Consumption of beef tongue: Human BSE risk associated with exposure to lymphoid tissue in bovine tongue in consideration of new research findings 1
|
|
|
- Solomon Dorsey
- 5 years ago
- Views:
Transcription
1 The EFSA Journal (2008) 700 Consumption of beef tongue: Human BSE risk associated with exposure to lymphoid tissue in bovine tongue in consideration of new research findings 1 Scientific Opinion of the Panel on Biological Hazards (Question No EFSA-Q ) Adopted on 17 April 2008 DISCLAIMER This document represents revision 1 of the document originally published in EFSA website on 29/04/2008, as it has been realized that a mistake was included in the document originally published. In this revision, the original sentence Buschmann and Groschup (2005) provide data on a Tgbov mice model giving a fold greater sensitivity than the cattle bioassay. at lines 6-8 of page 12 of the document has been replaced by the sentence Buschmann and Groschup (2005) provide data on a Tgbov mice model giving a 10 fold greater sensitivity than the cattle intra-cerebral bioassay. The mistake identified did not have any impact neither in any conclusion drawn from the text nor on the final outcome of the Opinion. A series of calculations on BSE infectivity in tonsil tissue were performed in this Opinion, but all of them took into account the correct figure and not the wrong one, so that all results were correct and correctly reported. 1 For citation purposes: Scientific Opinion of the Panel on Biological Hazards on a request from the European Commission on consumption of beef tongue: human BSE risk associated with exposure to lymphoid tissue in bovine tongue in consideration of new research findings. The EFSA Journal (2008) 700, 1-24 European Food Safety Authority, 2008
2 The EFSA Journal (2008) 700, 1-24 Consumption of beef tongue: Human BSE risk associated with exposure to lymphoid tissue in bovine tongue in consideration of new research findings 1 PANEL MEMBERS Scientific Opinion of the Panel on Biological Hazards (Question No EFSA-Q ) Adopted on 17 April 2008 Olivier Andreoletti, Herbert Budka, Sava Buncic, Pierre Colin, John D Collins, Aline De Koeijer, John Griffin, Arie Havelaar, James Hope, Günter Klein, Hilde Kruse, Simone Magnino, Antonio Martínez López, James McLauchlin, Christophe Nguyen-The, Karsten Noeckler, Birgit Noerrung, Miguel Prieto Maradona, Terence Roberts, Ivar Vågsholm, Emmanuel Vanopdenbosch. SUMMARY Following a request from the European Commission, the Panel on Biological Hazards (BIOHAZ) was asked to deliver a scientific opinion on the human BSE risk associated with exposure to lymphoid tissue in bovine tongue in consideration of the findings included in a scientific article recently published on the consumption of beef tongue and the risk for public health. This scientific article describes the distribution of lymphoid tissue in bovine tongue and the location of bovine lingual tonsil. In addition, it concludes that the method currently prescribed for harvesting bovine tongues in slaughterhouses is not appropriate for removing all specified risk material (SRM) and proposes an alternative harvesting method. EFSA was requested (i) to evaluate the design of the study and its scientific validity in relation to the distribution of lymphoid tissue in bovine tongue and (ii) to evaluate the conclusions and recommendations of the study in relation to BSE risk from bovine tonsil following the harvesting method currently prescribed by EU legislation compared to the alternative one proposed in the study. The BIOHAZ Panel reviewed the scientific article and concluded that the study further confirms and extends observations that the lingual tonsil at the base of the tongue may not be entirely eliminated when harvesting tongues by means of the method currently prescribed. 1 For citation purposes: Scientific Opinion of the Panel on Biological Hazards on a request from the European Commission on consumption of beef tongue: human BSE risk associated with exposure to lymphoid tissue in bovine tongue in consideration of new research findings. The EFSA Journal (2008) 700, 1-24 European Food Safety Authority, 2008
3 Consumption of beef tongue In reply to the second request, the BIOHAZ Panel assessed different parameters in order to quantify the human exposure risk to BSE from bovine tonsil associated with the consumption of bovine tongue. It was concluded that, overall, the level of infectivity in bovine tonsil is low. This, together with the declining and overall low BSE prevalence and the current policy on SRM removal, suggests a very low, if not negligible, human BSE exposure risk associated with exposure to lymphoid tissue in bovine tongue harvested as currently prescribed by EU legislation. The BIOHAZ Panel further concluded that currently there are not sufficient quantitative data available allowing a comparison of the human BSE exposure risk reduction achieved by the alternative tongue harvesting method proposed by the study in comparison to the harvesting method currently prescribed. However, it is likely that the proposed method would only provide a marginal reduction in the risk from bovine tonsil compared to the one currently prescribed. Following to this, the BIOHAZ Panel made a series of recommendations on the topics that might be addressed in future studies on the subject. Key words: BSE, beef tongue, tonsil, lymphoid tissue. The EFSA Journal (2008) 700, 2-24
4 Consumption of beef tongue TABLE OF CONTENTS Panel Members...1 Summary...1 Table of Contents...3 Background as provided by the European Commission...4 Terms of reference as provided by the European Commission...4 Acknowledgements...4 Assessment Introduction Regulatory background Terminology Former SSC and EFSA assessments Review of the design of the study by Casteleyn et al. (2007) and of its scientific validity Risk assessment Level of infectivity associated with bovine tonsils Amount of lymphoid tissue associated with the tongue intended for human consumption Dynamics and prevalence of BSE in the EU cattle population BSE risk from bovine tonsil following different harvesting methods of bovine tongue Conclusions and Recommendations...16 Recommendations...17 Documentation provided to EFSA...17 References...18 Appendices...21 The EFSA Journal (2008) 700, 3-24
5 Consumption of beef tongue BACKGROUND AS PROVIDED BY THE EUROPEAN COMMISSION Regulation (EC) No 999/2001 of the European Parliament and of the Council lays down rules for the prevention, control and eradication of certain transmissible spongiform encephalopathies, including the provisions related to the removal of specified risk material. Point 1 of the Annex XI 2 to Regulation (EC) No 999/2001 lays down that tonsils of bovine animals of all ages are designated as specified risk material. Point 6 lays down special provisions when harvesting the bovine tongue at the slaughterhouse, i.e. Tongues of bovine animals of all ages intended for human or animal consumption shall be harvested at the slaughterhouse by a transverse cut rostral to the lingual process of the basihyoid bone. Recently, a scientific article on Consumption of beef tongue and sweetbread: risk for public health? has been published in the journal Vlaams Diergeneeskundig Tijdschrift 3. The paper describes the exact anatomical location of the bovine lingual tonsil and demonstrates that the currently prescribed technique to harvest bovine tongues at the slaughterhouse is not appropriate to remove all SRM. TERMS OF REFERENCE AS PROVIDED BY THE EUROPEAN COMMISSION The European Food Safety Authority (EFSA) is invited to provide a scientific Opinion on: 1. the design of the study and its scientific validity, in particular with reference to the distribution of lymphoid tissue in the tongue of bovine animals; 2. the conclusions and recommendations of the study in respect to current and future BSE risk from bovine tonsil following the harvesting method of bovine tongues at the level of the slaughterhouse as currently set out by Regulation (EC) No 999/2001 compared to the harvesting method proposed in the study. Clarification of the ToR The scientific article submitted to EFSA for assessment includes a part referring specifically to bovine thymus. It was clarified with the EU Commission Services that the scientific opinion was requested only with respect to the bovine tongue related aspects of the study and not with respect to its considerations on bovine thymus. In addition, and after consultation with the EU Commission Services, the ToR were amended in order to limit the scope of this assessment to the human BSE risk associated with exposure to lymphoid tissue in bovine tongue. ACKNOWLEDGEMENTS The European Food Safety Authority wishes to thank the members of the Working Group for the preparation of this opinion: Olivier Andreoletti, John Griffin (Chairman), Martin Groschup, Günter Klein, Emmanuel Vanopdenbosch and Gerald Wells (Rapporteur). The BIOHAZ panel members would like to acknowledge Dr. Christophe Casteleyn for his availability when clarifying certain aspects related to the findings of the research that he and his colleagues carried out. 2 3 Annex XI of Regulation (EC) No 999/2001 was deleted by Regulation (EC) No 722/2007 (OJ L 164, , p.7) and the list of SRM was moved, together with other provisions, into Annex V of the Regulation, as described in Section 1.1 of this Opinion. Casteleyn, C., Breugelmans, S., Muylle, S., Van den Broeck, W. and Simoens, P. (2007) Consumptie van rundertong en kalfszwezeriken: een risico voor de volksgezondheid? Vlaams Diergeneeskundig Tijdschrift, 76, The EFSA Journal (2008) 700, 4-24
6 Consumption of beef tongue ASSESSMENT 1. Introduction 1.1. Regulatory background Point 1 of Annex V to Regulation (EC) No 999/ defines, also depending on the country or region of origin of the animals, the following bovine tissues as specified risk material (SRM): The following tissues shall be designated as specified risk material if they come from animals whose origin is in a Member State or third country or of one of their region with a controlled or undetermined BSE risk: (a) as regards bovine animals: i) the skull excluding the mandible and including the brain and eyes, and the spinal cord of animals aged over 12 months; ii) the vertebral column excluding the vertebrae of the tail, the spinous and transverse process of the cervical, thoracic and lumbar vertebrae and the medial sacral crest and wings of the sacrum, but including the dorsal root ganglia of animals aged over 24 months 5 ; and iii) the tonsils, the intestines from the duodenum to the rectum and the mesentery of animals of all ages.. Point 7 of Annex V to Regulation (EC) No 999/2001 lays down provisions when harvesting the bovine tongue at the slaughterhouse: Tongues of bovine animals of all ages intended for human or animal consumption shall be harvested at the slaughterhouse by a transverse cut rostral to the lingual process of the basihyoid bone Terminology Tonsils are not specifically defined in Regulation (EC) No 999/2001. Nickel et al. (1979) provide a definition of bovine tonsils, as reported below. The tonsils are the independent lymphatic organs of the pharynx. They are composed of accumulations of lymphatic tissue, that may be temporary, diffuse and unorganised, or permanent lymph nodules, appearing solitary or organised into small or large aggregates. In the bovine the tonsils form six named parts according to their location: lingual, palatine, soft palate, paraepiglottic, pharyngeal and tubal (see Figures 1 and 2). The definition of the tonsils has three important aspects in the context of this Opinion, which concerns principally the lingual tonsil: 4 5 OJ L 147, , p.1. Regulation as last amended by Regulation (EC) No 315/2008 (OJ L 94, , p.3). After the adoption of this Opinion, and before its publication, Regulation (EC) No 357/2008 (OJ L 111, , p.3) was published, increasing this age to 30 months. The EFSA Journal (2008) 700, 5-24
7 Consumption of beef tongue a) Recognition of tonsil tissue from visual inspection of the pharynx can be made only by features on the surface of the mucosa. In the bovine the main concentrations of tonsillar lymphatic tissue are the palatine tonsil and lingual tonsil: The palatine tonsil is the largest of the tonsillar lymphoid aggregations and in adult cattle comprises a discrete 3 cm diameter structure embedded in the lateral wall of the oropharnyx. It is evident from the pharyngeal mucosa by a single opening, the tonsillar sinus. The lingual tonsil is evident from the dorsal surface of the base of the tongue as numerous distinct fossulae (invaginations or crypts) which radiate in curvi-linear rows, rostro-laterally. The most rostral limit of these fossulae relative to other mucosal features is subject to some biological variation between animals (Wells et al., 2005). b) Diffuse and unorganised lymphoid accumulations which, by definition, occur in tonsils in addition to organised aggregations of lymphoid nodules, are not visible on inspection of the mucosa as they are covered only by a smooth epithelial layer. In the bovine lingual tonsil, lymphoid tissue (in the form of lymphoid nodules and diffuse lymphoid accumulations not associated with fossulae) extends rostral to the visible array of fossulae (Wells et al., 2005; Kuhne et al., 2005; Cocquyt et al., 2007; Casteleyn et al., 2007). c) Diffuse and unorganised lymphoid tissue that is temporary or transient cannot be distinguished from inflammatory infiltrations, the extent of which will be variable, depending upon many physiological factors, including the age of the animal, diet and management and possible specific disease related factors. In summary, the lingual tonsil can be defined by visual inspection to identify the area of the mucosa in which fossulae are evident, but lymphoid tissue of the tongue extends rostral to this area and may vary in extent depending on physiological variables and the possible occurrence of inflammatory changes. The EFSA Journal (2008) 700, 6-24
