The Paleontograph. A newsletter for those interested in all aspects of Paleontology Volume 1 Issue 1 January, 2012
|
|
|
- Dwight Ramsey
- 5 years ago
- Views:
Transcription
1 The Paleontograph A newsletter for those interested in all aspects of Paleontology Volume 1 Issue 1 January, 2012 From Your Editor So, here we are at the beginning of something. I think that it would be good to tell you how this venture came about. I ve edited The NJPS Paleontograph for sixteen years. NJPS, being the New Jersey Paleontological Society. Like many gem, mineral or fossil clubs, NJPS has started to fade away. There is a lack of members willing to step forward and do work for the group. It became a club that had a great newsletter but nothing else, no programs, field trips or activities. I grew tired of the situation and tried to get people to become more active but that was not to be, so I resigned. I don t know what will happen to the club, some are trying to keep it going. But I enjoyed putting out the newsletter and felt proud of it. I was blessed with one main, very prolific, contributor, Bob Sheridan, who made me the envy of all other newsletter editors that knew my newsletter. Unlike other newsletter editors, I never had to write articles myself, in order to fill my newsletter. It turns out that Bob feels the same way I do about the whole thing. So that brings us to here, today. First, the name. I like it and so I did a spin off from the old name. Not quite stealing it but very close. If someone complains, maybe I ll change it but for now this is it. I will distribute this newsletter to the former members of NJPS as well as my former complimentary subscribers. I also went out to the Paleolist, from which I received some subscribers as well as my first new contributor. I ll stop now. Please let me know what you think. I hope you all enjoy this new thing, we will see where it goes. It will be up to you. The Paleontograph was created in 2012 to continue what was originally the newsletter of The New Jersey Paleontological Society. The Paleontograph publishes articles, book reviews, personal accounts, and anything else that relates to Paleontology and fossils. Feel free to submit both technical and non-technical work. We try to appeal to a wide range of people interested in fossils. Articles about localities, specific types of fossils, fossil preparation, shows or events, museum displays, field trips, websites are all welcome. This newsletter is meant to be one by and for the readers. Issues will come out when there is enough content to fill an issue. I encourage all to submit contributions. It will be interesting, informative and fun to read. It can become whatever the readers and contributors want it to be, so it will be a work in progress. TC, January 2012 Edited by Tom Caggiano and distributed at no charge Tomcagg@aol.com
2 PALEONTOGRAPH Volume 1 Issue 01\2012 Page 1 The Stability of Jaw Anatomy in Devonian Fish Bob Sheridan August 20, 2011 Fish with jaws (gnathosomes) appeared first in the Silurian, and by the end of the Devonian almost all fish were jawed. How different types of jaws arose among different types of fish is the subject of study by Anderson et al. (2011). They looked at 31 mandibular traits of 198 gnathosomes from the Silurian and Devonian divided into 8 time bins spanning ~420 Myr. to ~360 Myr. The gnathosomes can be divided into five kinds of fish: Acanthodii ("spiny sharks"), Actinopterygii (ray-finned fish-- which are the most common type today), Chondrichthyes (cartilaginous fish, i.e. sharks), Placoderms (armored fish), and Sarcopterygii (lobefinned fish). First, examining assemblages of vertebrate fossils, these authors find that in the Upper Silurian, 0-50% of the animals were gnathosomes, depending on the assemblage. In the Early Lower Devonian, assemblages may contain anywhere from 0-100% gnathosomes. By the Middle Devonian assemblages contain % gnathosomes. By the Late Upper Devonian, all the assemblages contain nearly 100% gnathosomes. This is where we still are today, although the non-gnathosomes have changed. In the Devonian, almost all non-gnathosomes were Ostracoderms, armored jawless fish, but today the only jawless fish are lampreys and hagfish. Here we need an aside about "disparity," which has to do, in this case, with how different the different genera are from each other in the 31 mandibular traits. These authors use non-metric multidimensional scaling to project the mandibular traits into a two dimensional "morpho-space". (Two is a useful dimension because it can be displayed on paper.) Distance in this morpho-space is a measure of disparity. If the fish are spread out in morphospace, that means there is more anatomic variety. (There is usually an assumption that anatomic variety implies functional variety as well; however we usually do not have a handle on function in extinct animals.) Anderson et al. find that disparity started out low in the Upper Silurian, peaked in the Late Lower Devonian, and then settled into a low disparity after the Middle Devonian. Interestingly, which group of fish contribute to the disparity changes with time. In the Late Silurian, almost all the variation was within the Acanthodii. At the end of the Devonian, most of the disparity is from the Sarcopterygii and Placoderms. The jaws of Condrichthyes and Actinopterigyii never varied very much. The authors make two points: Since jawed and jawless fish coexisted for millions of years even after the disparity in jawed fishes had peaked, this argues against jawed fish replacing jawless fish in specific ecological niches because they are somehow superior. It is more likely gnathosomes were able to exploit lifestyles not available to jawless fish, which were probably confined to bottom-feeding. The disparity of jaws is constant during the parts of the Devonian where there were a number of environmental upheavals: sea level changes, an extinction event (~375 Myr.), and increase in oxygen (~400 Myr). Anderson, P.S.L.; Friedman, M.; Brazeau, M.D.; Rayfeld, E.J. "Initial radiation of jaws demonstrated stability despite faunal and environmental change." Nature 2011, 476, Wooly Before the Ice Age Bob Sheridan September 5, 2011 There are two types of animals we know of that came in a "wooly" version in the Ice Age whereas the modern version is essentially hairless: elephants (as represented by the mammoth) and rhinos (as represented by the wooly rhino). This story is about the wooly rhino (genus Coelodonta--"cavity tooth"), which is best know from its Pleistocene fossils in northern Europe and Asia. The wooly rhino went extinct with other megafauna ~12,000 years ago. Aside from being wooly, there are several differences between Coelodonta and modern rhinos: The skull is especially long and low. The limbs are stocky. The two nasal horns are especially long, pointed forward, and narrow from side to side. Cont d
3 PALEONTOGRAPH Volume 1 Issue 01\2012 Page 2 Wooly Cont d Since rhino horns are not made of bone, they are not usually preserved; hair is not usually preserved either. However, as with mammoths, there have been a few specimens mummified in permafrost or tarpits. These have horn and wool intact. Also as with mammoths, there are many cave paintings that record the appearance of the living animal. One suggestion as to why the wooly rhino horn is long, narrow, and pointed forward is that it was used to scrape snow away so the rhino could browse on ground-level plants. (A similar suggestion is made for the extra-long tusks of the mammoth.) This week Deng et al. (2010) describe a new wellpreserved wooly mammoth skull and mandible for a new species they call Coelodonta thibetana. The skull is about three feet long. This specimen is unusual in two respects: It is found in the Zanda Formation in southern Tibet at the foothills of the Himalayas. Most specimens are found in northern Asia. It is 3.7 Myr. old (middle Pliocene), whereas most specimens are from the Pleistocene (less than 0.4 Myr.). Unfortunately, the horn and wool are not preserved for this specimen, so one must go by the bone characters, and only for the skull. However, the nasal boss in the specimen is very large and is consistent with a flattened horn. Phylogenetic analysis of the new specimen with 15 other specimens of rhino, wooly and otherwise, shows that Coelodanta thibetana is most like the three other wooly rhino species, and the most primitive wooly rhino. Isotope analysis of the new rhino specimen, plus other large mammals living at the same time suggests they all fed on C3 vegetation and they lived in a high-altitude cold climate. The authors suggest that Coelodanta thibetana represents the first cold-adapted rhino, the cold being due to the altitude at which it lived, rather than the global climate. Being pre-adapted for cold, it was able to migrate to low-altitude parts of northern Asia and then to all of Eurasia when the Ice Ages came. Deng, T.; Wang,. X.; Fortelius, M.; Li, Q.; Wang, Y.; Tseng, Z.J.; Takeuchi, G.; Saylor, J.E.; Saila, L.K.; Xie, G. "Out of Tibet: Pliocene wooly rhino suggests a highplateau origin of ice age megaherbivores." Science 2011, 333, K-T Extinction of Birds Bob Sheridan September 18, 2011 Birds originated in the Late Jurassic (if you count Archaeopteryx as the first bird) and diversified in the Cretaceous. (Most well-preserved Early Cretaceous birds are found in China.) In the Cretaceous there were two major classes of birds: Enantiornithes and Ornithurae which are distinguished by how the ankle and foot bones are fused. The Enantiornithes were the most abundant and diverse, but only the Ornithurae survive to the present day. The families of modern birds definitely existed in the Paleocene. Since data is scarce, two scenarios for the later evolution of birds have remained plausible: Modern bird families existed in the Cretaceous and most survived the K-T extinction. Almost all birds were wiped out at the K-T and the modern families arose later from the few survivors. One way of addressing this is to see if "archaic" birds, i.e. not in modern families, survived to the end of the Cretaceous. Longrich et al. (2011) examined bird fauna fossils from the Maastrichtian (the latest 1.5 Myr. of the Cretaceous). These are from the Hell Creek Formation (Montana), Lance Formation (Wyoming), and the Frenchman Formation (Salskatchewan). The main approach is to generate a phylogenetic analysis of the species existing at that time. Interestingly, these authors confined their analysis mostly to characteristics of the coracoid bone. In mammals, the coracoid is part of our shoulder blade, but in birds, the coracoid is a long bone. The humerus articulates at one end of the coracoid, and the other end braces against the sternum. The clavicle (in birds called the furcula--or wishbone) also articulates with the coracoid. Apparently the coracoid is very distinctive for different types of birds. The authors also include some data about the tarsometatarsi bones for which the coracoid is not preserved in this set of fossil. In birds the ankle bones (tarsi) and foot bones (metatarsi) are fused, and these also differ among birds, especially between enantiornithes and ornithurines, as stated above. The authors identified 17 types of coracoid, 15 of which could be assigned to a previously known fossil species. There are a number of enantiornithines and ornithurines. The coracoids show a reasonable range of bird sizes, from 200 grams to 5 kg. Cont d
