE
 (DRAFT) RECOMMENDATIONS FOR THE CONTROL OF MULTI-DRUG RESISTANT GRAM-NEGATIVES: CARBAPENEM RESISTANT ENTEROBACTERIACEAE John Ferguson (Hunter New England, NSW) on behalf of MRGN Task Force Acknowledgement
(DRAFT) RECOMMENDATIONS FOR THE CONTROL OF MULTI-DRUG RESISTANT GRAM-NEGATIVES: CARBAPENEM RESISTANT ENTEROBACTERIACEAE John Ferguson (Hunter New England, NSW) on behalf of MRGN Task Force Acknowledgement
Antibiotic Susceptibility Pattern of Vibrio cholerae Causing Diarrohea Outbreaks in Bidar, North Karnataka, India
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 4 Number 9 (2015) pp. 957-961 http://www.ijcmas.com Original Research Article Antibiotic Susceptibility Pattern
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 4 Number 9 (2015) pp. 957-961 http://www.ijcmas.com Original Research Article Antibiotic Susceptibility Pattern
Brief Report THE DEVELOPMENT OF VANCOMYCIN RESISTANCE IN A PATIENT WITH METHICILLIN-RESISTANT STAPHYLOCOCCUS AUREUS INFECTION
 Brief Report THE DEVELOPMENT OF VANCOMYCIN RESISTANCE IN A PATIENT WITH METHICILLIN-RESISTANT STAPHYLOCOCCUS AUREUS INFECTION KRZYSZTOF SIERADZKI, PH.D., RICHARD B. ROBERTS, M.D., STUART W. HABER, M.D.,
Brief Report THE DEVELOPMENT OF VANCOMYCIN RESISTANCE IN A PATIENT WITH METHICILLIN-RESISTANT STAPHYLOCOCCUS AUREUS INFECTION KRZYSZTOF SIERADZKI, PH.D., RICHARD B. ROBERTS, M.D., STUART W. HABER, M.D.,
HEALTHCARE-ACQUIRED INFECTIONS AND ANTIMICROBIAL RESISTANCE
 Universidade de São Paulo Departamento de Moléstias Infecciosas e Parasitárias HEALTHCARE-ACQUIRED INFECTIONS AND ANTIMICROBIAL RESISTANCE Anna S. Levin 4 main lines! Epidemiology of HAS and resistance!
Universidade de São Paulo Departamento de Moléstias Infecciosas e Parasitárias HEALTHCARE-ACQUIRED INFECTIONS AND ANTIMICROBIAL RESISTANCE Anna S. Levin 4 main lines! Epidemiology of HAS and resistance!
ARCH-Vet. Summary 2013
 Federal Department of Home Affairs FDHA FSVO ARCH-Vet Report on sales of antibiotics in veterinary medicine and antibiotic resistance monitoring of livestock in Switzerland Summary 2013 Published by Federal
Federal Department of Home Affairs FDHA FSVO ARCH-Vet Report on sales of antibiotics in veterinary medicine and antibiotic resistance monitoring of livestock in Switzerland Summary 2013 Published by Federal
RESEARCH NOTE THE EVALUATION OF ANTIMICROBIAL SUSCEPTIBILITY OF URINE ENTEROCOCCI WITH THE VITEK 2 AUTOMATED SYSTEM IN EASTERN TURKEY
 Southeast Asian J Trop Med Public Health RESEARCH NOTE THE EVALUATION OF ANTIMICROBIAL SUSCEPTIBILITY OF URINE ENTEROCOCCI WITH THE VITEK 2 AUTOMATED SYSTEM IN EASTERN TURKEY Sibel AK 1, Köroglu Mehmet
Southeast Asian J Trop Med Public Health RESEARCH NOTE THE EVALUATION OF ANTIMICROBIAL SUSCEPTIBILITY OF URINE ENTEROCOCCI WITH THE VITEK 2 AUTOMATED SYSTEM IN EASTERN TURKEY Sibel AK 1, Köroglu Mehmet
MRSA ST398 from swine and cattle
 Novel antimicrobial resistance genes among livestock-associated MRSA ST398 from swine and cattle Kristina Kadlec, Andrea Feßler and Stefan Schwarz Institute of Farm Animal Genetics,, Friedrich-Loeffler
Novel antimicrobial resistance genes among livestock-associated MRSA ST398 from swine and cattle Kristina Kadlec, Andrea Feßler and Stefan Schwarz Institute of Farm Animal Genetics,, Friedrich-Loeffler
Multidrug-Resistant Salmonella enterica in the Democratic Republic of the Congo (DRC)
 Multidrug-Resistant Salmonella enterica in the Democratic Republic of the Congo (DRC) Octavie Lunguya 1, Veerle Lejon 2, Sophie Bertrand 3, Raymond Vanhoof 3, Jan Verhaegen 4, Anthony M. Smith 5, Benedikt
Multidrug-Resistant Salmonella enterica in the Democratic Republic of the Congo (DRC) Octavie Lunguya 1, Veerle Lejon 2, Sophie Bertrand 3, Raymond Vanhoof 3, Jan Verhaegen 4, Anthony M. Smith 5, Benedikt
Project Summary. Principal Investigators: Ross Beier 1, T. Poole 1, Dayna Harhay 2, and Robin Anderson 1 1
 Project Summary Antibiotic and Disinfectant Susceptibility Profiles of Escherichia coli O157:H7 Cattle Feces, Hide, Carcass, and Ground Meat Isolates from the United States Principal Investigators: Ross
Project Summary Antibiotic and Disinfectant Susceptibility Profiles of Escherichia coli O157:H7 Cattle Feces, Hide, Carcass, and Ground Meat Isolates from the United States Principal Investigators: Ross
Antimicrobial susceptibility testing of Campylobacter jejuni and C. coli. CRL Training course in AST Copenhagen, Denmark 23-27th Feb.