8 Consumption of beef tongue Palatine tonsil Lingual tonsil Root of tongue Vallate papillae Torus linguae Figure 1. Bovine tongue (adapted from Nickel/Schummer/Seiferle: Lehrbuch der Anatomie der Haustiere, Bd. II Eingeweide, 9/e, Parey in MVS Medizinverlage Stuttgart GmbH & Co. KG, 2004). The EFSA Journal (2008) 700, 7-24
9 Consumption of beef tongue l n Palatine tonsil Torus linguae Root of tongue Figure 2. Bovine tongue (adapted from Nickel/Schummer/Seiferle: Lehrbuch der Anatomie der Haustiere, Bd. II Eingeweide, 9/e, Parey in MVS Medizinverlage Stuttgart GmbH & Co. KG, 2004) Former SSC and EFSA assessments In its Opinion on Transmissible Spongiform Encephalopathies (TSEs) infectivity distribution in ruminant tissues (SSC, 2002), the Scientific Steering Committee (SSC) concluded that the tonsil of a bovine animal of any age should be regarded as posing a risk and that the tongue of animals certified safe for human consumption does indeed not pose a risk if contamination with CNS and tonsil material is avoided for animals of any age, but this would imply that the harvested section of the tongue is shortened [to the short tongue ] to avoid, by a cautious margin, removal with the tongue of that part of the root of the tongue containing lingual tonsil. In 2004, EFSA was requested to consider updating the mentioned SSC Opinion with regard to the Bovine Spongiform Encephalopathy (BSE) risk from bovine tonsil. Considering the already established precautionary measures, the very low tonsil infectivity titre and the absence of new positive findings in tonsil infectivity from the ongoing cattle bioassay study, EFSA concluded The EFSA Journal (2008) 700, 8-24
10 Consumption of beef tongue that the re-assessment of tonsil infectivity did not suggest the need for updating the SSC Opinion of TSE infectivity in ruminant tissues (EFSA, 2004). In addition, EFSA also concluded that in the case of the detection of PrP Sc in tongues following tongue or intra-cerebral infection as recently observed in one specific animal model a quantitative risk analysis appears currently not to be possible in view of the total absence of data for other strains and species. The study cited (Bartz et al., 2003) reported the detection of PrP Sc in tongue tissue using the Western Blot method following the intracerebral inoculation of hamsters with hamster adapted strains of Transmissible Mink Encephalopathy (HY TME) agent. The authors suggested the likely route of infection of the tongue was axonal transport of PrP Sc from the brain to the tongue. Bartz et al. (2003) also inoculated the TME agent into the tongue of hamsters and showed that this was 100,000-fold more efficient than infection by oral route. In 2005, EFSA was requested to evaluate the conclusions of an OIE-consultation group of experts as well as other scientific information available and, if appropriate, to review the age limit for the removal of certain bovine tissues identified as SRM. With regard to bovine tonsils, EFSA concluded that there was no scientific basis to raise the age limit for removal of tonsil (EFSA, 2005). 2. Review of the design of the study by Casteleyn et al. (2007) and of its scientific validity. The scientific publication Consumptie van rundertong en kalfszwezeriken: een risico voor de volksgezondheid? 6 (Casteleyn et al., 2007) describes the anatomical location of the bovine lingual tonsil based on the examination of a sample of 15 tongues from adult cattle immediately after slaughter. Semi-serial haematoxylin and eosin stained histological sections were prepared from full width slices of the mucosa of the tongue, extending from the most caudal vallate papilla to the epiglottis (10 tongues) and from the most caudal vallate papilla to 5 cm rostrally (4 tongues). A histological section for the identification of fossulae was also prepared from 1 tongue. Visual macroscopic examination of the tongues showed the rostral termination of lingual tonsil fossulae being located approximately 2 cm caudal to the most caudal vallate papilla. In keeping with the anatomical textbook definition of tonsil, these authors also observed by histological examination that subepithelial lymphoid follicles (nodules) occur without associated fossulae and are distributed between the fossulae. Lymphocyte infiltration of the epithelium, consistent with inflammatory changes, was also noted in 2 out of 15 tongues. In the area of tongue 3cm rostral to and 2cm caudal to the most caudal vallate papilla only a few primary follicles and diffuse accumulations of lymphocytes were detected. Rostral to this none were found. As described under section 1.2, these lymphoid accumulations are considered part of normal tonsillar tissue but could also be inflammatory in origin. The article states that the lingual tonsil is not confined to the area where crypt openings are found. We have in fact been able to demonstrate lymphocyte aggregations that were not associated with a crypt. These findings are consistent with those of other studies carried out in Great Britain (Wells et al., 2005) and Germany (Rebmann et al., submitted for publication). Casteleyn et al., (2007) conclude that certain parts of bovine tongue currently intended for human consumption could represent a risk of exposure to BSE for consumers and propose an alternative cutting method for harvesting bovine tongue at slaughter. 6 Consumption of beef tongue and sweet bread: risk for public health? The EFSA Journal (2008) 700, 9-24
11 The study findings present the following interpretations: Consumption of beef tongue The sample size comprised tongues from only 15 animals, and only tongues from 4 animals were examined rostral to the most caudal vallate papilla. The significance of a study with such a low number of samples may be questionable, in view of the intrinsic variability of the distribution of lymphoid formations in the bovine tongue. For example, the precision of the position of the most rostral fossulae as being 2cm caudal to the most caudal vallate papilla, may not be justified based on this sample size and in view of the anatomical variation found previously in the position of the most rostral fossulae in a sample of 100 tongues (Wells et al., 2005). Despite this limitation, the histological examination of the bovine tongue is extended more rostrally than the study of Wells et al. (2005) and the findings concur with the observations of other studies (Kuhne et al., 2005; Cocquyt et al., 2007; Rebmann et al., submitted for publication) that lymphoid tissue, albeit relatively sparse, may be present in bovine tongues rostral to the level of the most caudal vallate papilla. The more rostral part of the tongue, which might harbour part of the lingual tonsil, is not taken into account. The study provides no quantitative elements of the relative amounts of lymphoid tissue in the different areas of tongue examined. The study does not provide a quantitative comparison of the amount of lymphoid tissue that would be removed from bovine tongue by implementing the proposed alternative harvesting method with the amount removed by the currently applied harvesting method. A conclusion is drawn that incomplete removal of lymphoid tissue from the tongue poses a limited risk to public health, but no other risk factors, particularly in relation to BSE infectivity in bovine tonsillar tissue or BSE prevalence in EU Member States, are considered. 3. Risk assessment In this assessment it is considered that three main parameters have to be evaluated in order to quantify the human exposure risk to BSE from bovine tonsil associated with the consumption of bovine tongue: a. the level of infectivity associated with bovine tonsils; b. the amount of lymphoid tissue associated with the tongue intended for human consumption; c. the dynamics and prevalence of BSE in the EU cattle population Level of infectivity associated with bovine tonsils The EFSA Opinion of 2004 (EFSA, 2004) summarised as follows the findings reported in an assessment carried out by DNV (SEAC, 2003). The bovine intra-cerebral infectivity dose of tonsil tissue was estimated to be not higher than 10 0 to 10 1 ID 50 (DNV report; Prof. Wells, personal communication). Considering a decrease of effectiveness of infection in the oral route by a factor of (at least) 10-5 this would equal a bovine oral infectivity of 10-5 to 10-4 ID 50. Thus a whole tonsil of 50 g would carry no more than The EFSA Journal (2008) 700, 10-24
12 Consumption of beef tongue boid 50. In the DNV report, where a more tentative approach was used (resulting in a factor of for the decrease of infectivity from intra-cerebral to oral route), infectivity for one whole bovine tonsil (50 g) was calculated to be 0.25 boid 50. Additional facts have to be taken into account as sources of variation in risk quantification, such as reduction of tonsil tissue due to meat inspection, current regulations and slaughtering practices, and in particular the fact that the one positive finding relates to the palatine tonsil and not lingual tonsil tissue. Since the publication of this opinion new data have become available. However, the number of BSE infected cattle in which infectivity in tonsillar tissue has been investigated is very limited Natural BSE clinical cases. Studies of infectivity in the palatine tonsils of naturally occurring clinical cases of BSE have been conducted on only two cows: one in Great Britain, using RIII mice as the assay model (Fraser and Foster, 1994) and one in Germany (Buschmann and Groschup, 2005) using both RIII and Tgbov XV mice for the assays. In both of these studies mice were inoculated (0,02ml intracerebrally and 0,1ml intraperitoneally) with ten per cent tissue homogenates in saline. No infectivity was detected even by the Tg mouse assay which provides more than 4 logs greater sensitivity of detection than the conventional wild type mouse assay. In a study in Japan, three dairy cattle, infected naturally with BSE, did not show PrP Sc in the palatine tonsils using WB and IHC (Iwata et al., 2006). Due to the low number of cases investigated, no definite conclusions can be drawn Experimental BSE cases after oral challenge. In a sequential kill pathogenesis study of BSE in which calves were experimentally infected by oral exposure to 100g of a pool of BSE affected brainstems (a dose considered far in excess of most natural exposures (Arnold et al., 2007)), palatine tonsil was assayed in conventional mouse strains from all time points (2-40 months post-exposure). No infectivity was detected (Wells et al., 1998; Wells et al., 2005). Palatine tonsil from this sequential kill pathogenesis study was further assayed by intracerebral inoculation of cattle, which provides a 500 fold (log ) greater sensitivity of detection of the BSE agent than the RIII mouse assay (Wells et al., 2005). A pooled inoculum was prepared from palatine tonsil of cattle (3, 3, 3 and 1 respectively) at each of 6, 10, 18, and 26 month periods post-exposure (corresponding to 10, 14, 22 and 30 months of age) from the oral challenge study. One ml of a ten per cent tissue homogenate in saline was injected by the intracerebral route into groups of 5 calves. Results indicated traces of infectivity in the palatine tonsil of cattle killed ten months after experimental oral exposure, with transmission occurring in 1 of the 5 challenged calves (Wells et al., 2005). An infection rate of 1 out of 5 suggests that the infectivity is close to the limit of detection of the assay and that the titre of infectivity in tonsil is less than 1 cattle i.c. ID 50 /g. The study was completed in 2006, without further transmission to any of the 4 remaining calves (Veterinary Laboratory Agency, unpublished data). Palatine tonsil collected from a further sequential-kill pathogenesis study of BSE in which 100 calves were exposed to 100g of a pool of BSE affected brainstems (Arnold et al 2007) was also bioassayed. In this study, Espinosa et al. (2007) inoculated tonsil tissue intracerebrally into mice expressing bovine PrP (BoPrP-Tg110). The inocula originated from The EFSA Journal (2008) 700, 11-24