4 PALEONTOGRAPH Volume 1 Issue 01\2012 Page 3 Bird Extinction Cont d This is somewhat narrower than the range of modern birds, and the difference is probably due to the small number of samples, but it does suggest birds already filled a variety of ecological roles. We know from this sample that archaic birds such as enantiornithines survived until the end of the Cretaceous, although they were not as abundant as ornithurines at that time. Earlier work from the authors showed that no enanthiornithines survived to the Paleocene. Also, there are no modern families of birds represented in this sample. Overall the data is consistent with a model where birds were diversified in the Mesozoic, almost all were wiped out at the K-T extinction, and the few survivors diversified into modern bird families. Longrich, N.R.; Tokaryk, T.; Field, D.J. "Mass extinction of birds at the Cretaceous- Paleogene (K-Pg) boundary." Proc. Natl. Acad. Sci. USA 2011, 108, Crops in Fossil Birds Bob Sheridan September 24, 2011 First a quick review of the unique digestive system of birds. Many birds have a crop, which is a sack-like extension of the esophagus. This usually is at the base of the throat in front of the furcula (wishbone) and outside the ribcage. Below the stomach, and inside the ribcage, is the gizzard, which is a very muscular organ that may incorporate gravel (or gastroliths). The crop provides a way to gather food quickly so that it can be digested at leisure at a safer location. The gizzard provides a way of grinding harder foods (since teeth are absent in birds). Not surprisingly, the presence of a gizzard and crop are usually associated with a diet of seeds in living birds. There is much fossil evidence for gizzards in birds and in dinosaurs since gizzards contain stones, and there have been fossil birds with seeds among the stomach contents, for example in Jeholornis. However, evidence for crops in fossil birds is harder to come by. Zheng et al. (2011), after examining hundreds of bird fossils at the Tianyu Museum of Nature, report the presence of a crop in two types of birds from the Early Cretaceous of China. Sapeornis chaoyangensis ( bird from Chaoyang ) is a basal bird with a beak but with a few premaxillary teeth. Two specimens of Sapeornis are preserved with a circular mass in font of the throat, which is packed with seeds. One specimen shows a mass of polished gastroliths in the ribcage where the gizzard ought to be. Hongshanornis longicresta ( bird of the Hongshan culture ) is a basal ornithurine (which means it is in the class of modern birds) with a toothless beak and long legs. One specimen has a seed mass in front of the furcula and also has gastroliths in the ribcage. The authors suggest that the earliest birds were insectivores and the presence of teeth and a long snout was necessary to handle that diet. They further suggest that a diet of seeds may have given rise to the presence of crop and gizzard, which in turn may have led to the loss of teeth. Since Sapeornis and Hongshanornis are not closely related, the gizzard and crop may have evolved several times in birds. Whether crop and gizzard always go together and whether they are associated with seed eating is not clear because the crop is seldom preserved. The authors point out that of nearly 100 Sapeornis specimens they examined, only the two mentioned here show evidence of a crop. Also Jeholornis, which is a known seed-eater based on stomach contents, shows no evidence of a crop in more than 100 specimens. Zheng, X.; Martin, L.D.; Zhou, Z.; Burnham, D.A.; Zhang, F.; Miao, D. Fossil evidence of avian crops from the Early Cretaceous of China. Proc. Natl. Acad. Sci. USA 2011, 108, Hongshanornis Hongshanornis From Wikipedia Pavel Riha
5 PALEONTOGRAPH Volume 1 Issue 01\2012 Page 4 Mosasaurs Kenneth Quinn mosasaur47@msn.com Mosasaurs were true reptiles that first appeared in the Cretaceous and disappeared at the very end of that period. It was long thought that their closest living relatives were the monitor lizards such as the Komodo Dragon, but recent research indicates that snakes are instead closer. There were a number of genera of mosasaurs and they showed a wide range of ecological slots; one, Globidens, had teeth designed to crush shellfish. There is indirect evidence that some kinds preyed on the squidlike ammonites - the bite marks on at least one ammonite shell exactly match the pattern of the teeth in a mosasaur jaw. In another genus of mosasaur, there are defects in the bones that are identical to those seen in the bones of whales that dive to great depths to seek their prey - giant squid! These defects are caused by the deep dives, so apparently these mosasaurs also sought their food at great depths. The name of these reptiles comes from the area where they were first found - the valley of the Meuse River. To be precise, the first one discovered was found in a quarry near Maastricht, a Dutch city, in My own experience with mosasaurs began on November 18, 1969; that date is easy to remember because it was my first wedding anniversary. I asked my wife what she wanted to do and she said - bless her! - "Let's go fossil hunting!" At that time I was still an undergraduate geology student at the University of Alabama and it was only an hour's drive to outcrops of the Mooreville Chalk, the northernmost formation of the chalky Cretaceous deposits of southern Alabama. We first tried an outcrop we had visited before but we found very few worthwhile fossils, so we started cruising rural roads for other outcrops. Near the tiny community of West Greene, we spotted roadside exposures and got out to look at them. At first, we saw very little to get excited about. My wife's method was to sit down in one spot and search it thoroughly, while mine was to roam the whole outcrop, so I started walking down a narrow strip right along the edge of the road. About 25 yards away from her, I looked down and started screaming! She looked up, thinking I had fallen and broken something. Instead, I had spotted dark brown bone - I had seen scraps of bone in this formation before, but not this big, and not several connected pieces! We started digging, and more of the animal emerged. By the time we were finished, we had about ten feet of reptilian skeleton exposed; missing were the tail and the left hind leg. At that time I had no knowledge of enclosing skeletons in a plaster of Paris jacket, so we just picked up the thoroughly mineralized bones. The owner of the adjacent private land had in the meantime driven by and I got permission to cross the fence and look for more fossils on his property but found nothing, not even a clam shell! Still, that was a great way to celebrate an anniversary! Since then, I have found fragments of mosasaurs in Nebraska and Arkansas but no articulated bones. Nevertheless, these are no doubt the most common reptilian marine remains in Late Cretaceous deposits and anyone who looks for fossils in such deposits has a god chance of finding at least a piece of bone or one of their conical, curved teeth. Articulated skeletons - even partial ones - are of course much rarer, and should be reported to a reputable museum. I know, I know - but I should have! And I did donate all but a very few bones of my discovery to someone doing research on mosasaurs. My mosasaur was in the genus Clidastes. This was no big surprise - it was perhaps the most common genus of mosasaur. However, there are currently 27 genera of mosasaurs! They ranged in size from 10 feet to 57 feet, and were almost worldwide in distribution. Fossils of them have been found in Europe, both Americas, Africa, Australia, New Zealand, and an island off the coast of Antarctica. I am not aware of any having been found on mainland Asia. Ed. Note For all you ever wanted to know about Mosasaurs, go to Mike Everhart s great website at:
6 PALEONTOGRAPH Volume 1 Issue 01\2012 Page 5 Trace Copper as a Marker for Melanin in Fossil Feathers Bob Sheridan October 1, 2011 This story needs three bits of background. One is about x-ray spectroscopy. Three varieties are important here, with the acronyms EXAFS, XANES, and SRS-XRF. XANES measures the absorbance of x-rays in a material as a function of the wavelength of the x-ray--very much like visible spectroscopy. EXAFS measures the energy of scattered electrons ejected from the atoms in the material by the x-rays. In EXAFS the energy of the scattered electrons is a measure of the distance of other atoms to the atoms doing the absorbance. SRS-XRF is a type of x-ray fluorescence, where atoms absorbing x-rays emit visible light and one measures the visible light as a function of wavelength. Typically metals or other heavy elements are the ones that absorb x-rays. One has to have a strong source of x-rays to do x- ray spectroscopy, and this usually requires a synchotron, a large expensive piece of equipment. The other story has to do with melanosomes. Modern feathers contain micrometer-size bodies called melanosomes that are embedded in the keratin. Melanosomes contain the pigment melanin, of which there are many varieties. The color of the pigment seems to be associated with the shape of the melanosomes: rod shapes (eumelanosomes) tend to contain black or grey pigment, more spherical shapes (pheomelanosomes) contain red or brown pigments. There have been at least two reports in the literature that feathers in fossil birds or dinobirds (from China) contain melanosomes, and the shape of the melanosomes in the fossils has been used to infer the color of those extinct animals. Also relevant to this story is the fact that melanin is synthesized by the copper-containing enzyme tyrosinase, and copper atoms are incorporated into melanin as it is synthesized. Wogelius et al. (2011) use all three types of x-ray spectroscopy to examine the feathers in a specimen of Confusciusornis, an early true bird from Early Cretaceous China. (Earlier the same lab used SRS- XRF to study the feathers of Archaeopteryx.) SRS- XRS shows that the downy feathers and some of the wing feathers closest to the body (but not the flight feathers at the tips of the wings) of Confusciusornis fluoresce when exposed to x-rays of the wavelength absorbed by copper. The inference is that the copper signature reflects the presence of copper incorporated into melanin; the incorporation is probably in the form of a metal-chelation, much as the heme in hemoglobin binds iron. The following experiments from the same paper support this claim: The EXAFS and XANES spectra of the downy feathers are more consistent with modern coppercontaining melanin than inorganic sources of copper like copper oxide. Eumelanosomes can be detected in the fossil feathers by scanning electron microscopy in the same places as the copper is detected. The copper signature can be seen in the darker parts of a fossil feather and eye of a fossil fish from Green River. The same thing is seen in a fossil squid. The copper signature can be seen in the colored parts of two modern feathers from an eagle and a blue jay. Similarly with an extant squid. The authors infer that Confusciusornis was probably dark colored close to the body but most of the wings were white. Also they suggest that copper detected by x-ray spectroscopy can be a marker for melanin in fossils where melanosomes themselves are not preserved. Wogelius, R.A.; Manning, P.L.; Barden, H.E.; Edwards, N.P.; Webb, S.M.; Sellers, W.I.; Taylor, K.G.; Larson, P.L.; Dodson, P.; You, H.; Da-qing, L.; Bergmann, U. "Trace metals as biomarkers for eumelanin pigments in the fossil record." Science 201, 333, Other Chemical Evidence for Melanin in Fossil Feathers Bob Sheridan October 22, 2011 The last story I wrote described work by Wogelius et al. (2011). These authors used a variety of x-ray spectroscopic methods to link the presence of copper in fossil feathers with the presence of the pigment melanin (of which "eumelanin" is a subtype). Cont d