 Antimicrobial susceptibility testing of Campylobacter jejuni and C. coli CRL Training course in AST Copenhagen, Denmark 23-27th Feb. 2009 Methodologies E-test by AB-biodisk A dilution test based on the
Antimicrobial susceptibility testing of Campylobacter jejuni and C. coli CRL Training course in AST Copenhagen, Denmark 23-27th Feb. 2009 Methodologies E-test by AB-biodisk A dilution test based on the
Intrinsic, implied and default resistance
 Appendix A Intrinsic, implied and default resistance Magiorakos et al. [1] and CLSI [2] are our primary sources of information on intrinsic resistance. Sanford et al. [3] and Gilbert et al. [4] have been
Appendix A Intrinsic, implied and default resistance Magiorakos et al. [1] and CLSI [2] are our primary sources of information on intrinsic resistance. Sanford et al. [3] and Gilbert et al. [4] have been
Antibiotics in vitro : Which properties do we need to consider for optimizing our therapeutic choice?
 Antibiotics in vitro : Which properties do we need to consider for optimizing our therapeutic choice? With the support of Wallonie-Bruxelles-International 1-1 In vitro evaluation of antibiotics : the antibiogram
Antibiotics in vitro : Which properties do we need to consider for optimizing our therapeutic choice? With the support of Wallonie-Bruxelles-International 1-1 In vitro evaluation of antibiotics : the antibiogram
Phenotypic & genotypic characterization of vancomycin resistant Enterococcus isolates from clinical specimens
 Indian J Med Res 138, October 2013, pp 549-556 Phenotypic & genotypic characterization of vancomycin resistant Enterococcus isolates from clinical specimens Ira Praharaj, S. Sujatha & Subhash Chandra Parija
Indian J Med Res 138, October 2013, pp 549-556 Phenotypic & genotypic characterization of vancomycin resistant Enterococcus isolates from clinical specimens Ira Praharaj, S. Sujatha & Subhash Chandra Parija
Identification of vancomycin-resistant enterococci clones and inter-hospital spread during an outbreak in Taiwan
 Lee et al. BMC Infectious Diseases 2013, 13:163 RESEARCH ARTICLE Open Access Identification of vancomycin-resistant enterococci clones and inter-hospital spread during an outbreak in Taiwan Sai-Cheong
Lee et al. BMC Infectious Diseases 2013, 13:163 RESEARCH ARTICLE Open Access Identification of vancomycin-resistant enterococci clones and inter-hospital spread during an outbreak in Taiwan Sai-Cheong
Summary of the latest data on antibiotic resistance in the European Union
 Summary of the latest data on antibiotic resistance in the European Union EARS-Net surveillance data November 2017 For most bacteria reported to the European Antimicrobial Resistance Surveillance Network
Summary of the latest data on antibiotic resistance in the European Union EARS-Net surveillance data November 2017 For most bacteria reported to the European Antimicrobial Resistance Surveillance Network
Antimicrobial use in poultry: Emerging public health problem
 Antimicrobial use in poultry: Emerging public health problem Eric S. Mitema, BVM, MS, PhD CPD- Diagnosis and Treatment of Poultry Diseases FVM, CAVS, 6 th. August, 2014 AMR cont Antibiotics - Natural or
Antimicrobial use in poultry: Emerging public health problem Eric S. Mitema, BVM, MS, PhD CPD- Diagnosis and Treatment of Poultry Diseases FVM, CAVS, 6 th. August, 2014 AMR cont Antibiotics - Natural or
A retrospective analysis of urine culture results issued by the microbiology department, Teaching Hospital, Karapitiya
 A retrospective analysis of urine culture results issued by the microbiology department, Teaching Hospital, Karapitiya LU Edirisinghe 1, D Vidanagama 2 1 Senior Registrar in Medicine, 2 Consultant Microbiologist,
A retrospective analysis of urine culture results issued by the microbiology department, Teaching Hospital, Karapitiya LU Edirisinghe 1, D Vidanagama 2 1 Senior Registrar in Medicine, 2 Consultant Microbiologist,
Research Article Genotyping of Methicillin Resistant Staphylococcus aureus Strains Isolated from Hospitalized Children
 International Pediatrics, Article ID 314316, 4 pages http://dx.doi.org/10.1155/2014/314316 Research Article Genotyping of Methicillin Resistant Staphylococcus aureus Strains Isolated from Hospitalized
International Pediatrics, Article ID 314316, 4 pages http://dx.doi.org/10.1155/2014/314316 Research Article Genotyping of Methicillin Resistant Staphylococcus aureus Strains Isolated from Hospitalized