13 Consumption of beef tongue pooled samples from three cattle at each of five time points (20, 24, 27, 30 and 33 months) after the oral exposure of calves. Infectivity was detected at all of the time points tested, with infection rates in the mice of 1/6, 1/6, 1/5, 1/6 and 1/6 respectively. Interestingly, this would seem to indicate a relatively low constant plateau level of infectivity in the tonsil throughout this largely preclinical period. The low infection rate (1 out of 6) is consistent with a level of infectivity lower than 1 i.c.id 50 in Tgbov mice. Buschmann and Groschup (2005) provide data on a Tgbov mice model giving a 10 fold greater sensitivity than the cattle intra-cerebral bioassay. While the Tgbov mice (tg110) in the study of Espinosa et al. (2007) were different from the TgbovXV mice used by Buschmann and Groschup (2005), their sensitivity is likely to be similar based on comparable levels of over expression of the bovine PrP gene in each model. From data provided by titrations of BSE infectivity in brain by the i.c. and the oral routes in cattle it has been calculated that one bovine oral ID 50 = bovine i.c. ID 50 (Wells et al., 2007). On this basis, the titre of infectivity in tonsil is less than bovine oral ID 50 /g; an estimate, using additional data, that is closely similar to that provided previously (EFSA 2004). Given the greater sensitivity of the Tgbov mouse assays than the cattle i.c. assay, it would seem probable that even in cattle after exposure to a 100g oral dose, the titre in terms of bovine oral ID 50 /g may be at least one order of magnitude less (i.e bovine oral ID 50 /g), for at least part of the incubation period. As the majority of exposures in the epidemic were probably less than 1g (Arnold et al., 2007), the infectivity in tonsil might well have occurred at an even lower titre and have peaked later in incubation. The most optimistic estimate might be that with doses of the order of less than 1g, tonsil never contains detectable levels at any time in the disease course. A table summarising the key parameters of the infectivity studies mentioned in section 3.1 can be found in Appendix A. In summary, the available data on BSE infectivity in tonsil indicate that: The frequency with which detectable infectivity occurs in tonsillar tissue of a BSE naturally infected animal is difficult to estimate. Although only two cases have been investigated employing biological assay, infectivity has not been detected in palatine tonsil from naturally occurring clinical cases. From sequential kill pathogenesis studies in experimentally infected cattle, infectivity associated with tonsil could be detected at 10 months post-exposure and, in the preclinical period, months post-exposure (Wells et al., 2005; Espinosa et al., 2007), suggesting that, in this model, infectivity in tonsil probably persists throughout the disease course. In the experimental exposure to a 100g dose, infectivity titer can be estimated from the data available to be bovine oral ID 50 per gram of tonsilar lymphoid tissue. It must also be noted that all available data on infectivity relate to palatine tonsil. Occurrence and comparable levels of infectivity in lingual tonsil is an assumption. The EFSA Journal (2008) 700, 12-24
14 Consumption of beef tongue 3.2. Amount of lymphoid tissue associated with the tongue intended for human consumption As mentioned above, the findings of the study from Casteleyn et al. (2007) are consistent with those of other studies carried out in Great Britain (Wells et al., 2005) and Germany (Rebmann et al., submitted for publication), but provide no quantification of lymphoid tissue remaining in tongues intended for human consumption. Examination of 251 tongues, derived from 15 abattoirs in Great Britain after removal of SRM and intended for human consumption, showed that visible identifiable lingual tonsillar tissue, indicated by fossulae, remained in 76.5% of them (192 out of 251) (Wells et al., 2005). Even in the tongues in which no visible tonsillar tissue remained, histological examination revealed lymphoid tissue in more than 90% of them. Variations in the distribution of the lingual tonsil suggested that even after the most rigorous trimming of the tongue traces of tonsillar tissue may remain. However, the histological examination did not extend rostral to the most caudal of the filiform papillae, which occur caudal to the most caudal vallate papillae. In Germany (Rebmann et al., submitted for publication), specimens of the lingual mucosa were taken from thirty cattle immediately after slaughter. The main parameter to identify the lymphoreticular tissue in this study was the immunohistochemical identification of the follicular dendritic cells (FDC). Lymph follicles were detected in areas up to 30 mm rostral to a given macroscopic landmark (i.e. the most caudal of the vallate papilla). This is an area which would not be removed from the tongue when implementing the measures foreseen by Regulation (EC) No 999/2001. Alternative technical approaches for the removal of the lingual tonsil s tissue, similar to those by Casteleyn et al. in 2007, are proposed by the authors. These scientific studies did not take into account the rostral part of the tongue which can harbor solitary primary lymph nodules or diffuse accumulations of T and B lymphocytes. Some information on more rostral areas of the tongue was provided in a more recent study (Kato and Sawada, 2008). In that study, examination of the bovine tongue was carried out from the tip of the torus linguae to the root of the tongue. The study confirmed previous results in relation to the distribution of lymphoid tissue in bovine tongue. In addition, 1 out of the 20 specimens collected rostrally to the most rostral vallate papilla contained lingual tonsillar tissue. Based on the above data, neither qualitative nor quantitative estimation of the significance of the lymphoid formations located rostrally to the most caudal vallate papilla is feasible at present. In the DNV risk assessment cited in section 3.1 (EFSA, 2004; SEAC, 2003), a total weight of bovine tonsil of 50g is assumed. However, in the DNV risk assessment, it is stated that the weights of the various tissues were taken from the LFRA ruminant products audit (LFRA, 1997). The total weight of tonsillar tissue in a typical bovine, as given from literature derived offal weights in the LFRA audit, is estimated at 200g. This suggests that the value of 50g in the DNV report is in fact referring to the weight of lingual tonsil. If the largest contribution to the total weight of tonsillar material is the palatine tonsil, it would be reasonable to estimate lingual tonsil as approximately 50g. It is further assumed in the DNV risk assessment that, after removal of all visible tonsillar tissue, the realistic upper limit of tissue that would remain in the tongue would be 10% i.e. 5g. Since the palatine tonsil is a circumscribed structure and easily identified, complete removal, compared to the lingual tonsil, is ensured, so this estimate remains valid as the quantity of tonsillar tissue that might not be removed. In conclusion, based on the data available: The EFSA Journal (2008) 700, 13-24
15 Consumption of beef tongue it is not possible to know exactly the quantity of lymphoid tissue remaining in bovine tongue intended for human consumption when harvested according to the harvesting method currently applied, even if it can be estimated to be 5g; it is not possible to estimate how much lymphoid tissue is removed by the alternative harvesting method compared with the one currently applied Dynamics and prevalence of BSE in the EU cattle population The European Union (EU) has developed a comprehensive set of measures on TSEs in order to protect human health from BSE and to control and eventually eradicate TSEs in animals (Regulation (EC) No 999/2001 and related EU legislation). In addition to these control measures, extensive epidemiological surveillance of BSE 7 has been carried out in order to monitor the dynamics of the disease over time and check the efficacy of the control measures put in place. Over the last 6.5 years more than 67 million tests have been carried out in the EU. Detailed epidemiological information on BSE monitoring can be found in the TSE annual/monthly reports released by the EU Commission (EC, 2001; EC, 2002; EC, 2003; EC, 2004; EC, 2005; EC, 2006; EC, 2007). Some key figures on the prevalence of BSE in cattle and on the number of tests carried out in the EU since July 2001 are reported in Appendix B. A constant decline (about 30-40% per year) in the number of detected BSE positive cases has been recorded: from 2,167 cases in 2001 to 320 cases in In 2006, no BSE cases were found in Denmark, Estonia, Greece, Cyprus, Latvia, Lithuania, Luxemburg, Hungary, Malta, Slovakia, Finland, Bulgaria and Romania. So far, only 27 BSE cases are related to animals born after the implementation of the EU wide total feed ban in Altogether these data indicate a dramatic decrease of the number of BSE cases in Europe over the period. As concerns the average age of detection of BSE cases in the EU, the data available point out its constant increase through the years. For healthy slaughtered animals the average age of detection has increased from 76 months in 2001 to 116 months in This is in accordance with an overall decrease of BSE in cattle over the years (Saegerman et al., 2006). In the current context, human exposure to BSE would result from the entry into the food chain of undetected BSE incubating animals. This situation could arise if animals under 30 months of age (not tested), or animals over 30 months of age (testing negative) were incubating BSE when slaughtered for human consumption. Indeed, capacity of BSE tests to identify infected animals is limited to the late stage of the incubation period. Based on the first BSE pathogenesis studies realized in cattle after a 100g oral challenge, the likely detection of positive cases with rapid BSE tests was considered to be at about ¾ of the incubation time (EFSA 2007). Recently, a more realistic level of field exposure (1g) has been considered. In that situation, detection of cases by active surveillance is predicted by modelling to be only 1-2 months prior to the onset of clinical disease (Arnold et al., 2007). 7 Over the years data have been collected from the different EU Member States (in some cases also during pre-accession periods) starting at different time points. In particular, data are available since July 2001 from countries of the EU-15, since 2002 also from Bulgaria, since 2003 also from the other countries of the EU-27 except Romania, for which data are available since The EFSA Journal (2008) 700, 14-24
16 Consumption of beef tongue In this context, the efficacy of BSE testing to exclude the entry of BSE infected animals into the food chain appears limited. This underlines how prevention of human exposure to BSE agent mainly relies on the SRM removal policy. An estimation of the number of undetected BSE infected bovines entering into the food chain was not performed in the framework of this mandate. Such an estimation would assist in quantifying the residual BSE infectivity entering the food chain. In conclusion: The number of detected BSE cases continues to decline in EU with about 30-40% per year, which indicates a continuous decrease of BSE prevalence in the EU cattle population and consequently a reduction of food-borne human exposure risk to BSE agent. SRM removal and destruction remains the most effective measure to protect human health from the risk of exposure to BSE. 4. BSE risk from bovine tonsil following different harvesting methods of bovine tongue. As mentioned in section 1.1, EU legislation currently in force prescribes that a transverse cut rostral to the lingual process of the basihyoid bone is performed while harvesting bovine tongue intended for human consumption. Such a procedure was introduced by Regulation (EC) No 1139/2003 8, in order to ensure that bovine tonsils are excluded from human consumption and eliminated as SRM. There are no scientific research data available to judge to what extent this achieves the removal of the lingual tonsil from the bovine tongue. While the discrete structures formed by the palatine tonsil are easily identified, the limits of the lingual tonsil are far less definable. Visual inspection of the base of the tongue allows the identification of lingual tonsil only by the presence of fossulae. If it were considered insufficient to remove only the visually defined tonsil tissue, then the method of harvesting and trimming of the tongue for human consumption would be required to be considerably more prescriptive than provided by current EU legislation. As already reported under section 3.2, based on the DNV risk assessment (EFSA, 2004; SEAC, 2003) and the LFRA ruminant products audit (LFRA, 1997), a bovine lingual tonsil weight of 50g is assumed. It is further assumed in the DNV risk assessment that after removal of all visible tonsillar tissue, the realistic upper limit of tissue that would remain in the tongue would be 10% i.e. 5g. The assessment uses an upper limit of infectivity in tonsil of 10 1 bovine i.c. ID 50 /g, and a factor of for the decrease of infectivity from intra-cerebral to oral route giving the infectivity for the whole bovine lingual tonsil (50 g) as 0.25 bovine oral ID 50, or bovine oral ID 50. With the revised estimates for tonsil infectivity of less than 1 cattle i.c. ID50/g and the efficiency of the route of one bovine oral ID 50 = bovine i.c. ID 50 (Wells et al., 2007), 50g of tonsillar tissue would contain at most bovine oral ID50. If residual lymphoid tissue in tongue is considered to be 5g, then the greatest potential human exposure from the unlikely event of eating all of a bovine tongue would be bovine oral ID50. If 99% of tonsillar lymphoid tissue were removed this exposure would be reduced by a further factor of 10, to bovine oral ID50. 8 OJ L 160, , p.22. The EFSA Journal (2008) 700, 15-24