7 PALEONTOGRAPH Volume 1 Issue 01\2012 Page 6 Melanin Cont d This was done for the Early Cretaceous bird Confusciusornis, plus some modern feathers and squid. In case you don't have that article handy, here is the recap: Modern feathers contain micrometersize bodies called melanosomes that are embedded in the keratin. Melanosomes contain the pigment melanin, of which there are many varieties. The color of the pigment seems to be associated with the shape of the melanosomes: rod shapes (eumelanosomes) tend to contain black or grey pigment, more spherical shapes (pheomelanosomes) contain red or brown pigments. There have been at least two reports in the literature that feathers in fossil birds or dinobirds (from China) contain melanosomes, and the shape of the melanosomes in the fossils has been used to infer the color of those extinct animals. However, the problem with "melanosomes" in fossil feathers is that it is hard to rule out that they might be other types of round or elongated shapes a few microns in size, like modern bacteria. This week I came across another study from the same laboratory (Barden et al., 2011) that extends the work with other forms of spectroscopy: energy dispersive x-ray spectroscopy (EDS), Fourier-transform infrared (FTIR) spectroscopy, electron paramagnetic resonance (PIR), and pyrolysis gas chromatography/mass spectroscopy (py-gcms). These were applied to the fossil feathers of two specimens of another Early Cretaceous bird from China called Gansus, and also applied to feathers from two modern birds: a black feather from a Maribou stork and a white feather from a White-napped crane. EDS is much like scanning-electron microscopy, i.e. forms images of small objects, but one may also analyze the objects being scanned for their elemental composition. Melanosomes were seen in the dark parts of the fossil feather, and in the black modern feather, and not in the white modern feathers. Elemental composition of the modern feathers shows the presence of carbon, oxygen, and sulfur (which is not a surprise), but there is more calcium in the dark feather. Composition of the fossil feathers shows more carbon in the dark areas. Elemental composition of the fossil matrix is silicon and oxygen, again not surprising for rock. The interpretation of the elemental composition in terms of relating modern to fossil feathers is unclear, but it is clear that the original organic material is present in the fossil fossil. Infrared spectroscopy measures the absorbance of infrared light by a material, and one may see the bond stretch frequencies of common chemical groups. The bottom line here is that the IR spectrum of the dark part of fossil feathers looks something like that of isolated melanin, and something like that a modern dark feather. The match is fairly vague, in my opinion, but it does again suggest that the fossil feathers are organic. There is no evidence for the chemical bonds expected for modern bacterial biofilms. EPR reveals the environment of unpaired electrons. The EPR spectrum of the dark part of the fossil feathers, the black modern feather, and isolated melanin look about the same. The white modern feather has very little EPR signal. This is consistent with melanin being present in the fossil feathers. py-gcms uses heat to break molecules into fragments, whereupon the fragments are separated by molecular weight by mass spectroscopy. Interpretation of this, again, is not straightforward. However, one can be sure that the mass spectrum of the fossil feathers is different from that of the surrounding matrix. The studies in Barden et al. are on the borderline of being doable, and harder to interpret than the those in Wogelius et al.. This is not surprising since these authors are trying to chemically analyze a very degraded, very thin film on a rock. The best one can say is that it appears that fossil feathers are not just impressions in the rock, but contain original feather material, including melanin. Barden, H.E.; Wogelius, R.A.; Li, D.; Manning, P.L.; Edwards, N.P.; van Dongen, B.E. "Morphological and geochemical evidence of eumelanin preservation in the feathers in the Early Cretaceous bird Gansus yumenensis." PLoS ONE 2011, 6, e Wogelius, R.A.; Manning, P.L.; Barden, H.E.; Edwards, N.P.; Webb, S.M.; Sellers, W.I.; Taylor, K.G.; Larson, P.L.; Dodson, P.; You, H.; Da-qing, L.; Bergmann, U. "Trace metals as biomarkers for eumelanin pigments in the fossil record." Science 201, 333,
8 PALEONTOGRAPH Volume 1 Issue 01\2012 Page 7 Triceratops to Nedoceratops to Torosaurus? Bob Sheridan January 7, 2012 For most dinosaurs we have only few specimens. That makes it hard to guess the normal variation between members of a species, and it is therefore very easy to interpret a juvenile or a female, or an individual with some minor variation, as a different species or even a different genus. Even up to the middle of the 20th Century it was common for new dinosaur species to be named based on small differences. For instance, the animal in today's topic, Triceratops, has had 16 species assigned to it. Based on the variation of anatomy in Triceratops, for which many specimens are known, It was suggested by Ostrom and Wellnhoffer in 1990 that there were at most one or two Triceratops species. Some workers, currently Jack Horner being the strongest proponent, look for ways to consolidate species based on growth stages. This becomes possible once one can estimate age of individual specimens based on bone histology. For example, he feels that among the bone-head dinosaurs Dracorex, Stygimoloch, and Pachycephalosaurus represent increasing old specimens of the same animal. To work out such a relationship one must show that there is a smooth transition in anatomy between the animals (making it plausible that one animal can "grow" into another) and the sizes and ages of the animals are consistent. Of course, if the number of specimens is small, it is possible to go overboard in this direction and falsely lump together different species. For example, if the few specimens you have of animal A happen to be young and the few of animal B happen to be old, you might be tempted to say A is the juvenile form of B although the age differences could be due purely to chance. Today's story deals with two horned dinosaurs, the familiar Triceratops, and the less familiar Torosaurus. They both are large, have two long brow horns, a short nose horn, and short horns on the cheek bones. The major difference is in the frill. Triceratops has a shorter solid frill, whereas Torosaurus has a longer frill with large openings (or fenestrae). The brow horns of Triceratops point slightly upward while those of Torosaurus point forward. In 1990 Ostrom and Wellnhoffer suggested that Torosaurus could be a "male" Triceratops. However, in 2010 Scannella and Horner (Museum of the Rockies) suggested instead that Torosaurus represents the oldest Triceratops. This is based on three points: 1. Triceratops has a thinning of the frill in the locations where Torosaurus has fenestrae, i.e. there is plausible transition between species. 2.The bone histology age of known Torosaurus specimens is high. 3.In Triceratops that the brow horns tend to point more forward in the larger (presumably older) animals, consistent with the forward pointing horns of Torosaurus. Early in 2011 Scannella and Horner joined another similar animal Nedoceratops (previously known as Diceratops) into this series. There is only one specimen of Nedoceratops (USNM 2412), but it makes a reasonable intermediate between Triceratops and Torosaurus. It has small fenestrae in the frill at the same place as the large fenestrae of Torosaurus. However, another worker Andrew Farke (Raymond M. Alf Museum of Paleontology) argued that Nedoceratops was not equivalent to Triceratops because it lacked a nose horn and the single specimen was already very old. Also, Nedoceratops has few episquamosals (bony growths at the margin of the frill) like Triceratops and not like Torosaurus. A new paper by Scannella and Horner (2011) rebuts the points from Farke. In particular, the paper notes that the number of squamosals and presence of a nose horn core varies so much among Triceratops, and that the preservation of these features is also variable, so that those features cannot be used to rule out a relationship of Nedoceratops and Triceratops. This is once again a case of taking some ambiguous evidence from few specimens and arguing about which side has the burden of proof, the side of "splitters" (more species) or that of "lumpers" (fewer species). My personal prejudice is to apply Occam's Razor in ambiguous cases: don't assume more species than there is clear evidence for. Scannella, J.B.; Horner J.R. "'Nedoceratops': and example of a transitional morphology." PLoS ONE 2011, 6, e28705
Origin and Evolution of Birds. Read: Chapters 1-3 in Gill but limited review of systematics
 Origin and Evolution of Birds Read: Chapters 1-3 in Gill but limited review of systematics Review of Taxonomy Kingdom: Animalia Phylum: Chordata Subphylum: Vertebrata Class: Aves Characteristics: wings,
Origin and Evolution of Birds Read: Chapters 1-3 in Gill but limited review of systematics Review of Taxonomy Kingdom: Animalia Phylum: Chordata Subphylum: Vertebrata Class: Aves Characteristics: wings,
From Reptiles to Aves
 First Vertebrates From Reptiles to Aves Evolutions of Fish to Amphibians Evolution of Amphibians to Reptiles Evolution of Reptiles to Dinosaurs to Birds Common Ancestor of Birds and Reptiles: Thecodonts
First Vertebrates From Reptiles to Aves Evolutions of Fish to Amphibians Evolution of Amphibians to Reptiles Evolution of Reptiles to Dinosaurs to Birds Common Ancestor of Birds and Reptiles: Thecodonts
Living Dinosaurs (3-5) Animal Demonstrations
 Living Dinosaurs (3-5) Animal Demonstrations At a glance Students visiting the zoo will be introduced to live animals and understand their connection to a common ancestor, dinosaurs. Time requirement One
Living Dinosaurs (3-5) Animal Demonstrations At a glance Students visiting the zoo will be introduced to live animals and understand their connection to a common ancestor, dinosaurs. Time requirement One
Dinosaurs and Dinosaur National Monument
 Page 1 of 6 Dinosaurs and Dinosaur National Monument The Douglass Quarry History of Earl's Excavation... Geology of the Quarry Rock Formations and Ages... Dinosaur National Monument protects a large deposit
Page 1 of 6 Dinosaurs and Dinosaur National Monument The Douglass Quarry History of Earl's Excavation... Geology of the Quarry Rock Formations and Ages... Dinosaur National Monument protects a large deposit
MOR CHANGE TEACHERS. TRICERATOPS GROWTH Activity Overview BIG IDEA
 MOR CHANGE 10 TRICERATOPS GROWTH Activity Overview BIG IDEA Triceratops, like other dinosaurs, changed in appearance as they grew up. As babies, their horns pointed backward, then shifted as they grew
MOR CHANGE 10 TRICERATOPS GROWTH Activity Overview BIG IDEA Triceratops, like other dinosaurs, changed in appearance as they grew up. As babies, their horns pointed backward, then shifted as they grew
Origin and Evolution of Birds. Read: Chapters 1-3 in Gill but limited review of systematics
 Origin and Evolution of Birds Read: Chapters 1-3 in Gill but limited review of systematics Review of Taxonomy Kingdom: Animalia Phylum: Chordata Subphylum: Vertebrata Class: Aves Characteristics: wings,
Origin and Evolution of Birds Read: Chapters 1-3 in Gill but limited review of systematics Review of Taxonomy Kingdom: Animalia Phylum: Chordata Subphylum: Vertebrata Class: Aves Characteristics: wings,
When Dinosaurs Ruled the Earth
 Buffalo Geosciences Program: Lesson Plan #2 When Dinosaurs Ruled the Earth Objectives: By the end of the program, the participants should be able to understand the earth and its creatures during the Triassic,
Buffalo Geosciences Program: Lesson Plan #2 When Dinosaurs Ruled the Earth Objectives: By the end of the program, the participants should be able to understand the earth and its creatures during the Triassic,
Red Eared Slider Secrets. Although Most Red-Eared Sliders Can Live Up to Years, Most WILL NOT Survive Two Years!
 Although Most Red-Eared Sliders Can Live Up to 45-60 Years, Most WILL NOT Survive Two Years! Chris Johnson 2014 2 Red Eared Slider Secrets Although Most Red-Eared Sliders Can Live Up to 45-60 Years, Most
Although Most Red-Eared Sliders Can Live Up to 45-60 Years, Most WILL NOT Survive Two Years! Chris Johnson 2014 2 Red Eared Slider Secrets Although Most Red-Eared Sliders Can Live Up to 45-60 Years, Most
Anatomy. Name Section. The Vertebrate Skeleton
 Name Section Anatomy The Vertebrate Skeleton Vertebrate paleontologists get most of their knowledge about past organisms from skeletal remains. Skeletons are useful for gleaning information about an organism
Name Section Anatomy The Vertebrate Skeleton Vertebrate paleontologists get most of their knowledge about past organisms from skeletal remains. Skeletons are useful for gleaning information about an organism
VERTEBRATE READING. Fishes
 VERTEBRATE READING Fishes The first vertebrates to become a widespread, predominant life form on earth were fishes. Prior to this, only invertebrates, such as mollusks, worms and squid-like animals, would
VERTEBRATE READING Fishes The first vertebrates to become a widespread, predominant life form on earth were fishes. Prior to this, only invertebrates, such as mollusks, worms and squid-like animals, would
The Fossil Record of Vertebrate Transitions
 The Fossil Record of Vertebrate Transitions The Fossil Evidence of Evolution 1. Fossils show a pattern of change through geologic time of new species appearing in the fossil record that are similar to
The Fossil Record of Vertebrate Transitions The Fossil Evidence of Evolution 1. Fossils show a pattern of change through geologic time of new species appearing in the fossil record that are similar to
Vertebrate Evolution
 Vertebrate Evolution Torsten Bernhardt Redpath Museum, McGill University This teaching resource was made possible with funding from the PromoScience programme of NSERC. McGill University 2010 History of
Vertebrate Evolution Torsten Bernhardt Redpath Museum, McGill University This teaching resource was made possible with funding from the PromoScience programme of NSERC. McGill University 2010 History of
Evolution of Birds. Summary:
 Oregon State Standards OR Science 7.1, 7.2, 7.3, 7.3S.1, 7.3S.2 8.1, 8.2, 8.2L.1, 8.3, 8.3S.1, 8.3S.2 H.1, H.2, H.2L.4, H.2L.5, H.3, H.3S.1, H.3S.2, H.3S.3 Summary: Students create phylogenetic trees to
Oregon State Standards OR Science 7.1, 7.2, 7.3, 7.3S.1, 7.3S.2 8.1, 8.2, 8.2L.1, 8.3, 8.3S.1, 8.3S.2 H.1, H.2, H.2L.4, H.2L.5, H.3, H.3S.1, H.3S.2, H.3S.3 Summary: Students create phylogenetic trees to
Non-fiction: Sea Monsters. A new wave of fossils reveals the oceans prehistoric giants.
 Sea Monsters By Stephen Fraser A new wave of fossils reveals the oceans prehistoric giants. Way back when Tyrannosaurus rex shook the ground, another giant reptile lurked in the prehistoric oceans. A 50-foot
Sea Monsters By Stephen Fraser A new wave of fossils reveals the oceans prehistoric giants. Way back when Tyrannosaurus rex shook the ground, another giant reptile lurked in the prehistoric oceans. A 50-foot
Fossilized remains of cat-sized flying reptile found in British Columbia
 Fossilized remains of cat-sized flying reptile found in British Columbia By Washington Post, adapted by Newsela staff on 09.06.16 Word Count 768 An artist's impression of the small-bodied, Late Cretaceous
Fossilized remains of cat-sized flying reptile found in British Columbia By Washington Post, adapted by Newsela staff on 09.06.16 Word Count 768 An artist's impression of the small-bodied, Late Cretaceous
Evolution of Tetrapods
 Evolution of Tetrapods Amphibian-like creatures: The earliest tracks of a four-legged animal were found in Poland in 2010; they are Middle Devonian in age. Amphibians arose from sarcopterygians sometime
Evolution of Tetrapods Amphibian-like creatures: The earliest tracks of a four-legged animal were found in Poland in 2010; they are Middle Devonian in age. Amphibians arose from sarcopterygians sometime
Evolution as Fact. The figure below shows transitional fossils in the whale lineage.
 Evolution as Fact Evolution is a fact. Organisms descend from others with modification. Phylogeny, the lineage of ancestors and descendants, is the scientific term to Darwin's phrase "descent with modification."
Evolution as Fact Evolution is a fact. Organisms descend from others with modification. Phylogeny, the lineage of ancestors and descendants, is the scientific term to Darwin's phrase "descent with modification."
Life in the Paleozoic
 Life in the Paleozoic Ocean Planet & The Great Migration Paleozoic Late Middle Early 543-248 Myr P r e c a m b r i a n Eon P h a n e r o z o i c Proterozoic Archean Hadean Geologic Time Scale Era Period
Life in the Paleozoic Ocean Planet & The Great Migration Paleozoic Late Middle Early 543-248 Myr P r e c a m b r i a n Eon P h a n e r o z o i c Proterozoic Archean Hadean Geologic Time Scale Era Period
It came from N.J.: A prehistoric croc Scientists' rare find will go on display. Tom Avril INQUIRER STAFF WRITER
 January 14, 2006 Section: LOCAL Edition: CITY-D Page: A01 Philadelphia Inquirer, The (PA) It came from N.J.: A prehistoric croc Scientists' rare find will go on display. Tom Avril INQUIRER STAFF WRITER
January 14, 2006 Section: LOCAL Edition: CITY-D Page: A01 Philadelphia Inquirer, The (PA) It came from N.J.: A prehistoric croc Scientists' rare find will go on display. Tom Avril INQUIRER STAFF WRITER
Fishes, Amphibians, Reptiles
 Fishes, Amphibians, Reptiles Section 1: What is a Vertebrate? Characteristics of CHORDATES Most are Vertebrates (have a spinal cord) Some point in life cycle all chordates have: Notochord Nerve cord that
Fishes, Amphibians, Reptiles Section 1: What is a Vertebrate? Characteristics of CHORDATES Most are Vertebrates (have a spinal cord) Some point in life cycle all chordates have: Notochord Nerve cord that
Juehuaornis gen. nov.
 34 1 2015 3 GLOBAL GEOLOGY Vol. 34 No. 1 Mar. 2015 1004 5589 2015 01 0007 05 Juehuaornis gen. nov. 1 1 1 2 1. 110034 2. 110034 70% Juehuaornis zhangi gen. et sp. nov Q915. 4 A doi 10. 3969 /j. issn. 1004-5589.
34 1 2015 3 GLOBAL GEOLOGY Vol. 34 No. 1 Mar. 2015 1004 5589 2015 01 0007 05 Juehuaornis gen. nov. 1 1 1 2 1. 110034 2. 110034 70% Juehuaornis zhangi gen. et sp. nov Q915. 4 A doi 10. 3969 /j. issn. 1004-5589.