17 In conclusion: Consumption of beef tongue The available evidence indicates that the infectivity of bovine tonsil is low, but it is not possible to determine precisely the extent to which the proposed method would reduce the amount of infectivity remaining in the part of the tongue intended for human consumption compared with the harvesting method currently applied. However, in view of the overall low level of infectivity, it is likely that the proposed method would only provide a marginal reduction in the risk from bovine tonsil compared with the one currently applied. For example, if 10% of all bovine lingual tonsil material would remain with the current method and 1% would remain with the proposed method, the infective load would decrease from to bovine oral ID50. CONCLUSIONS AND RECOMMENDATIONS CONCLUSIONS ToR 1 on the design of the study and its scientific validity, in particular with reference to the distribution of lymphoid tissue in the tongue of bovine animals. In general, the study by Casteleyn et al. (2007) provides valuable insights into the anatomomorphological aspects of lymphoid tissue distribution in bovine tongue. However, the significance of the study from Casteleyn and colleagues with such a low number of samples analysed may be questionable, in view of the intrinsic variability of the distribution of lymphoid formations in the bovine tongue. The study has further confirmed and extended observations that the lingual tonsil at the base of the tongue may not be entirely eliminated by severing and removal at slaughter according to the implementation of measures as currently set out by Regulation (EC) No 999/2001. No quantitative elements are provided by the study, in particular in relation to the relative amounts of lymphoid tissue in the different areas of tongue examined and in relation to the comparison of the amount of lymphoid tissue that would be removed from bovine tongue by implementing the proposed alternative harvesting method with the amount removed by the harvesting method currently applied. As a limitation to this assessment, the study did not take into account the rostral part of the tongue which may also harbor part of the lingual tonsil. Therefore, both qualitative and quantitative estimation of the importance of these lymphoid formations by comparison with the area of the tongue caudal to vallate papillae is not feasible at present. ToR 2 on the conclusions and recommendations of the study in respect to current and future BSE risk from bovine tonsil following the harvesting method of bovine tongues at the level of the slaughterhouse as currently set out by Regulation (EC) No 999/2001 compared to the harvesting method proposed in the study. Overall, the level of infectivity in bovine tonsil is low. This, together with the declining and overall low BSE prevalence and the current policy on SRM removal, suggests a very low, if not negligible, human BSE exposure risk associated with exposure to lymphoid tissue in bovine tongue harvested as currently prescribed by EU legislation. The EFSA Journal (2008) 700, 16-24
18 Consumption of beef tongue Currently there are not sufficient quantitative data available allowing a comparison of the human BSE exposure risk reduction achieved by the alternative tongue harvesting method proposed by the study in comparison to the harvesting method currently prescribed by Regulation (EC) No 999/2001. However, it is likely that the proposed method would only provide a marginal reduction in the risk from bovine tonsil compared to the one currently prescribed. RECOMMENDATIONS To quantitatively assess the efficacy of the procedure currently prescribed in Regulation (EC) 999/2001 for the harvesting of bovine tongue in achieving the removal of bovine lingual tonsil. Should a quantification of the distribution of lymphoid tissue in the bovine tongue be necessary, additional morphological and immunohistochemical investigations should be performed. Should future studies of BSE infectivity in bovine tonsils be performed, bioassay of both palatine and lingual tonsils should be considered. This opinion only considers the risk associated with lymphoid formations in bovine tongue handled according to the measures currently set out by EU legislation. Infectivity in other tissues or structures of the tongue, as described in TSE animal models other than cattle, may contribute to infectivity in this organ. If new data become available with respect to involvement of such other tissues or structures in cattle BSE, further risk assessment should be performed. An estimation of the number of undetected BSE infected bovines entering into the food chain would assist in quantifying the residual BSE infectivity after SRM removal. DOCUMENTATION PROVIDED TO EFSA 1. Letter Consumption of beef tongue. June Submitted by the European Commission. 2. Scientific Article Casteleyn, C., Breugelmans, S., Muylle, S., Van den Broeck, W. and Simoens, P. (2007) Consumptie van rundertong en kalfszwezeriken: een risico voor de volksgezondheid? Vlaams Diergeneeskundig Tijdschrift, 76, Month Submitted by the European Commission. The Translation Centre for the Bodies of the European Union translated the Article into English. The validity and accuracy of this translation was verified and validated by Dr. Casteleyn. A copy of the English translation is attached to the Opinion as Annex 1, with permission of the authors. The EFSA Journal (2008) 700, 17-24
19 Consumption of beef tongue REFERENCES Arnold, M. E., Ryan, J. B. M., Konold, T., Simmons, M. M., Spencer, Y. I., Wear, A., Chaplin, M., Stack, M., Czub, S., Mueller, R., Webb, P. R., Davis, A., Spiropoulos, J., Holdaway, J., Hawkins, S. A. C., Austin, A. R. and Wells, G. A. H Estimating the temporal relationship between PrPSc detection and incubation period in experimental bovine spongiform encephalopathy of cattle. Journal Of General Virology 88: Bartz, J. C., Kincaid, A. E. and Bessen, R. A Rapid prion neuroinvasion following tongue infection. Journal Of Virology 77 (1): Buschmann, A. and Groschup, M. H Highly bovine spongiform encephalopathysensitive transgenic mice confirm the essential restriction of infectivity to the nervous system in clinically diseased cattle. Journal Of Infectious Diseases 192 (5): Casteleyn, C., Breugelmans, S., Muylle, S., Van den Broeck, W. and Simoens, P Consumption of beef tongue and sweetbread: risk for public health? Vlaams Diergeneeskundig Tijdschrift 76 (2): Cocquyt, G., Simoens, P., Muylle, S., Van den Broeck, W Anatomical and histological aspects of the bovine lingual tonsil. In press: Res. Vet. Sci., doi: /j.rvsc EC Report on the monitoring and testing of bovine animals for the presence of bovine spongiform encephalopathy (BSE) in EC Report on the monitoring and testing of ruminants for the presence of bovine spongiform encephalopathy (BSE) in EC Report on the monitoring and testing of ruminants for the presence of transmissible spongiform encephalopathy (TSE) in the EU in 2003, including the results of the survey of prion protein genotypes in sheep breeds. EC Report on the monitoring and testing of ruminants for the presence of transmissible spongiform encephalopathy (TSE) in the EU in EC Report on the monitoring and testing of ruminants for the presence of transmissible spongiform encephalopathy (TSE) in the EU in EC Report on the monitoring and testing of ruminants for the presence of transmissible spongiform encephalopathy (TSE) in the EU in EC BSE testing in EFSA Opinion of the Scientific Panel on Biological Hazards of the European Food Safety Authority on BSE risk from bovine tonsil and consumption of bovine tongue. The EFSA Journal 41: The EFSA Journal (2008) 700, 18-24
20 Consumption of beef tongue EFSA Opinion of the Scientific Panel on Biological Hazards on the assessment of the age limit in cattle for the removal of certain Specific Risk Materials (SRM). The EFSA Journal 220: EFSA Opinion of the Scientific Panel on Biological Hazards on the assessment of the likelihood of the infectivity in SRM derived from cattle at different age groups estimated by back calculation modelling. The EFSA Journal 476: Espinosa, J. C., Morales, M., Castilla, J., Rogers, M. and Torres, J. M Progression of prion infectivity in asymptomatic cattle after oral bovine spongiform encephalopathy challenge. Journal of General Virology 88: Fraser, H. and Foster, J. D Transmission to mice, sheep and goats and bioassay of bovine tissues. In Transmissible Spongiform Encephalopathies. Procedures of a consultation on BSE with the Scientific Veterinary Committee of the Commission of the European Communities held in Brussels from 14 to 15 September 1993: Iwata, N., Sato, Y., Higuchi, Y., Nohtomi, K., Nagata, N., Hasegawa, H., Tobiume, M., Nakamura, Y., Hagiwara, K., Furuoka, H., Horiuchi, M., Yamakawa, Y. and Sata, T Distribution of PrPSc in cattle with bovine spongiform encephalopathy slaughtered at abattoirs in Japan. Japanese Journal of Infectious Diseases 59 (2): Kato, K. and Sawada, Y Distribution of the lingual tonsils of cattle designated as specified risk materials. J. Vet. Med. Sci. 70 (3): Kühne, M., Klein, G. and Gasse, H Shortening of the bovine tongue according to regulation (EC) 999/2001 is not complying with the current legal definition of specified risk material - a macroscopical and histological preliminary study. Journal Of Veterinary Medicine Series B-Infectious Diseases And Veterinary Public Health 52 (2): LFRA Audit of bovine and ovine slaughter and by-products sector (ruminant products audit) for MAFF (MAFF Project No. SE0217). Nickel, R., Schummer, A. and Seiferle, E Digestive System. In The Viscera of Domestic Mammals. 2nd edn. Berlin, Verlag Paul Parey. Rebmann, S., Kühne, M., Klein, G. and Gasse, H. submitted for publication. The bovine lingual tonsil: topographical distribution of lympho-reticular tissue with special reference to regulations about the removal of specified risk material (SRM). Saegerman, C., Speybroeck, N., Vanopdenbosch, E., Wilesmith, J. W. and Berkvens, D Trends in age at detection in cases of bovine spongiform encephalopathy in Belgium: an indicator of the epidemic curve. Veterinary Record 159 (18): SEAC DNV risk assessment report on ox tongue and associated tonsil tissue. Paper No: SEAC 78/2. SSC Update of the Opinion on TSE infectivity distribution in ruminants tissues. Wells, G. A. H., Hawkins, S. A. C., Green, R. B., Austin, A. R., Dexter, I., Spencer, Y. I., Chaplin, M. J., Stack, M. J. and Dawson, M Preliminary observations on the pathogenesis of experimental bovine spongiform encephalopathy (BSE): an update. Veterinary Record 142 (5): The EFSA Journal (2008) 700, 19-24
Opinion of the Scientific Panel on Biological Hazards of the European Food Safety Authority on:
 Opinion of the Scientific Panel on Biological Hazards of the European Food Safety Authority on: A quantitative assessment of risk posed to humans by tissues of small ruminants in case BSE is present in
Opinion of the Scientific Panel on Biological Hazards of the European Food Safety Authority on: A quantitative assessment of risk posed to humans by tissues of small ruminants in case BSE is present in
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL
 EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Scientific Opinions C1 - Follow-up and dissemination of scientific opinions SCIENTIFIC STEERING COMMITTEE OPINION ON
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Scientific Opinions C1 - Follow-up and dissemination of scientific opinions SCIENTIFIC STEERING COMMITTEE OPINION ON
(Text with EEA relevance)
 L 225/76 19.8.2016 COMMISSION REGULATION (EU) 2016/1396 of 18 August 2016 amending certain Annexes to Regulation (No 999/2001 of the European Parliament and of the Council laying down rules for the prevention,
L 225/76 19.8.2016 COMMISSION REGULATION (EU) 2016/1396 of 18 August 2016 amending certain Annexes to Regulation (No 999/2001 of the European Parliament and of the Council laying down rules for the prevention,
PRE-EMPTIVE RISK ASSESSMENT SHOULD BSE IN SMALL RUMINANTS BE FOUND UNDER DOMESTIC CONDITIONS.
 EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate B - Scientific Health Opinions Unit B1 - Monitoring and dissemination of scientific opinions Scientific Steering Committee
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate B - Scientific Health Opinions Unit B1 - Monitoring and dissemination of scientific opinions Scientific Steering Committee
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL BLOOD AND CARCASS WHEN APPLYING CERTAIN STUNNING METHODS.)
 EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL SCIENTIFIC OPINION ON STUNNING METHODS AND BSE RISKS (THE RISK OF DISSEMINATION OF BRAIN PARTICLES INTO THE BLOOD AND CARCASS WHEN APPLYING
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL SCIENTIFIC OPINION ON STUNNING METHODS AND BSE RISKS (THE RISK OF DISSEMINATION OF BRAIN PARTICLES INTO THE BLOOD AND CARCASS WHEN APPLYING
Assignment 13.1: Proofreading Bovine Spongiform Encephalopathy
 Technical Editing, A 13.1, Proofreading Technical Editing Assignment 13.1: Proofreading Bovine Spongiform Encephalopathy The context This document is now set in type as it will appear in print unless corrected.
Technical Editing, A 13.1, Proofreading Technical Editing Assignment 13.1: Proofreading Bovine Spongiform Encephalopathy The context This document is now set in type as it will appear in print unless corrected.