Vertebrate Structure and Function
 Vertebrate Structure and Function Part 1 - Comparing Structure and Function Classification of Vertebrates a. Phylum: Chordata Common Characteristics: Notochord, pharyngeal gill slits, hollow dorsal nerve
Vertebrate Structure and Function Part 1 - Comparing Structure and Function Classification of Vertebrates a. Phylum: Chordata Common Characteristics: Notochord, pharyngeal gill slits, hollow dorsal nerve
Animal Evolution The Chordates. Chapter 26 Part 2
 Animal Evolution The Chordates Chapter 26 Part 2 26.10 Birds The Feathered Ones Birds are the only animals with feathers Descendants of flying dinosaurs in which scales became modified as feathers Long
Animal Evolution The Chordates Chapter 26 Part 2 26.10 Birds The Feathered Ones Birds are the only animals with feathers Descendants of flying dinosaurs in which scales became modified as feathers Long
The Cretaceous Period
 The Cretaceous Period By Doug and Claudia Mann Illustrated by David Cobb Copyright 2007 www.fossils-facts-and-finds.com Mesozoic Era Triassic Jurassic Cretaceous The Cretaceous Period: Flowers Bloom For
The Cretaceous Period By Doug and Claudia Mann Illustrated by David Cobb Copyright 2007 www.fossils-facts-and-finds.com Mesozoic Era Triassic Jurassic Cretaceous The Cretaceous Period: Flowers Bloom For
Vertebrates. Vertebrates are animals that have a backbone and an endoskeleton.
 Vertebrates Vertebrates are animals that have a backbone and an endoskeleton. The backbone replaces the notochord and contains bones called vertebrae. An endoskeleton is an internal skeleton that protects
Vertebrates Vertebrates are animals that have a backbone and an endoskeleton. The backbone replaces the notochord and contains bones called vertebrae. An endoskeleton is an internal skeleton that protects
C O L O S S A L F I S H
 COLOSSAL FISH GIANT DEVONIAN ARMORED FISH SKULL Titanichthys Termieri Lower Femannian, Upper Devonian Tafilalt, Morocco The Titanichthys was an immense armored fish, part of the Arthrodire order that ruled
COLOSSAL FISH GIANT DEVONIAN ARMORED FISH SKULL Titanichthys Termieri Lower Femannian, Upper Devonian Tafilalt, Morocco The Titanichthys was an immense armored fish, part of the Arthrodire order that ruled
Page # Diversity of Arthropoda Crustacea Morphology. Diversity of Arthropoda. Diversity of Arthropoda. Diversity of Arthropoda. Arthropods, from last
 Arthropods, from last time Crustacea are the dominant marine arthropods Crustacea are the dominant marine arthropods any terrestrial crustaceans? Should we call them shellfish? sowbugs 2 3 Crustacea Morphology
Arthropods, from last time Crustacea are the dominant marine arthropods Crustacea are the dominant marine arthropods any terrestrial crustaceans? Should we call them shellfish? sowbugs 2 3 Crustacea Morphology
Barney to Big Bird: The Origin of Birds. Caudipteryx. The fuzzy raptor. Solnhofen Limestone, cont d
 Barney to Big Bird: The Origin of Birds Caudipteryx The fuzzy raptor The discovery of feathered dinosaurs in Liaoning, China, has excited the many paleontologists who suspected a direct link between dinosaurs
Barney to Big Bird: The Origin of Birds Caudipteryx The fuzzy raptor The discovery of feathered dinosaurs in Liaoning, China, has excited the many paleontologists who suspected a direct link between dinosaurs
Biology 1B Evolution Lecture 11 (March 19, 2010), Insights from the Fossil Record and Evo-Devo
 Biology 1B Evolution Lecture 11 (March 19, 2010), Insights from the Fossil Record and Evo-Devo Extinction Important points on extinction rates: Background rate of extinctions per million species per year:
Biology 1B Evolution Lecture 11 (March 19, 2010), Insights from the Fossil Record and Evo-Devo Extinction Important points on extinction rates: Background rate of extinctions per million species per year:
Tuesday, December 6, 11. Mesozoic Life
 Mesozoic Life Review of Paleozoic Transgression/regressions and Mountain building events during the paleoozoic act as driving force of evolution. regression of seas and continental uplift create variety
Mesozoic Life Review of Paleozoic Transgression/regressions and Mountain building events during the paleoozoic act as driving force of evolution. regression of seas and continental uplift create variety
CLIL READERS. Level headwords. Level headwords. Level 5. Level headwords. Level 6 1,200 headwords. Level headwords
 dino _5 cover_apeikonisi.qxp_cover Time 21/9/16 7:02 PM Page 1 Level 5 Level 1 300 headwords Level 2 450 headwords Level 3 600 headwords Level 4 800 headwords CLIL READERS ISBN 978-1-4715-3303-7 Level
dino _5 cover_apeikonisi.qxp_cover Time 21/9/16 7:02 PM Page 1 Level 5 Level 1 300 headwords Level 2 450 headwords Level 3 600 headwords Level 4 800 headwords CLIL READERS ISBN 978-1-4715-3303-7 Level
LABORATORY EXERCISE 6: CLADISTICS I
 Biology 4415/5415 Evolution LABORATORY EXERCISE 6: CLADISTICS I Take a group of organisms. Let s use five: a lungfish, a frog, a crocodile, a flamingo, and a human. How to reconstruct their relationships?
Biology 4415/5415 Evolution LABORATORY EXERCISE 6: CLADISTICS I Take a group of organisms. Let s use five: a lungfish, a frog, a crocodile, a flamingo, and a human. How to reconstruct their relationships?
Domesticated dogs descended from an ice age European wolf, study says
 Domesticated dogs descended from an ice age European wolf, study says By Los Angeles Times, adapted by Newsela staff on 11.22.13 Word Count 952 Chasing after a pheasant wing, these seven-week-old Labrador
Domesticated dogs descended from an ice age European wolf, study says By Los Angeles Times, adapted by Newsela staff on 11.22.13 Word Count 952 Chasing after a pheasant wing, these seven-week-old Labrador
What is the evidence for evolution?
 What is the evidence for evolution? 1. Geographic Distribution 2. Fossil Evidence & Transitional Species 3. Comparative Anatomy 1. Homologous Structures 2. Analogous Structures 3. Vestigial Structures
What is the evidence for evolution? 1. Geographic Distribution 2. Fossil Evidence & Transitional Species 3. Comparative Anatomy 1. Homologous Structures 2. Analogous Structures 3. Vestigial Structures
Get the other MEGA courses!
 www.thesimplehomeschool.com Simple Schooling BUGS MEGA course is ten weeks of all about bugs! This course grabs your student s attention and never lets go! Grades K-3 Get the other MEGA courses! Simple
www.thesimplehomeschool.com Simple Schooling BUGS MEGA course is ten weeks of all about bugs! This course grabs your student s attention and never lets go! Grades K-3 Get the other MEGA courses! Simple
Outline 17: Reptiles and Dinosaurs
 Outline 17: Reptiles and Dinosaurs Evolution of Reptiles The first reptiles appeared in the Mississippian. They evolved from amphibians, which first appeared in the Devonian. The evolutionary jump was
Outline 17: Reptiles and Dinosaurs Evolution of Reptiles The first reptiles appeared in the Mississippian. They evolved from amphibians, which first appeared in the Devonian. The evolutionary jump was
B D. C D) Devonian E F. A) Cambrian. B) Ordovician. C) Silurian. E) Carboniferous. F) Permian. Paleozoic Era
 Paleozoic Era A) Cambrian A B) Ordovician B D C) Silurian C D) Devonian E) Carboniferous F) Permian E F The Cambrian explosion refers to the sudden appearance of many species of animals in the fossil record.
Paleozoic Era A) Cambrian A B) Ordovician B D C) Silurian C D) Devonian E) Carboniferous F) Permian E F The Cambrian explosion refers to the sudden appearance of many species of animals in the fossil record.
2 nd Term Final. Revision Sheet. Students Name: Grade: 11 A/B. Subject: Biology. Teacher Signature. Page 1 of 11
 2 nd Term Final Revision Sheet Students Name: Grade: 11 A/B Subject: Biology Teacher Signature Page 1 of 11 Nour Al Maref International School Riyadh, Saudi Arabia Biology Worksheet (2 nd Term) Chapter-26
2 nd Term Final Revision Sheet Students Name: Grade: 11 A/B Subject: Biology Teacher Signature Page 1 of 11 Nour Al Maref International School Riyadh, Saudi Arabia Biology Worksheet (2 nd Term) Chapter-26
Evolution by Natural Selection
 Evolution by Natural Selection 225 Permian Seed Plants Flowering Plants Birds Land Plants Mammals Insects Reptiles Teleost Fish Amphibians Chordates Molluscs Arthropods Dinosaurs 180 Triassic Jawless Fish
Evolution by Natural Selection 225 Permian Seed Plants Flowering Plants Birds Land Plants Mammals Insects Reptiles Teleost Fish Amphibians Chordates Molluscs Arthropods Dinosaurs 180 Triassic Jawless Fish
Some Facts about... Amphibians
 Amphibians Amphibians are cold-blooded vertebrates that live part of their lives in water and part on land. Amphibians eggs are laid in water and they are born there. They begin their lives with gills
Amphibians Amphibians are cold-blooded vertebrates that live part of their lives in water and part on land. Amphibians eggs are laid in water and they are born there. They begin their lives with gills
Name Date Class. From the list below, choose the term that best completes each sentence.