Safefood helpline from the South from the North The Food Safety Promotion Board Abbey Court, Lower Abbey Street, Dublin 1
 Safefood helpline from the South 1850 40 4567 from the North 0800 085 1683 The Food Safety Promotion Board Abbey Court, Lower Abbey Street, Dublin 1 Food Safety Promotion Board Prepared by Food Safety
Safefood helpline from the South 1850 40 4567 from the North 0800 085 1683 The Food Safety Promotion Board Abbey Court, Lower Abbey Street, Dublin 1 Food Safety Promotion Board Prepared by Food Safety
Opinion of the Scientific Steering Committee on the GEOGRAPHICAL RISK OF BOVINE SPONGIFORM ENCEPHALOPATHY (GBR) in New Zealand
 Scientific Steering Committee November 2002 Opinion of the Scientific Steering Committee on the GEOGRAPHICAL RISK OF BOVINE SPONGIFORM ENCEPHALOPATHY (GBR) in New Zealand adopted by the SSC on 7 November
Scientific Steering Committee November 2002 Opinion of the Scientific Steering Committee on the GEOGRAPHICAL RISK OF BOVINE SPONGIFORM ENCEPHALOPATHY (GBR) in New Zealand adopted by the SSC on 7 November
About Food Health Impact Assessment
 Food Safety No. 1015001 from the Ministry of Health, Labour and Welfare Consumer Safety No. 5410, 2004 October 15, 2004 To: Mr. Masaaki Terada, Chairman Food Safety Commission Hidehisa Otsuji Minister
Food Safety No. 1015001 from the Ministry of Health, Labour and Welfare Consumer Safety No. 5410, 2004 October 15, 2004 To: Mr. Masaaki Terada, Chairman Food Safety Commission Hidehisa Otsuji Minister
Questions and Answers on TSE in sheep and goats
 MEMO/03/157 Brussels, 24 July 2003 Questions and Answers on TSE in sheep and goats What are Transmissible Spongiform Encephalopathies (TSEs)? TSEs are a family of diseases occurring in man and animals
MEMO/03/157 Brussels, 24 July 2003 Questions and Answers on TSE in sheep and goats What are Transmissible Spongiform Encephalopathies (TSEs)? TSEs are a family of diseases occurring in man and animals
OVER 30 MONTH CATTLE SLAUGHTER RULE (OTM Rule)
 BACKGROUND FSA REVIEW OF BSE CONTROLS OVER 30 MONTH CATTLE SLAUGHTER RULE (OTM Rule) THE RULE 1. The Over 30 Month Rule, with some exceptions, prohibits the sale of meat for human consumption from cattle
BACKGROUND FSA REVIEW OF BSE CONTROLS OVER 30 MONTH CATTLE SLAUGHTER RULE (OTM Rule) THE RULE 1. The Over 30 Month Rule, with some exceptions, prohibits the sale of meat for human consumption from cattle
ANNEX. to the COMMISSION IMPLEMENTING DECISION
 EUROPEAN COMMISSION Brussels, 30.4.2015 C(2015) 3024 final ANNEX 1 ANNEX to the COMMISSION IMPLEMENTING DECISION on the adoption of the multiannual work programme for 2016-2017 for the implementation of
EUROPEAN COMMISSION Brussels, 30.4.2015 C(2015) 3024 final ANNEX 1 ANNEX to the COMMISSION IMPLEMENTING DECISION on the adoption of the multiannual work programme for 2016-2017 for the implementation of
BSE Update Meat Industry Perspective. Randall Huffman, Ph.D. V.P. Scientific Affairs American Meat Institute Foundation
 BSE Update Meat Industry Perspective Randall Huffman, Ph.D. V.P. Scientific Affairs American Meat Institute Foundation Tuesday, December 23 USDA Announcement Overview BSE and how it spreads Control measures
BSE Update Meat Industry Perspective Randall Huffman, Ph.D. V.P. Scientific Affairs American Meat Institute Foundation Tuesday, December 23 USDA Announcement Overview BSE and how it spreads Control measures
COMMISSION (2003/708/EC)
 10.10.2003 L 258/11 COMMISSION COMMISSION DECISION of 7 October 2003 amending Annex E to Council Directive 91/68/EEC and Annexes I and II to Decision 93/198/EEC as regards the updating of the model health
10.10.2003 L 258/11 COMMISSION COMMISSION DECISION of 7 October 2003 amending Annex E to Council Directive 91/68/EEC and Annexes I and II to Decision 93/198/EEC as regards the updating of the model health
1. DEFINITION OF BSE AND ITS TESTING METHODS. (1) Japan s BSE Measures. Screening
 FINAL REPORT JAPAN-UNITED STATES BSE WORKING GROUP July 22, 2004 Introduction Pursuant to the agreement reached between the Government of Japan and the Government of the United States (U.S.) at the Third
FINAL REPORT JAPAN-UNITED STATES BSE WORKING GROUP July 22, 2004 Introduction Pursuant to the agreement reached between the Government of Japan and the Government of the United States (U.S.) at the Third
May 4-6, 2004 University of Arkansas
 May 4-6, 2004 University of Arkansas BSE Update Meat Industry Perspective Randall Huffman, Ph.D. V.P. Scientific Affairs American Meat Institute Foundation Tuesday, December 23 USDA Announcement Overview
May 4-6, 2004 University of Arkansas BSE Update Meat Industry Perspective Randall Huffman, Ph.D. V.P. Scientific Affairs American Meat Institute Foundation Tuesday, December 23 USDA Announcement Overview
This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents
 2001R0999 EN 17.11.2012 036.001 This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents B REGULATION (EC) No 999/2001 OF THE EUROPEAN PARLIAMENT
2001R0999 EN 17.11.2012 036.001 This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents B REGULATION (EC) No 999/2001 OF THE EUROPEAN PARLIAMENT
EUROPEAN COMMISSION. The TSE Roadmap 2. Brussels, COM (2010) 384 final
 EUROPEAN COMMISSION The TSE Roadmap 2 Brussels, 16.7.2010 COM (2010) 384 final European Union, 2010 Reproduction is authorised provided the source is acknowledged EN EN EN EUROPEAN COMMISSION Brussels,
EUROPEAN COMMISSION The TSE Roadmap 2 Brussels, 16.7.2010 COM (2010) 384 final European Union, 2010 Reproduction is authorised provided the source is acknowledged EN EN EN EUROPEAN COMMISSION Brussels,
Specified risk material and its amounts removed from cattle carcasses. Eva Vařechová
 Specified risk material and its amounts removed from cattle carcasses Eva Vařechová Regional Veterinary Administration State Veterinary Administration for the Moravian-Silesian Region Nový Jičín, Czech
Specified risk material and its amounts removed from cattle carcasses Eva Vařechová Regional Veterinary Administration State Veterinary Administration for the Moravian-Silesian Region Nový Jičín, Czech
Scientific Opinion on BSE/TSE infectivity in small ruminant tissues 1
 SCIENTIFIC OPINION Scientific Opinion on BSE/TSE infectivity in small ruminant tissues 1 EFSA Panel on Biological Hazards (BIOHAZ) 2, 3 European Food Safety Authority (EFSA), Parma, Italy ABSTRACT The
SCIENTIFIC OPINION Scientific Opinion on BSE/TSE infectivity in small ruminant tissues 1 EFSA Panel on Biological Hazards (BIOHAZ) 2, 3 European Food Safety Authority (EFSA), Parma, Italy ABSTRACT The
COMMISSION DELEGATED REGULATION (EU)
 L 296/6 Official Journal of the European Union 15.11.2011 COMMISSION DELEGATED REGULATION (EU) No 1152/2011 of 14 July 2011 supplementing Regulation (EC) No 998/2003 of the European Parliament and of the
L 296/6 Official Journal of the European Union 15.11.2011 COMMISSION DELEGATED REGULATION (EU) No 1152/2011 of 14 July 2011 supplementing Regulation (EC) No 998/2003 of the European Parliament and of the
Animal Health Requirements for beef and beef offal to be exported to Japan from Norway
 Animal Health Requirements for beef and beef offal to be exported to Japan from Norway Animal health requirements for beef and beef offal to be exported to Japan from Norway are as follows: 1. Definitions
Animal Health Requirements for beef and beef offal to be exported to Japan from Norway Animal health requirements for beef and beef offal to be exported to Japan from Norway are as follows: 1. Definitions
Annual report of the Scientific Network on BSE-TSE 2015
 TECHNICAL REPORT APPROVED: 10 December 2015 PUBLISHED: 11 December 2015 Annual report of the Scientific Network on BSE-TSE 2015 Abstract European Food Safety Authority The EFSA Scientific Network on bovine
TECHNICAL REPORT APPROVED: 10 December 2015 PUBLISHED: 11 December 2015 Annual report of the Scientific Network on BSE-TSE 2015 Abstract European Food Safety Authority The EFSA Scientific Network on bovine
THE DEVELOPMENT OF A RISK BASED MEAT INSPECTION SYSTEM SANCO / 4403 / 2000
 FEDERATION OF VETERINARIANS OF EUROPE FVE/01/034 Final THE DEVELOPMENT OF A RISK BASED MEAT INSPECTION SYSTEM SANCO / 4403 / 2000 Members FVE COMMENTS Austria Belgium Croatia Cyprus Czech Republic Denmark
FEDERATION OF VETERINARIANS OF EUROPE FVE/01/034 Final THE DEVELOPMENT OF A RISK BASED MEAT INSPECTION SYSTEM SANCO / 4403 / 2000 Members FVE COMMENTS Austria Belgium Croatia Cyprus Czech Republic Denmark
Standard requirements for the submission of programmes of eradication and monitoring of TSE
 Member States seeking a financial contribution from the Community for national programmes for the control and monitoring of transmissible spongiform encephalopathies (TSEs), shall submit applications containing
Member States seeking a financial contribution from the Community for national programmes for the control and monitoring of transmissible spongiform encephalopathies (TSEs), shall submit applications containing
COMMISSION REGULATION (EU)
 L 179/60 Official Journal of the European Union 29.6.2013 COMMISSION REGULATION (EU) No 630/2013 of 28 June 2013 amending the Annexes to Regulation (EC) No 999/2001 of the European Parliament and of the
L 179/60 Official Journal of the European Union 29.6.2013 COMMISSION REGULATION (EU) No 630/2013 of 28 June 2013 amending the Annexes to Regulation (EC) No 999/2001 of the European Parliament and of the
Official Journal of the European Union L 280/5
 24.10.2007 Official Journal of the European Union L 280/5 COMMISSION REGULATION (EC) No 1237/2007 of 23 October 2007 amending Regulation (EC) No 2160/2003 of the European Parliament and of the Council
24.10.2007 Official Journal of the European Union L 280/5 COMMISSION REGULATION (EC) No 1237/2007 of 23 October 2007 amending Regulation (EC) No 2160/2003 of the European Parliament and of the Council
Mobile Slaughter Unit
 Mobile Slaughter Unit Name of the business/responsible entity USDA Facility Number: 00000 Standard Operating Procedures (SOP) Signature Page Slaughter: beef, swine, goat, and lamb (list all species you
Mobile Slaughter Unit Name of the business/responsible entity USDA Facility Number: 00000 Standard Operating Procedures (SOP) Signature Page Slaughter: beef, swine, goat, and lamb (list all species you
BSE: Diagnosis and control, the consequences for human health and geographical BSE risk. Emmanuel Vanopdenbosch* and Stefan Roels**
 BSE: Diagnosis and control, the consequences for human health and geographical BSE risk Emmanuel Vanopdenbosch* and Stefan Roels** *Head of the Department of Biocontrol Veterinary and Agrochemical Research
BSE: Diagnosis and control, the consequences for human health and geographical BSE risk Emmanuel Vanopdenbosch* and Stefan Roels** *Head of the Department of Biocontrol Veterinary and Agrochemical Research
COMMISSION DELEGATED REGULATION (EU) /... of XXX
 Ref. Ares(2017)4396495-08/09/2017 EUROPEAN COMMISSION Brussels, XXX SANTE/7009/2016 CIS Rev. 1 (POOL/G2/2016/7009/7009R1-EN CIS.doc) [ ](2016) XXX draft COMMISSION DELEGATED REGULATION (EU) /... of XXX
Ref. Ares(2017)4396495-08/09/2017 EUROPEAN COMMISSION Brussels, XXX SANTE/7009/2016 CIS Rev. 1 (POOL/G2/2016/7009/7009R1-EN CIS.doc) [ ](2016) XXX draft COMMISSION DELEGATED REGULATION (EU) /... of XXX
COMMISSION OF THE EUROPEAN COMMUNITIES REPORT FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT AND THE COUNCIL
 COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 8.10.2007 COM(2007) 578 final REPORT FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT AND THE COUNCIL in connection with Article 23 of Regulation (EC) No
COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 8.10.2007 COM(2007) 578 final REPORT FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT AND THE COUNCIL in connection with Article 23 of Regulation (EC) No
COMMISSION OF THE EUROPEAN COMMUNITIES. Proposal for a REGULATION OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL
 COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 16.6.2009 COM(2009) 268 final 2009/0077 (COD) C7-0035/09 Proposal for a REGULATION OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL amending Regulation (EC)
COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 16.6.2009 COM(2009) 268 final 2009/0077 (COD) C7-0035/09 Proposal for a REGULATION OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL amending Regulation (EC)
21st Conference of the OIE Regional Commission for Europe. Avila (Spain), 28 September 1 October 2004
 21st Conference of the OIE Regional Commission for Europe Avila (Spain), 28 September 1 October 2004 Recommendation No. 1: Recommendation No. 2: Recommendation No. 3: Contingency planning and simulation
21st Conference of the OIE Regional Commission for Europe Avila (Spain), 28 September 1 October 2004 Recommendation No. 1: Recommendation No. 2: Recommendation No. 3: Contingency planning and simulation
3. records of distribution for proteins and feeds are being kept to facilitate tracing throughout the animal feed and animal production chain.
 CANADA S FEED BAN The purpose of this paper is to explain the history and operation of Canada s feed ban and to put it into a broader North American context. Canada and the United States share the same
CANADA S FEED BAN The purpose of this paper is to explain the history and operation of Canada s feed ban and to put it into a broader North American context. Canada and the United States share the same
HEALTH & CONSUMERS DIRECTORATE-GENERAL
 EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL SANCO D D(2011) 1198550 SUMMARY RECORD OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD IN BRUSSELS ON 3 & 4 MAY 2010 (Section
EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL SANCO D D(2011) 1198550 SUMMARY RECORD OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD IN BRUSSELS ON 3 & 4 MAY 2010 (Section
EFSA s activities on Antimicrobial Resistance
 EFSA s activities on Antimicrobial Resistance CRL-AR, Copenhagen 23 April 2009 Annual Workshop of CRL - AR 1 Efsa s Role and Activities on AMR Scientific advices Analyses of data on AR submitted by MSs
EFSA s activities on Antimicrobial Resistance CRL-AR, Copenhagen 23 April 2009 Annual Workshop of CRL - AR 1 Efsa s Role and Activities on AMR Scientific advices Analyses of data on AR submitted by MSs
(Non-legislative acts) REGULATIONS
 8.9.2010 Official Journal of the European Union L 237/1 II (Non-legislative acts) REGULATIONS COMMISSION REGULATION (EU) No 790/2010 of 7 September 2010 amending Annexes VII, X and XI to Regulation (EC)
8.9.2010 Official Journal of the European Union L 237/1 II (Non-legislative acts) REGULATIONS COMMISSION REGULATION (EU) No 790/2010 of 7 September 2010 amending Annexes VII, X and XI to Regulation (EC)
Bovine Spongiform Encephalopathy
 Bovine Spongiform Encephalopathy Mad Cow Disease Warren J. Hess, DVM Acting State Veterinarian Utah Department of Agriculture and Food Transmissible Spongiform Encephalopathies Bovine (BSE) Sheep/Goats
Bovine Spongiform Encephalopathy Mad Cow Disease Warren J. Hess, DVM Acting State Veterinarian Utah Department of Agriculture and Food Transmissible Spongiform Encephalopathies Bovine (BSE) Sheep/Goats
Having regard to the Treaty establishing the European Community, and in particular Article 152(4)(b) thereof,
 14.10.2003 L 262/17 DIRECTIVE 2003/74/EC OF THE EUROPEAN PARLIAMT AND OF THE COUNCIL of 22 September 2003 amending Council Directive 96/22/EC concerning the prohibition on the use in stockfarming of certain
14.10.2003 L 262/17 DIRECTIVE 2003/74/EC OF THE EUROPEAN PARLIAMT AND OF THE COUNCIL of 22 September 2003 amending Council Directive 96/22/EC concerning the prohibition on the use in stockfarming of certain
This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents
 2003R2160 EN 27.10.2007 003.001 1 This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents B REGULATION (EC) No 2160/2003 OF THE EUROPEAN
2003R2160 EN 27.10.2007 003.001 1 This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents B REGULATION (EC) No 2160/2003 OF THE EUROPEAN
Joint WHO/FAO/OIE Technical Consultation on BSE: public health, animal health and trade
 Joint WHO/FAO/OIE Technical Consultation on BSE: OIE Headquarters, Paris, 11-14 June 2001 Conclusions and key recommendations World Organisation for Animal Health (Office International des Epizooties),
Joint WHO/FAO/OIE Technical Consultation on BSE: OIE Headquarters, Paris, 11-14 June 2001 Conclusions and key recommendations World Organisation for Animal Health (Office International des Epizooties),
Joint scientific report of ECDC, EFSA and EMEA on meticillin resistant Staphylococcus aureus (MRSA) in livestock, companion animals and food 1.
 16 June 2009 Joint scientific report of ECDC, EFSA and EMEA on meticillin resistant Staphylococcus aureus (MRSA) in livestock, companion animals and food 1. Summary of the scientific Opinion of the Panel
16 June 2009 Joint scientific report of ECDC, EFSA and EMEA on meticillin resistant Staphylococcus aureus (MRSA) in livestock, companion animals and food 1. Summary of the scientific Opinion of the Panel
L 39/12 Official Journal of the European Union
 L 39/12 Official Journal of the European Union 10.2.2009 COMMISSION REGULATION (EC) No 119/2009 of 9 February 2009 laying down a list of third countries or parts thereof, for imports into, or transit through,
L 39/12 Official Journal of the European Union 10.2.2009 COMMISSION REGULATION (EC) No 119/2009 of 9 February 2009 laying down a list of third countries or parts thereof, for imports into, or transit through,
Official Journal of the European Union. (Acts whose publication is obligatory)
 12.12.2003 L 325/1 I (Acts whose publication is obligatory) REGULATION (EC) No 2160/2003 OF THE EUROPEAN PARLIAMT AND OF THE COUNCIL of 17 November 2003 on the control of salmonella and other specified
12.12.2003 L 325/1 I (Acts whose publication is obligatory) REGULATION (EC) No 2160/2003 OF THE EUROPEAN PARLIAMT AND OF THE COUNCIL of 17 November 2003 on the control of salmonella and other specified
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL
 EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Scientific Opinions C1 - Follow-up and dissemination of scientific opinions SSC meeting of 6-7 September2001 / 6.2.a
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Scientific Opinions C1 - Follow-up and dissemination of scientific opinions SSC meeting of 6-7 September2001 / 6.2.a
Law on Special Measures Against Bovine Spongiform Encephalopathy (Law No. 70 of June 14, 2002)
 Law on Special Measures Against Bovine Spongiform Encephalopathy (Law No. 70 of June 14, 2002) Last amendment: Law No. 119 of July 16, 2003 (Laws and regulations yet to be enforced at the time of last
Law on Special Measures Against Bovine Spongiform Encephalopathy (Law No. 70 of June 14, 2002) Last amendment: Law No. 119 of July 16, 2003 (Laws and regulations yet to be enforced at the time of last
REPORT FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT AND THE COUNCIL
 EUROPEAN COMMISSION Brussels, 6.3.2018 COM(2018) 88 final REPORT FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT AND THE COUNCIL on the implementation of Article 5 of Regulation (EU) No 576/2013 on the
EUROPEAN COMMISSION Brussels, 6.3.2018 COM(2018) 88 final REPORT FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT AND THE COUNCIL on the implementation of Article 5 of Regulation (EU) No 576/2013 on the
ANNEX. to the. Commission Implementing Decision
 EUROPEAN COMMISSION Brussels, 2.5.2017 C(2017) 2841 final ANNEX 1 ANNEX to the Commission Implementing Decision on the adoption of the multiannual work programme for 2018, 2019 and 2020 for the implementation
EUROPEAN COMMISSION Brussels, 2.5.2017 C(2017) 2841 final ANNEX 1 ANNEX to the Commission Implementing Decision on the adoption of the multiannual work programme for 2018, 2019 and 2020 for the implementation
L 210/36 Official Journal of the European Union DECISIONS COMMISSION
 L 210/36 Official Journal of the European Union 10.8.2007 II (Acts adopted under the EC Treaty/Euratom Treaty whose publication is not obligatory) DECISIONS COMMISSION COMMISSION DECISION of 9 August 2007
L 210/36 Official Journal of the European Union 10.8.2007 II (Acts adopted under the EC Treaty/Euratom Treaty whose publication is not obligatory) DECISIONS COMMISSION COMMISSION DECISION of 9 August 2007
TEXTS ADOPTED Provisional edition. P8_TA-PROV(2018)0429 Animal welfare, antimicrobial use and the environmental impact of industrial broiler farming
 European Parliament 204-209 TEXTS ADOPTED Provisional edition P8_TA-PROV(208)0429 Animal welfare, antimicrobial use and the environmental impact of industrial broiler farming European Parliament resolution
European Parliament 204-209 TEXTS ADOPTED Provisional edition P8_TA-PROV(208)0429 Animal welfare, antimicrobial use and the environmental impact of industrial broiler farming European Parliament resolution
Conference on meat inspection
 Conference on meat inspection Animal health and welfare monitoring as integrated part of meat inspection Alberto Laddomada Head of Unit SANCO D1 Animal Health and Standing Committees Prevention is better
Conference on meat inspection Animal health and welfare monitoring as integrated part of meat inspection Alberto Laddomada Head of Unit SANCO D1 Animal Health and Standing Committees Prevention is better
EN SANCO/745/2008r6 EN EN
 SANCO/745/2008r6 COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, C(2008) Commission staff working document GUIDANCE DOCUMT On the minimum requirements for Salmonella control programmes to be recognised
SANCO/745/2008r6 COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, C(2008) Commission staff working document GUIDANCE DOCUMT On the minimum requirements for Salmonella control programmes to be recognised
Bovine spongiform encephalopathy: an update *
 Rev. sci. tech. Off. int. Epiz., 1996, IS (3), 1087-1118 Bovine spongiform encephalopathy: an update * Summary: A specialist group of the Office International des Epizooties met in May 1996 to prepare
Rev. sci. tech. Off. int. Epiz., 1996, IS (3), 1087-1118 Bovine spongiform encephalopathy: an update * Summary: A specialist group of the Office International des Epizooties met in May 1996 to prepare
Opinion of the Committee for Medicinal Products for Veterinary Use pursuant to Article 30(3) of Regulation (EC) No 726/2004
 11 December 2014 EMA/CVMP/761582/2014 Veterinary Medicines Division EMEA/V/A/107 Opinion of the Committee for Medicinal Products for Veterinary Use pursuant to Article 30(3) of Regulation (EC) No 726/2004