 Name Date Class Structure and Function of Vertebrates Review and Reinforce Birds Understanding Main Ideas Answer the following questions. 1. What are four characteristics that all birds share? 2. What
Name Date Class Structure and Function of Vertebrates Review and Reinforce Birds Understanding Main Ideas Answer the following questions. 1. What are four characteristics that all birds share? 2. What
Sauropterygia. Lepidosauromorpha
 Sauropterygia Lepidosauromorpha ***cladogram of lepids*** Pachypleurosauridae Nothosauria Pliosauroidea Plesiosauroidea Mosasauridae Thalattosauriformes? Placodontia Pistosauridae Plesiosauria Sauropterygia
Sauropterygia Lepidosauromorpha ***cladogram of lepids*** Pachypleurosauridae Nothosauria Pliosauroidea Plesiosauroidea Mosasauridae Thalattosauriformes? Placodontia Pistosauridae Plesiosauria Sauropterygia
Class Reptilia Testudines Squamata Crocodilia Sphenodontia
 Class Reptilia Testudines (around 300 species Tortoises and Turtles) Squamata (around 7,900 species Snakes, Lizards and amphisbaenids) Crocodilia (around 23 species Alligators, Crocodiles, Caimans and
Class Reptilia Testudines (around 300 species Tortoises and Turtles) Squamata (around 7,900 species Snakes, Lizards and amphisbaenids) Crocodilia (around 23 species Alligators, Crocodiles, Caimans and
Fossils Test Holt 2016 Answer Key. Test Key
 Fossils Test Holt 2016 Answer Key Test Key Station #1 1. What is the genus of this specific specimen? Favosites. 2. What Class was this specimen? Anthozoa 3. What period on the geologic timescale did the
Fossils Test Holt 2016 Answer Key Test Key Station #1 1. What is the genus of this specific specimen? Favosites. 2. What Class was this specimen? Anthozoa 3. What period on the geologic timescale did the
Non-Dinosaurians of the Mesozoic
 Non-Dinosaurians of the Mesozoic Calling the Mesozoic the Age of Dinosaurs is actually not quite correct Not all reptiles of the Mesozoic were dinosaurs. Many reptiles (and other amniotes) have returned
Non-Dinosaurians of the Mesozoic Calling the Mesozoic the Age of Dinosaurs is actually not quite correct Not all reptiles of the Mesozoic were dinosaurs. Many reptiles (and other amniotes) have returned
HUMAN APPENDIX BATS & TROPICAL FLOWERS
 HUMAN APPENDIX In humans, the appendix is a short piece of tissue off the large intestine. It is not used by humans for digestive functions. In other mammals, like rabbits and deer, the cecum is a large
HUMAN APPENDIX In humans, the appendix is a short piece of tissue off the large intestine. It is not used by humans for digestive functions. In other mammals, like rabbits and deer, the cecum is a large
Reproduction in Seed Plants (pp )
 Structure and Function of Plants Reading/Notetaking Guide Reproduction in Seed Plants (pp. 388 397) This section gives examples of the group of seed plants known as gymnosperms and angiosperms and describes
Structure and Function of Plants Reading/Notetaking Guide Reproduction in Seed Plants (pp. 388 397) This section gives examples of the group of seed plants known as gymnosperms and angiosperms and describes
Today there are approximately 250 species of turtles and tortoises.
 I WHAT IS A TURTLE OR TORTOISE? Over 200 million years ago chelonians with fully formed shells appeared in the fossil record. Unlike modern species, they had teeth and could not withdraw into their shells.
I WHAT IS A TURTLE OR TORTOISE? Over 200 million years ago chelonians with fully formed shells appeared in the fossil record. Unlike modern species, they had teeth and could not withdraw into their shells.
Phylogeny of Animalia (overview)
 The Diversity of Animals 2 Chapter 23 Phylogeny of Animalia (overview) Key features of Chordates Phylum Chordata (the Chordates) includes both invertebrates and vertebrates that share (at some point in
The Diversity of Animals 2 Chapter 23 Phylogeny of Animalia (overview) Key features of Chordates Phylum Chordata (the Chordates) includes both invertebrates and vertebrates that share (at some point in
Early Birds: Early Birds: Fossils and Feathers A Reading A Z Leveled Y Benchmark Book Word Count: 1,240. Fossils and Feathers BENCHMARK Y
 Early Birds: Fossils and Feathers A Reading A Z Leveled Y Benchmark Book Word Count: 1,240 BENCHMARK Y Early Birds: Fossils and Feathers Written by Alfred J. Smuskiewicz Visit www.readinga-z.com for thousands
Early Birds: Fossils and Feathers A Reading A Z Leveled Y Benchmark Book Word Count: 1,240 BENCHMARK Y Early Birds: Fossils and Feathers Written by Alfred J. Smuskiewicz Visit www.readinga-z.com for thousands
DARWIN LECTURE SERIES..
 http://ky.aipg.org Mailing Address Kentucky Section AIPG P.O. Box 24690 Lexington, KY 40524-4690 2011 EXECUTIVE COMMITTEE President: Frank Ettensohn President-elect: Charlie Mason Vice President: Trent
http://ky.aipg.org Mailing Address Kentucky Section AIPG P.O. Box 24690 Lexington, KY 40524-4690 2011 EXECUTIVE COMMITTEE President: Frank Ettensohn President-elect: Charlie Mason Vice President: Trent
Vertebrates. skull ribs vertebral column
 Vertebrates skull ribs vertebral column endoskeleton in cells working together tissues tissues working together organs working together organs systems Blood carries oxygen to the cells carries nutrients
Vertebrates skull ribs vertebral column endoskeleton in cells working together tissues tissues working together organs working together organs systems Blood carries oxygen to the cells carries nutrients
Lecture 11 Wednesday, September 19, 2012
 Lecture 11 Wednesday, September 19, 2012 Phylogenetic tree (phylogeny) Darwin and classification: In the Origin, Darwin said that descent from a common ancestral species could explain why the Linnaean
Lecture 11 Wednesday, September 19, 2012 Phylogenetic tree (phylogeny) Darwin and classification: In the Origin, Darwin said that descent from a common ancestral species could explain why the Linnaean
CHAPTER 26. Animal Evolution The Vertebrates
 CHAPTER 26 Animal Evolution The Vertebrates Impacts, Issues: Interpreting and Misinterpreting the Past No one was around to witness the transitions in the history of life Fossils allow us glimpses into
CHAPTER 26 Animal Evolution The Vertebrates Impacts, Issues: Interpreting and Misinterpreting the Past No one was around to witness the transitions in the history of life Fossils allow us glimpses into
LABORATORY #10 -- BIOL 111 Taxonomy, Phylogeny & Diversity
 LABORATORY #10 -- BIOL 111 Taxonomy, Phylogeny & Diversity Scientific Names ( Taxonomy ) Most organisms have familiar names, such as the red maple or the brown-headed cowbird. However, these familiar names
LABORATORY #10 -- BIOL 111 Taxonomy, Phylogeny & Diversity Scientific Names ( Taxonomy ) Most organisms have familiar names, such as the red maple or the brown-headed cowbird. However, these familiar names
Mesozoic Marine Life Invertebrate Vertebrate
 Mesozoic Marine Life Invertebrate Vertebrate Cenozoic Marine Life - Invertebrates (Mollusks) Cenozoic Marine Life - Invertebrates (Arthropods) Cenozoic Marine Life - Vertebrates Marine fossils are abundant
Mesozoic Marine Life Invertebrate Vertebrate Cenozoic Marine Life - Invertebrates (Mollusks) Cenozoic Marine Life - Invertebrates (Arthropods) Cenozoic Marine Life - Vertebrates Marine fossils are abundant
TUSKS! Exhibit Guide
 TUSKS! Exhibit Guide Assembling the Guide The pages of this guide are meant to be glued or photocopied back to back in the following order: Page 32/1 (facing down) with Page 2/31 (facing up) Page 30/3
TUSKS! Exhibit Guide Assembling the Guide The pages of this guide are meant to be glued or photocopied back to back in the following order: Page 32/1 (facing down) with Page 2/31 (facing up) Page 30/3
ALFRED GILLETT AND FOSSILS FROM STREET
 ALFRED GILLETT AND FOSSILS FROM STREET This collection of local fossils was formerly in the Crispin Hall, Street. Most of these fossils came from Alfred Gillett (1814-1904), a retired ironmonger who lived
ALFRED GILLETT AND FOSSILS FROM STREET This collection of local fossils was formerly in the Crispin Hall, Street. Most of these fossils came from Alfred Gillett (1814-1904), a retired ironmonger who lived
LABORATORY EXERCISE 7: CLADISTICS I
 Biology 4415/5415 Evolution LABORATORY EXERCISE 7: CLADISTICS I Take a group of organisms. Let s use five: a lungfish, a frog, a crocodile, a flamingo, and a human. How to reconstruct their relationships?
Biology 4415/5415 Evolution LABORATORY EXERCISE 7: CLADISTICS I Take a group of organisms. Let s use five: a lungfish, a frog, a crocodile, a flamingo, and a human. How to reconstruct their relationships?
Resources. Visual Concepts. Chapter Presentation. Copyright by Holt, Rinehart and Winston. All rights reserved.
 Chapter Presentation Visual Concepts Transparencies Standardized Test Prep Introduction to Vertebrates Table of Contents Section 1 Vertebrates in the Sea and on Land Section 2 Terrestrial Vertebrates Section
Chapter Presentation Visual Concepts Transparencies Standardized Test Prep Introduction to Vertebrates Table of Contents Section 1 Vertebrates in the Sea and on Land Section 2 Terrestrial Vertebrates Section
Activity Three: The Mystery Fossil Bones Activity
 Activity Three: The Mystery Fossil Bones Activity This was one of my favorites. I often used this as a culminating activity for my Geo. History Unit. Students from 9th - 12th loved it. And I m sure middle
Activity Three: The Mystery Fossil Bones Activity This was one of my favorites. I often used this as a culminating activity for my Geo. History Unit. Students from 9th - 12th loved it. And I m sure middle
Animal Diversity III: Mollusca and Deuterostomes
 Animal Diversity III: Mollusca and Deuterostomes Objectives: Be able to identify specimens from the main groups of Mollusca and Echinodermata. Be able to distinguish between the bilateral symmetry on a
Animal Diversity III: Mollusca and Deuterostomes Objectives: Be able to identify specimens from the main groups of Mollusca and Echinodermata. Be able to distinguish between the bilateral symmetry on a
Arizona s Raptor Experience, LLC November 2017 ~Newsletter~
 Arizona s Raptor Experience, LLC November 2017 ~Newsletter~ Greetings from Chino Valley! We hope you are well. This month s issue of our newsletter will focus on topics inspired by special days in November
Arizona s Raptor Experience, LLC November 2017 ~Newsletter~ Greetings from Chino Valley! We hope you are well. This month s issue of our newsletter will focus on topics inspired by special days in November
REPTILES. Scientific Classification of Reptiles To creep. Kingdom: Animalia Phylum: Chordata Subphylum: Vertebrata Class: Reptilia
 Scientific Classification of Reptiles To creep Kingdom: Animalia Phylum: Chordata Subphylum: Vertebrata Class: Reptilia REPTILES tetrapods - 4 legs adapted for land, hip/girdle Amniotes - animals whose
Scientific Classification of Reptiles To creep Kingdom: Animalia Phylum: Chordata Subphylum: Vertebrata Class: Reptilia REPTILES tetrapods - 4 legs adapted for land, hip/girdle Amniotes - animals whose
Evolution by Natural Selection
 Evolution by Natural Selection 2006-2007 DOCTRINE TINTORETTO The Creation of the Animals 1550 But the Fossil record OBSERVATION Anaerobic Bacteria Photosynthetic Bacteria Dinosaurs Green Algae Multicellular
Evolution by Natural Selection 2006-2007 DOCTRINE TINTORETTO The Creation of the Animals 1550 But the Fossil record OBSERVATION Anaerobic Bacteria Photosynthetic Bacteria Dinosaurs Green Algae Multicellular
1 Describe the anatomy and function of the turtle shell. 2 Describe respiration in turtles. How does the shell affect respiration?
 GVZ 2017 Practice Questions Set 1 Test 3 1 Describe the anatomy and function of the turtle shell. 2 Describe respiration in turtles. How does the shell affect respiration? 3 According to the most recent
GVZ 2017 Practice Questions Set 1 Test 3 1 Describe the anatomy and function of the turtle shell. 2 Describe respiration in turtles. How does the shell affect respiration? 3 According to the most recent
Evolution by Natural Selection
 Evolution by Natural Selection 2006-2007 DOCTRINE But the Fossil record OBSERVATION Quaternary 1.5 Tertiary 63 Cretaceous 135 Jurassic 180 Triassic 225 Permian 280 Carboniferous 350 Devonian 400 Silurian
Evolution by Natural Selection 2006-2007 DOCTRINE But the Fossil record OBSERVATION Quaternary 1.5 Tertiary 63 Cretaceous 135 Jurassic 180 Triassic 225 Permian 280 Carboniferous 350 Devonian 400 Silurian
Differences between Reptiles and Mammals. Reptiles. Mammals. No milk. Milk. Small brain case Jaw contains more than one bone Simple teeth
 Differences between Reptiles and Mammals Reptiles No milk Mammals Milk The Advantage of Being a Furball: Diversification of Mammals Small brain case Jaw contains more than one bone Simple teeth One ear
Differences between Reptiles and Mammals Reptiles No milk Mammals Milk The Advantage of Being a Furball: Diversification of Mammals Small brain case Jaw contains more than one bone Simple teeth One ear
Your web browser (Safari 7) is out of date. For more security, comfort and the best experience on this site: Update your browser Ignore
 Your web browser (Safari 7) is out of date. For more security, comfort and the best experience on this site: Update your browser Ignore Activityengage HU NTERS IN THE AIR What characteristics helped pterosaurs
Your web browser (Safari 7) is out of date. For more security, comfort and the best experience on this site: Update your browser Ignore Activityengage HU NTERS IN THE AIR What characteristics helped pterosaurs
Animal Diversity wrap-up Lecture 9 Winter 2014
 Animal Diversity wrap-up Lecture 9 Winter 2014 1 Animal phylogeny based on morphology & development Fig. 32.10 2 Animal phylogeny based on molecular data Fig. 32.11 New Clades 3 Lophotrochozoa Lophophore:
Animal Diversity wrap-up Lecture 9 Winter 2014 1 Animal phylogeny based on morphology & development Fig. 32.10 2 Animal phylogeny based on molecular data Fig. 32.11 New Clades 3 Lophotrochozoa Lophophore:
HONR219D Due 3/29/16 Homework VI
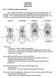 Part 1: Yet More Vertebrate Anatomy!!! HONR219D Due 3/29/16 Homework VI Part 1 builds on homework V by examining the skull in even greater detail. We start with the some of the important bones (thankfully
Part 1: Yet More Vertebrate Anatomy!!! HONR219D Due 3/29/16 Homework VI Part 1 builds on homework V by examining the skull in even greater detail. We start with the some of the important bones (thankfully
The Descendants WOMG. Is a newfound prehistoric species our direct ancestor?
 Name By Bobby Oerzen AUSTRALOPtTHBCUS AFAftBN&S (Lucy) 5,3 rsm>{;o yust, $
Name By Bobby Oerzen AUSTRALOPtTHBCUS AFAftBN&S (Lucy) 5,3 rsm>{;o yust, $
L E T 'S L E T 'S L O O K L E T 'S L O O K LOOK
 L E T 'S L O O K Roar! Roar! L E T 'S L O O K Dinosaurs DK Publishing, Inc. Dinosaur bones Dinosaurs lived long ago before there were people. Scientists look at their bones to learn about them. tail leg
L E T 'S L O O K Roar! Roar! L E T 'S L O O K Dinosaurs DK Publishing, Inc. Dinosaur bones Dinosaurs lived long ago before there were people. Scientists look at their bones to learn about them. tail leg
Where Animals and Plants Are Found
 Section 8: Physical Systems Where Animals and Plants Are Found About Animals and Plants What I Need to Know Vocabulary ecosystem food chain food web marine prairie Many animals live on Earth. Many plants
Section 8: Physical Systems Where Animals and Plants Are Found About Animals and Plants What I Need to Know Vocabulary ecosystem food chain food web marine prairie Many animals live on Earth. Many plants
AP Biology. AP Biology
 Evolution by Natural Selection 2006-2007 DOCTRINE TINTORETTO The Creation of the Animals 1550 But the Fossil record OBSERVATION mya Quaternary 1.5 Tertiary 63 Cretaceous 135 Jurassic 180 Triassic 225 Permian
Evolution by Natural Selection 2006-2007 DOCTRINE TINTORETTO The Creation of the Animals 1550 But the Fossil record OBSERVATION mya Quaternary 1.5 Tertiary 63 Cretaceous 135 Jurassic 180 Triassic 225 Permian
Characteristics of a Reptile. Vertebrate animals Lungs Scaly skin Amniotic egg
 Reptiles Characteristics of a Reptile Vertebrate animals Lungs Scaly skin Amniotic egg Characteristics of Reptiles Adaptations to life on land More efficient lungs and a better circulator system were develope
Reptiles Characteristics of a Reptile Vertebrate animals Lungs Scaly skin Amniotic egg Characteristics of Reptiles Adaptations to life on land More efficient lungs and a better circulator system were develope
Shedding Light on the Dinosaur-Bird Connection
 Shedding Light on the Dinosaur-Bird Connection This text is provided courtesy of the American Museum of Natural History. When people think of dinosaurs, two types generally come to mind: the huge herbivores
Shedding Light on the Dinosaur-Bird Connection This text is provided courtesy of the American Museum of Natural History. When people think of dinosaurs, two types generally come to mind: the huge herbivores
Non-fiction: The Descendants
 Non-fiction:The Descendants The Descendants By Bobby Oerzen Is a newfound prehistoric species our direct ancestor? Matthew Berger wasn t looking to revise the story of human origins. He was just chasing
Non-fiction:The Descendants The Descendants By Bobby Oerzen Is a newfound prehistoric species our direct ancestor? Matthew Berger wasn t looking to revise the story of human origins. He was just chasing
Evidence for Evolution by Natural Selection. Hunting for evolution clues Elementary, my dear, Darwin!
 Evidence for Evolution by Natural Selection Hunting for evolution clues Elementary, my dear, Darwin! 2006-2007 Evidence supporting evolution Fossil record shows change over time Anatomical record comparing
Evidence for Evolution by Natural Selection Hunting for evolution clues Elementary, my dear, Darwin! 2006-2007 Evidence supporting evolution Fossil record shows change over time Anatomical record comparing
May 10, SWBAT analyze and evaluate the scientific evidence provided by the fossil record.