11 December 2014 EMA/CVMP/761582/2014 Veterinary Medicines Division EMEA/V/A/107 Opinion of the Committee for Medicinal Products for Veterinary Use pursuant to Article 30(3) of Regulation (EC) No 726/2004
COMMISSION OF THE EUROPEAN COMMUNITIES REPORT FROM THE COMMISSION TO THE COUNCIL AND THE EUROPEAN PARLIAMENT
 COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 20.1.2005 COM(2005) 7 final. REPORT FROM THE COMMISSION TO THE COUNCIL AND THE EUROPEAN PARLIAMENT FOURTH REPORT ON THE STATISTICS ON THE NUMBER OF ANIMALS
COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 20.1.2005 COM(2005) 7 final. REPORT FROM THE COMMISSION TO THE COUNCIL AND THE EUROPEAN PARLIAMENT FOURTH REPORT ON THE STATISTICS ON THE NUMBER OF ANIMALS
Tentative translation
 Tentative translation Risk assessment concerning the comparability between risks of consuming beef and internal organs regulated by the beef export verification program of the United States/Canada and
Tentative translation Risk assessment concerning the comparability between risks of consuming beef and internal organs regulated by the beef export verification program of the United States/Canada and
Council of the European Union Brussels, 13 June 2016 (OR. en)
 Council of the European Union Brussels, 13 June 2016 (OR. en) 9952/16 SAN 241 AGRI 312 VETER 58 NOTE From: To: General Secretariat of the Council Council No. prev. doc.: 9485/16 SAN 220 AGRI 296 VETER
Council of the European Union Brussels, 13 June 2016 (OR. en) 9952/16 SAN 241 AGRI 312 VETER 58 NOTE From: To: General Secretariat of the Council Council No. prev. doc.: 9485/16 SAN 220 AGRI 296 VETER
and suitability aspects of food control. CAC and the OIE have Food safety is an issue of increasing concern world wide and
 forum Cooperation between the Codex Alimentarius Commission and the OIE on food safety throughout the food chain Information Document prepared by the OIE Working Group on Animal Production Food Safety
forum Cooperation between the Codex Alimentarius Commission and the OIE on food safety throughout the food chain Information Document prepared by the OIE Working Group on Animal Production Food Safety
OIE Standards for: Animal identification and traceability Antimicrobials
 OIE Standards for: Animal identification and traceability Antimicrobials OIE regional seminar on food safety Singapore, 12-14 October 2010 Yamato Atagi 1 Deputy Head, International Trade Department, OIE
OIE Standards for: Animal identification and traceability Antimicrobials OIE regional seminar on food safety Singapore, 12-14 October 2010 Yamato Atagi 1 Deputy Head, International Trade Department, OIE
RESTRAINING SYSTEMS FOR BOVINE ANIMALS SLAUGHTERED WITHOUT STUNNING WELFARE AND SOCIO-ECONOMIC IMPLICATIONS
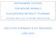 RESTRAINING SYSTEMS FOR BOVINE ANIMALS SLAUGHTERED WITHOUT STUNNING WELFARE AND SOCIO-ECONOMIC IMPLICATIONS EXECUTIVE SUMMARY & KEY MESSAGES JUNE 2015 SCOPE AND BACKGROUND The study exclusively refers
RESTRAINING SYSTEMS FOR BOVINE ANIMALS SLAUGHTERED WITHOUT STUNNING WELFARE AND SOCIO-ECONOMIC IMPLICATIONS EXECUTIVE SUMMARY & KEY MESSAGES JUNE 2015 SCOPE AND BACKGROUND The study exclusively refers
REPORT FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT AND THE COUNCIL. on systems restraining bovine animals by inversion or any unnatural position
 EUROPEAN COMMISSION Brussels, 8.2.2016 COM(2016) 48 final REPORT FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT AND THE COUNCIL on systems restraining bovine animals by inversion or any unnatural position
EUROPEAN COMMISSION Brussels, 8.2.2016 COM(2016) 48 final REPORT FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT AND THE COUNCIL on systems restraining bovine animals by inversion or any unnatural position
EFSA s activities on Antimicrobial resistance in the food chain. Dr. Ernesto Liebana Head of BIOCONTAM Unit. EFSA
 EFSA s activities on Antimicrobial resistance in the food chain Dr. Ernesto Liebana Head of BIOCONTAM Unit. EFSA EFSA IS The reference body for risk assessment of food and feed in the European Union. Its
EFSA s activities on Antimicrobial resistance in the food chain Dr. Ernesto Liebana Head of BIOCONTAM Unit. EFSA EFSA IS The reference body for risk assessment of food and feed in the European Union. Its
The EFSA s BIOHAZ Panel perspective on food microbiology and hygiene
 The EFSA s BIOHAZ Panel perspective on food microbiology and hygiene Dr Eirini Tsigarida Unit of Biological Hazards BIOHAZ Unit: Marta Hugas, Bart Goossens, Tobin Robinson, Fulvio Barizzone, Luis Vivas-
The EFSA s BIOHAZ Panel perspective on food microbiology and hygiene Dr Eirini Tsigarida Unit of Biological Hazards BIOHAZ Unit: Marta Hugas, Bart Goossens, Tobin Robinson, Fulvio Barizzone, Luis Vivas-
EUROPEAN COMMISSION DIRECTORATE-GENERAL FOR HEALTH AND FOOD SAFETY
 EUROPEAN COMMISSION DIRECTORATE-GENERAL FOR HEALTH AND FOOD SAFETY Health and food audits and analysis DG(SANTE) 2017-6110 FINAL REPORT OF A FACT-FINDING MISSION CARRIED OUT IN TURKEY FROM 05 SEPTEMBER
EUROPEAN COMMISSION DIRECTORATE-GENERAL FOR HEALTH AND FOOD SAFETY Health and food audits and analysis DG(SANTE) 2017-6110 FINAL REPORT OF A FACT-FINDING MISSION CARRIED OUT IN TURKEY FROM 05 SEPTEMBER
IMPORT HEALTH STANDARD FOR THE IMPORTATION INTO NEW ZEALAND OF RABBIT MEAT FOR HUMAN CONSUMPTION FROM THE EUROPEAN COMMUNITY
 IMPORT HEALTH STANDARD FOR THE IMPORTATION INTO NEW ZEALAND OF RABBIT MEAT FOR HUMAN CONSUMPTION FROM THE EUROPEAN COMMUNITY ANNEX A ASSIGNED NUMBERS (AN): 4C.2, 4D.1, 5C.2, 5D.1, 6C.1, 6D.2, Issued pursuant
IMPORT HEALTH STANDARD FOR THE IMPORTATION INTO NEW ZEALAND OF RABBIT MEAT FOR HUMAN CONSUMPTION FROM THE EUROPEAN COMMUNITY ANNEX A ASSIGNED NUMBERS (AN): 4C.2, 4D.1, 5C.2, 5D.1, 6C.1, 6D.2, Issued pursuant
Working for organic farming in Europe
 Working for organic farming in Europe International Federation of Organic Agriculture Movements EU Regional Group 9 st November 2012 President: Christopher Stopes Director: Marco Schlüter European Office
Working for organic farming in Europe International Federation of Organic Agriculture Movements EU Regional Group 9 st November 2012 President: Christopher Stopes Director: Marco Schlüter European Office
Regulatory Information
 Home Regulatory Information Search for FDA Guidance Documents Regulatory Information The Sourcing and Processing of Gelatin to Reduce the Potential Risk Posed by Bovine Spongiform Encephalopathy (BSE)
Home Regulatory Information Search for FDA Guidance Documents Regulatory Information The Sourcing and Processing of Gelatin to Reduce the Potential Risk Posed by Bovine Spongiform Encephalopathy (BSE)
Official Journal of the European Union L 162/3
 21.6.2008 Official Journal of the European Union L 162/3 COMMISSION REGULATION (EC) No 584/2008 of 20 June 2008 implementing Regulation (EC) No 2160/2003 of the European Parliament and of the Council as
21.6.2008 Official Journal of the European Union L 162/3 COMMISSION REGULATION (EC) No 584/2008 of 20 June 2008 implementing Regulation (EC) No 2160/2003 of the European Parliament and of the Council as
EVALUATION OF A RAPID ANTE MORTEM BSE TEST
 EVALUATION OF A RAPID ANTE MORTEM BSE TEST Scientific Report of the Scientific Expert Working Group of the European Food Safety Authority on Transmissible Spongiform Encephalopathy (TSE) 1 Adopted on 27
EVALUATION OF A RAPID ANTE MORTEM BSE TEST Scientific Report of the Scientific Expert Working Group of the European Food Safety Authority on Transmissible Spongiform Encephalopathy (TSE) 1 Adopted on 27
Standard requirements for the submission of programmes of eradication and monitoring of TSE
 Member States seeking a financial contribution from the Community for national programmes for the control and monitoring of transmissible spongiform encephalopathies (TSEs), shall submit applications containing
Member States seeking a financial contribution from the Community for national programmes for the control and monitoring of transmissible spongiform encephalopathies (TSEs), shall submit applications containing
COMMISSION DELEGATED REGULATION (EU) /... of XXX
 Ref. Ares(2018)4937331-26/09/2018 EUROPEAN COMMISSION Brussels, XXX SANTE/10193/2017 CIS Rev. 2 (POOL/G4/2017/10193/10193R2-EN CIS.doc) [ ](2018) XXX draft COMMISSION DELEGATED REGULATION (EU) /... of
Ref. Ares(2018)4937331-26/09/2018 EUROPEAN COMMISSION Brussels, XXX SANTE/10193/2017 CIS Rev. 2 (POOL/G4/2017/10193/10193R2-EN CIS.doc) [ ](2018) XXX draft COMMISSION DELEGATED REGULATION (EU) /... of
REPORT OF THE MEETING OF THE OIE AD HOC GROUP TO REVIEW THE BOVINE SPONGIFORM ENCEPHALOPATHY CHAPTER IN THE OIE TERRESTRIAL ANIMAL HEALTH CODE
 Original: English September 2003 REPORT OF THE MEETING OF THE OIE AD HOC GROUP TO REVIEW THE BOVINE SPONGIFORM ENCEPHALOPATHY CHAPTER IN THE OIE TERRESTRIAL ANIMAL HEALTH CODE Paris, 22-24 September 2003
Original: English September 2003 REPORT OF THE MEETING OF THE OIE AD HOC GROUP TO REVIEW THE BOVINE SPONGIFORM ENCEPHALOPATHY CHAPTER IN THE OIE TERRESTRIAL ANIMAL HEALTH CODE Paris, 22-24 September 2003
Assessment Panel mapping document for
 Assessment Panel mapping document for Last updated: December 2015 Aim: To provide the candidate with knowledge, understanding and application of animal health, welfare, food hygiene and feed hygiene legislation.
Assessment Panel mapping document for Last updated: December 2015 Aim: To provide the candidate with knowledge, understanding and application of animal health, welfare, food hygiene and feed hygiene legislation.