 May 10, 2017 Aims: SWBAT analyze and evaluate the scientific evidence provided by the fossil record. Agenda 1. Do Now 2. Class Notes 3. Guided Practice 4. Independent Practice 5. Practicing our AIMS: E.3-Examining
May 10, 2017 Aims: SWBAT analyze and evaluate the scientific evidence provided by the fossil record. Agenda 1. Do Now 2. Class Notes 3. Guided Practice 4. Independent Practice 5. Practicing our AIMS: E.3-Examining
Lab 2 Skeletons and Locomotion
 Lab 2 Skeletons and Locomotion Objectives The objectives of this and next week's labs are to introduce you to the comparative skeletal anatomy of vertebrates. As you examine the skeleton of each lineage,
Lab 2 Skeletons and Locomotion Objectives The objectives of this and next week's labs are to introduce you to the comparative skeletal anatomy of vertebrates. As you examine the skeleton of each lineage,
Biology Slide 1 of 50
 Biology 1 of 50 2 of 50 What Is a Reptile? What are the characteristics of reptiles? 3 of 50 What Is a Reptile? What Is a Reptile? A reptile is a vertebrate that has dry, scaly skin, lungs, and terrestrial
Biology 1 of 50 2 of 50 What Is a Reptile? What are the characteristics of reptiles? 3 of 50 What Is a Reptile? What Is a Reptile? A reptile is a vertebrate that has dry, scaly skin, lungs, and terrestrial
Ceri Pennington VELOCIRAPTOR
 Ceri Pennington VELOCIRAPTOR The Velociraptor - meaning swift seizer - lived during the late Cretaceous period - 75-71 million years ago. They were a genus of dromaeosaurid theropod dinosaur and there
Ceri Pennington VELOCIRAPTOR The Velociraptor - meaning swift seizer - lived during the late Cretaceous period - 75-71 million years ago. They were a genus of dromaeosaurid theropod dinosaur and there
Ch 34: Vertebrate Objective Questions & Diagrams
 Ch 34: Vertebrate Objective Questions & Diagrams Invertebrate Chordates and the Origin of Vertebrates 1. Distinguish between the two subgroups of deuterostomes. 2. Describe the four unique characteristics
Ch 34: Vertebrate Objective Questions & Diagrams Invertebrate Chordates and the Origin of Vertebrates 1. Distinguish between the two subgroups of deuterostomes. 2. Describe the four unique characteristics
Name: GEOL 104 Dinosaurs: A Natural History Video Assignment. DUE: Wed. Oct. 20
 GEOL 104 Dinosaurs: A Natural History Video Assignment DUE: Wed. Oct. 20 Documentaries represent one of the main media by which scientific information reaches the general public. For this assignment, you
GEOL 104 Dinosaurs: A Natural History Video Assignment DUE: Wed. Oct. 20 Documentaries represent one of the main media by which scientific information reaches the general public. For this assignment, you
Evolution on Exhibit Hints for Teachers
 1 Evolution on Exhibit Hints for Teachers This gallery activity explores a variety of evolution themes that are well illustrated by gallery specimens and exhibits. Each activity is aligned with the NGSS
1 Evolution on Exhibit Hints for Teachers This gallery activity explores a variety of evolution themes that are well illustrated by gallery specimens and exhibits. Each activity is aligned with the NGSS
Geo 302D: Age of Dinosaurs. LAB 7: Dinosaur diversity- Saurischians
 Geo 302D: Age of Dinosaurs LAB 7: Dinosaur diversity- Saurischians Last lab you were presented with a review of major ornithischian clades. You also were presented with some of the kinds of plants that
Geo 302D: Age of Dinosaurs LAB 7: Dinosaur diversity- Saurischians Last lab you were presented with a review of major ornithischian clades. You also were presented with some of the kinds of plants that
Family Groups 1. a) b) c) d) e) f) g) h) i)
 Family Groups Dinosaurs evolved from the class of backboned animals called Reptiles. They are split into two major groups (orders) based on the structure of their pelvis (hip bone). These groups are then
Family Groups Dinosaurs evolved from the class of backboned animals called Reptiles. They are split into two major groups (orders) based on the structure of their pelvis (hip bone). These groups are then
Sample Questions: EXAMINATION I Form A Mammalogy -EEOB 625. Name Composite of previous Examinations
 Sample Questions: EXAMINATION I Form A Mammalogy -EEOB 625 Name Composite of previous Examinations Part I. Define or describe only 5 of the following 6 words - 15 points (3 each). If you define all 6,
Sample Questions: EXAMINATION I Form A Mammalogy -EEOB 625 Name Composite of previous Examinations Part I. Define or describe only 5 of the following 6 words - 15 points (3 each). If you define all 6,
What is evolution? Transitional fossils: evidence for evolution. In its broadest sense, evolution is simply the change in life through time.
 Transitional fossils: evidence for evolution http://domain- of- darwin.deviantart.com/art/no- Transitional- Fossils- 52231284 Western MA Atheists and Secular Humanists 28 May 2016 What is evolution? In
Transitional fossils: evidence for evolution http://domain- of- darwin.deviantart.com/art/no- Transitional- Fossils- 52231284 Western MA Atheists and Secular Humanists 28 May 2016 What is evolution? In
Evolution. Evolution is change in organisms over time. Evolution does not have a goal; it is often shaped by natural selection (see below).
 Evolution Evolution is change in organisms over time. Evolution does not have a goal; it is often shaped by natural selection (see below). Species an interbreeding population of organisms that can produce
Evolution Evolution is change in organisms over time. Evolution does not have a goal; it is often shaped by natural selection (see below). Species an interbreeding population of organisms that can produce
Chapter 22 Darwin and Evolution by Natural Selection
 Anaerobic Bacteria Photosynthetic Bacteria Dinosaurs Green Algae Multicellular Animals Flowering Molluscs Arthropods Chordates Jawless Fish Teleost Fish Amphibians Insects Reptiles Mammals Birds Land Plants
Anaerobic Bacteria Photosynthetic Bacteria Dinosaurs Green Algae Multicellular Animals Flowering Molluscs Arthropods Chordates Jawless Fish Teleost Fish Amphibians Insects Reptiles Mammals Birds Land Plants
Biology. Slide 1of 50. End Show. Copyright Pearson Prentice Hall
 Biology 1of 50 2of 50 Phylogeny of Chordates Nonvertebrate chordates Jawless fishes Sharks & their relatives Bony fishes Reptiles Amphibians Birds Mammals Invertebrate ancestor 3of 50 A vertebrate dry,
Biology 1of 50 2of 50 Phylogeny of Chordates Nonvertebrate chordates Jawless fishes Sharks & their relatives Bony fishes Reptiles Amphibians Birds Mammals Invertebrate ancestor 3of 50 A vertebrate dry,
Introduction to Biological Anthropology: Notes 23 A world full of Plio-pleistocene hominins Copyright Bruce Owen 2011 Let s look at the next chunk of
 Introduction to Biological Anthropology: Notes 23 A world full of Plio-pleistocene hominins Copyright Bruce Owen 2011 Let s look at the next chunk of time: 3.0 1.0 mya often called the Plio-pleistocene
Introduction to Biological Anthropology: Notes 23 A world full of Plio-pleistocene hominins Copyright Bruce Owen 2011 Let s look at the next chunk of time: 3.0 1.0 mya often called the Plio-pleistocene
DEUTEROSTOMES. This presentation contains copyrighted material under the educational fair use exemption to the U.S. copyright law.
 DEUTEROSTOMES This presentation contains copyrighted material under the educational fair use exemption to the U.S. copyright law. Deuterostome Echinodermata body plan! Body plan! Larvae are bilateral!
DEUTEROSTOMES This presentation contains copyrighted material under the educational fair use exemption to the U.S. copyright law. Deuterostome Echinodermata body plan! Body plan! Larvae are bilateral!
A R T I C L E S STRATIGRAPHIC DISTRIBUTION OF VERTEBRATE FOSSIL FOOTPRINTS COMPARED WITH BODY FOSSILS
 A R T I C L E S STRATIGRAPHIC DISTRIBUTION OF VERTEBRATE FOSSIL FOOTPRINTS COMPARED WITH BODY FOSSILS Leonard Brand & James Florence Department of Biology Loma Linda University WHAT THIS ARTICLE IS ABOUT
A R T I C L E S STRATIGRAPHIC DISTRIBUTION OF VERTEBRATE FOSSIL FOOTPRINTS COMPARED WITH BODY FOSSILS Leonard Brand & James Florence Department of Biology Loma Linda University WHAT THIS ARTICLE IS ABOUT
BY DINO DON LESSEM ILLUSTRATIONS BY JOHN BINDON. a LERNER PUBLICATIONS COMPANY / MINNEAPOLIS
 BY DINO DON LESSEM ILLUSTRATIONS BY JOHN BINDON a LERNER PUBLICATIONS COMPANY / MINNEAPOLIS To Brian Joseph, my first friend, who has nothing whatsoever to do with anything in this book Text copyright
BY DINO DON LESSEM ILLUSTRATIONS BY JOHN BINDON a LERNER PUBLICATIONS COMPANY / MINNEAPOLIS To Brian Joseph, my first friend, who has nothing whatsoever to do with anything in this book Text copyright
Title: Phylogenetic Methods and Vertebrate Phylogeny
 Title: Phylogenetic Methods and Vertebrate Phylogeny Central Question: How can evolutionary relationships be determined objectively? Sub-questions: 1. What affect does the selection of the outgroup have
Title: Phylogenetic Methods and Vertebrate Phylogeny Central Question: How can evolutionary relationships be determined objectively? Sub-questions: 1. What affect does the selection of the outgroup have
Carnivore An animal that feeds chiefly on the flesh of other animals.
 Name: School: Date: Bipedalism A form of terrestrial locomotion where an organism moves by means of its two rear limbs, or legs. An animal that usually moves in a bipedal manner is known as a biped, meaning
Name: School: Date: Bipedalism A form of terrestrial locomotion where an organism moves by means of its two rear limbs, or legs. An animal that usually moves in a bipedal manner is known as a biped, meaning
Dinosaurs. Lesson 1 Amazing dinosaurs. 1 Talk about it What do you know about dinosaurs?
 6 Dinosaurs We re going to: ask and answer questions about dinosaurs talk about time and dates describe and compare dinosaurs read about and discuss dinosaur discoveries Lesson 1 Amazing dinosaurs 1 Talk
6 Dinosaurs We re going to: ask and answer questions about dinosaurs talk about time and dates describe and compare dinosaurs read about and discuss dinosaur discoveries Lesson 1 Amazing dinosaurs 1 Talk