REGULATION (EC) No 854/2004 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 29 April 2004
 30.4.2004 EN Official Journal of the European Union L 155/206 REGULATION (EC) No 854/2004 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 29 April 2004 laying down specific rules for the organisation
30.4.2004 EN Official Journal of the European Union L 155/206 REGULATION (EC) No 854/2004 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 29 April 2004 laying down specific rules for the organisation
(Non-legislative acts) DECISIONS
 EN 5.6.2012 Official Journal of the European Union L 145/1 II (Non-legislative acts) DECISIONS COMMISSION IMPLEMENTING DECISION of 22 May 2012 amending Decision 2008/425/EC as regards standard requirements
EN 5.6.2012 Official Journal of the European Union L 145/1 II (Non-legislative acts) DECISIONS COMMISSION IMPLEMENTING DECISION of 22 May 2012 amending Decision 2008/425/EC as regards standard requirements
ISO INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 22442-2 First edition 2007-12-15 Medical devices utilizing animal tissues and their derivatives Part 2: Controls on sourcing, collection and handling Dispositifs médicaux utilisant
INTERNATIONAL STANDARD ISO 22442-2 First edition 2007-12-15 Medical devices utilizing animal tissues and their derivatives Part 2: Controls on sourcing, collection and handling Dispositifs médicaux utilisant
This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents
 2003L0099 EN 01.01.2007 001.001 1 This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents B DIRECTIVE 2003/99/EC OF THE EUROPEAN PARLIAMENT
2003L0099 EN 01.01.2007 001.001 1 This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents B DIRECTIVE 2003/99/EC OF THE EUROPEAN PARLIAMENT
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE VETERINARY MEDICINAL PRODUCT BLUEVAC BTV8 suspension for injection for cattle and sheep 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE VETERINARY MEDICINAL PRODUCT BLUEVAC BTV8 suspension for injection for cattle and sheep 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of
European Public MRL assessment report (EPMAR)
 18 March 2016 EMA/CVMP/619817/2015 Committee for Medicinal Products for Veterinary Use European Public MRL assessment report (EPMAR) Gentamicin (all mammalian food producing species and fin fish) On 3
18 March 2016 EMA/CVMP/619817/2015 Committee for Medicinal Products for Veterinary Use European Public MRL assessment report (EPMAR) Gentamicin (all mammalian food producing species and fin fish) On 3
ADDING VALUE TO THE SCOTTISH RED MEAT SUPPLY CHAIN
 Recovering Value from the 5th Quarter and Reducing Waste Topics of Common Interest An Industry Guide to the Identification of Category 1, 2 and 3 Material Animal by products (ABPs) are divided into three
Recovering Value from the 5th Quarter and Reducing Waste Topics of Common Interest An Industry Guide to the Identification of Category 1, 2 and 3 Material Animal by products (ABPs) are divided into three
This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents
 1996L0022 EN 18.12.2008 002.001 1 This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents B COUNCIL DIRECTIVE 96/22/EC of 29 April 1996 concerning
1996L0022 EN 18.12.2008 002.001 1 This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents B COUNCIL DIRECTIVE 96/22/EC of 29 April 1996 concerning
Summary of the latest data on antibiotic consumption in the European Union
 Summary of the latest data on antibiotic consumption in the European Union ESAC-Net surveillance data November 2016 Provision of reliable and comparable national antimicrobial consumption data is a prerequisite
Summary of the latest data on antibiotic consumption in the European Union ESAC-Net surveillance data November 2016 Provision of reliable and comparable national antimicrobial consumption data is a prerequisite
2010 No AGRICULTURE, ENGLAND. The Animals and Animal Products (Import and Export) (England) (Amendment) Regulations 2010
 STATUTORY INSTRUMENTS 2010 No. 1760 AGRICULTURE, ENGLAND The Animals and Animal Products (Import and Export) (England) (Amendment) Regulations 2010 Made - - - - 5th July 2010 Laid before Parliament 8th
STATUTORY INSTRUMENTS 2010 No. 1760 AGRICULTURE, ENGLAND The Animals and Animal Products (Import and Export) (England) (Amendment) Regulations 2010 Made - - - - 5th July 2010 Laid before Parliament 8th
COMMISSION OF THE EUROPEAN COMMUNITIES
 COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 22 December 2005 COM (2005) 0684 REPORT FROM THE COMMISSION TO THE COUNCIL ON THE BASIS OF MEMBER STATES REPORTS ON THE IMPLEMENTATION OF THE COUNCIL RECOMMENDATION
COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 22 December 2005 COM (2005) 0684 REPORT FROM THE COMMISSION TO THE COUNCIL ON THE BASIS OF MEMBER STATES REPORTS ON THE IMPLEMENTATION OF THE COUNCIL RECOMMENDATION
Overview of ongoing EFSA work on the meat inspection mandate
 Overview of ongoing EFSA work on the meat inspection mandate EFSA Stakeholder Consultative Platform 18th Meeting Pablo Romero Barrios Unit on Biological Hazards Outline Background for meat inspection mandate
Overview of ongoing EFSA work on the meat inspection mandate EFSA Stakeholder Consultative Platform 18th Meeting Pablo Romero Barrios Unit on Biological Hazards Outline Background for meat inspection mandate
Better Training for Safer Food
 Better Training for Safer Food Initiative Susanne Münstermann Better Training for Safer Food is an initiative of the European Commission aimed at organising an EU training strategy in the areas of food
Better Training for Safer Food Initiative Susanne Münstermann Better Training for Safer Food is an initiative of the European Commission aimed at organising an EU training strategy in the areas of food
LIFE.2.B EUROPEAN UNION. Brussels, 14 November 2018 (OR. en) 2014/0255 (COD) PE-CONS 43/18 AGRILEG 102 VETER 52 CODEC 1149
 EUROPEAN UNION THE EUROPEAN PARLIAMT THE COUNCIL Brussels, 14 November 2018 (OR. en) 2014/0255 (COD) PE-CONS 43/18 AGRILEG 102 VETER 52 CODEC 1149 LEGISLATIVE ACTS AND OTHER INSTRUMTS Subject: REGULATION
EUROPEAN UNION THE EUROPEAN PARLIAMT THE COUNCIL Brussels, 14 November 2018 (OR. en) 2014/0255 (COD) PE-CONS 43/18 AGRILEG 102 VETER 52 CODEC 1149 LEGISLATIVE ACTS AND OTHER INSTRUMTS Subject: REGULATION
Council Conclusions on Antimicrobial Resistance (AMR) 2876th EMPLOYMENT, SOCIAL POLICY, HEALTH AND CONSUMER AFFAIRS Council meeting
 COUNCIL OF THE EUROPEAN UNION Council Conclusions on Antimicrobial Resistance (AMR) 2876th EMPLOYMT, SOCIAL POLICY, HEALTH AND CONSUMER AFFAIRS Council meeting Luxembourg, 10 June 2008 The Council adopted
COUNCIL OF THE EUROPEAN UNION Council Conclusions on Antimicrobial Resistance (AMR) 2876th EMPLOYMT, SOCIAL POLICY, HEALTH AND CONSUMER AFFAIRS Council meeting Luxembourg, 10 June 2008 The Council adopted
This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents
 1996L0022 EN 18.12.2008 002.001 This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents B COUNCIL DIRECTIVE 96/22/EC of 29 April 1996 concerning
1996L0022 EN 18.12.2008 002.001 This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents B COUNCIL DIRECTIVE 96/22/EC of 29 April 1996 concerning
NIAA Resolutions Bovine Committee
 2016-2017 NIAA Resolutions Bovine Committee Mission: To bring the dairy cattle and beef cattle industries together for implementation and development of programs that assure the health and welfare of our
2016-2017 NIAA Resolutions Bovine Committee Mission: To bring the dairy cattle and beef cattle industries together for implementation and development of programs that assure the health and welfare of our
European Medicines Agency role and experience on antimicrobial resistance
 European Medicines Agency role and experience on antimicrobial resistance Regional Training Workshop on Antimicrobial Resistance (AMR) Responding to the global challenge of AMR threats: toward a one health
European Medicines Agency role and experience on antimicrobial resistance Regional Training Workshop on Antimicrobial Resistance (AMR) Responding to the global challenge of AMR threats: toward a one health
IMPORT HEALTH STANDARD FOR SHELF-STABLE PETFOODS CONTAINING ANIMAL PRODUCTS
 IMPORT HEALTH STANDARD FOR SHELF-STABLE PETFOODS CONTAINING ANIMAL PRODUCTS Issued pursuant to Section 24A of the Biosecurity Act 1993 Dated: 3 November 2014 12 May 2017 Under CTO Direction CTO 2017 029
IMPORT HEALTH STANDARD FOR SHELF-STABLE PETFOODS CONTAINING ANIMAL PRODUCTS Issued pursuant to Section 24A of the Biosecurity Act 1993 Dated: 3 November 2014 12 May 2017 Under CTO Direction CTO 2017 029
Franck Berthe Head of Animal Health and Welfare Unit (AHAW)
 EFSA s information meeting: identification of welfare indicators for monitoring procedures at slaughterhouses Parma, 30/01/2013 The role of EFSA in Animal Welfare Activities of the AHAW Unit Franck Berthe
EFSA s information meeting: identification of welfare indicators for monitoring procedures at slaughterhouses Parma, 30/01/2013 The role of EFSA in Animal Welfare Activities of the AHAW Unit Franck Berthe
EUROPEAN COMMISSION. General guidance on EU import and transit rules for live animals and animal products from third countries
 EUROPEAN COMMISSION General guidance on EU import and transit rules for live animals and animal products from third countries EUROPEAN COMMISSION General guidance on EU import and transit rules for live
EUROPEAN COMMISSION General guidance on EU import and transit rules for live animals and animal products from third countries EUROPEAN COMMISSION General guidance on EU import and transit rules for live
Minutes of the Scientific Steering Committee Meeting of September 2002
 Minutes of the Scientific Steering Committee Meeting of 12-13 September 2002 1. Welcome, apologies, introductory remarks and declaration of interest. The chairman Prof.G.Pascal welcomed the participants.
Minutes of the Scientific Steering Committee Meeting of 12-13 September 2002 1. Welcome, apologies, introductory remarks and declaration of interest. The chairman Prof.G.Pascal welcomed the participants.
FINAL REPORT OF THE INVESTIGATION INTO THE NORTH LEI...RSHIRE CLUSTER OF VARIANT CREUTZFELDT-JAKOB DISEASE
 SUMMARY OF THE FINAL REPORT OF THE INVESTIGATION INTO THE NORTH LEICESTERSHIRE CLUSTER OF VARIANT CREUTZFELDT-JAKOB DISEASE The investigation was carried out by Dr Gerry Bryant and Dr Philip Monk who have
SUMMARY OF THE FINAL REPORT OF THE INVESTIGATION INTO THE NORTH LEICESTERSHIRE CLUSTER OF VARIANT CREUTZFELDT-JAKOB DISEASE The investigation was carried out by Dr Gerry Bryant and Dr Philip Monk who have
Standard requirements for the submission of programmes of eradication and monitoring of TSE
 Standard requirements for the submission of programmes of eradication and monitoring of TSE Member States seeking a financial contribution from the Community for national programmes for the control and
Standard requirements for the submission of programmes of eradication and monitoring of TSE Member States seeking a financial contribution from the Community for national programmes for the control and
EUROPEAN COMMISSION DIRECTORATE-GENERAL FOR HEALTH AND FOOD SAFETY
 Ref. Ares(2016)105284-08/01/2016 EUROPEAN COMMISSION DIRECTORATE-GENERAL FOR HEALTH AND FOOD SAFETY Directorate F - Food and Veterinary Office DG(SANTE) 2015-7426 - MR FINAL REPORT OF AN AUDIT CARRIED
Ref. Ares(2016)105284-08/01/2016 EUROPEAN COMMISSION DIRECTORATE-GENERAL FOR HEALTH AND FOOD SAFETY Directorate F - Food and Veterinary Office DG(SANTE) 2015-7426 - MR FINAL REPORT OF AN AUDIT CARRIED
EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL
 EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL Directorate F - Food and Veterinary Office DG(SANCO) 2009-8137 - MR FINAL FINAL REPORT OF A MISSION CARRIED OUT IN CHINA FROM 17 TO 27 MARCH
EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL Directorate F - Food and Veterinary Office DG(SANCO) 2009-8137 - MR FINAL FINAL REPORT OF A MISSION CARRIED OUT IN CHINA FROM 17 TO 27 MARCH
Updated Guidance on the practice of animal dissection in standard school science experiments
 EA Circular Number: 2018-0411-1A Date: 27 th April 2018 To all EA Post Primary School Science Departments Updated Guidance on the practice of animal dissection in standard school science experiments Background:
EA Circular Number: 2018-0411-1A Date: 27 th April 2018 To all EA Post Primary School Science Departments Updated Guidance on the practice of animal dissection in standard school science experiments Background:
